Achieving Ah‑Level Zn–MnO2 Pouch Cells via Interfacial Solvation Structure Engineering
As the demand for safe, cost-effective energy storage solutions continues to grow, the limitations of traditional lithium-ion batteries in terms of safety and resource scarcity become more pronounced. Now, researchers from Zhejiang Wanli University, University of Waterloo, and Dalian Institute of Chemical Physics, led by Professor Xin Wang, Professor Aiping Yu, and Professor Dan Luo, have presented a breakthrough study on interfacial solvation engineering for aqueous zinc-ion batteries. This work offers valuable insights into the development of next-generation energy storage technologies that can overcome these limitations.
Why Interfacial Solvation Engineering Matters
- Enhanced Stability: Tailoring the interfacial solvation structure can significantly suppress hydrogen evolution and corrosion at the Zn anode, addressing the critical stability challenges in aqueous electrolytes.
- Rapid Desolvation Kinetics: By constructing a low-coordination Zn2+ solvation shell at the interface, the energy barrier for Zn2+ desolvation is reduced, enabling faster and more uniform Zn deposition.
- Practical Applications: The strategy enables the fabrication of high-capacity pouch cells (1.5 Ah) with long cycling stability, bridging the gap between laboratory research and commercial viability.
Innovative Design and Features
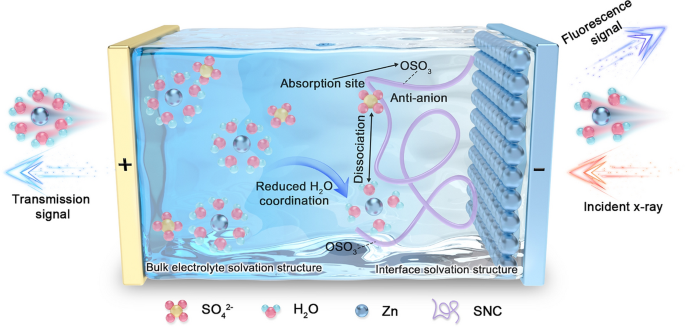
- Sulfated Nanocellulose Additive: The study introduces sulfated nanocellulose (SNC), an anion-rich biopolymer, to tailor the interfacial solvation structure without altering the bulk electrolyte composition. SNC forms a liquid/solid hybrid layer at the Zn anode interface, reducing interfacial water activity.
- Advanced Characterization Techniques: In-situ attenuated total reflection Fourier transform infrared (ATR-FTIR) spectroscopy and fluorescence interface-extended X-ray absorption fine structure (FI-EXAFS) were employed to reveal the formation of a low-coordination Zn2+solvation shell at the interface, confirming the mechanism of interfacial water reduction.
- Mechanism Insights: Electrostatic interactions between Zn2+ and anchored sulfate groups weaken H2O coordination, facilitating rapid desolvation kinetics and enhancing interfacial stability during cycling.
Applications and Future Outlook
- High Coulombic Efficiency: The SNC-induced aqueous zinc-ion batteries achieved a high average coulombic efficiency of 99.6% over 500 cycles in Zn|Ti asymmetric cells at 5 mA cm-2.
- Long-Term Cycling Stability: Zn|Zn symmetric cells exhibited superb cycling stability for over 1300 hours at 1 mA cm-2 and 1 mAh cm-2, and over 1000 hours even at a high current density of 5 mA cm-2.
- Practical Pouch Cell Performance: A 1.5 Ah Zn|MnO2pouch cell with a mass loading of 13.4 mg cm-2 remained stable over 250 cycles with 80% capacity retention, demonstrating the practical potential of this interfacial engineering strategy.
- Full Cell Enhancement: Zn|MnO2/CNT full cells using the optimized electrolyte maintained a steady coulombic efficiency of around 100% and possessed long-lasting stability (90.7% retention for over 1000 cycles).
- Challenges and Opportunities: The study highlights the importance of interfacial solvation structure engineering beyond traditional bulk electrolyte design. Future research will focus on further optimizing interfacial layers and exploring new biopolymer additives to fully realize the potential of aqueous zinc-ion batteries in large-scale energy storage.
This comprehensive study provides a roadmap for the development and application of interfacial solvation engineering in aqueous zinc-ion batteries. It highlights the importance of interdisciplinary research in materials science, electrochemistry, and advanced characterization techniques to drive innovation in this field. Stay tuned for more groundbreaking work from Professor Xin Wang, Professor Aiping Yu, and Professor Dan Luo and their teams!
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in