Bidirectionally Enhanced Reaction Kinetics in Vanadium Redox Flow Battery via Regulating Mixed‑Valence States in Perovskite Electrodes
As the climate crisis grows increasingly severe, the optimization of energy structure through renewable resources has emerged as a practical and effective national strategy. However, the inherent intermittent and fluctuating nature of renewable energy constitutes the primary technical bottleneck in the energy conversion process. Now, researchers from the School of Chemical Engineering at North China University of Science and Technology, led by Professor Zhangxing He, in collaboration with Queensland University of Technology and Tianjin University, have presented a breakthrough study on bidirectionally enhanced reaction kinetics in vanadium redox flow battery (VRFB) via regulating mixed-valence states in perovskite. This work offers valuable insights into the development of next-generation high-performance metal-based electrocatalysts for large-scale energy storage.
Why Bidirectional Electronic Structure Engineering Matters
- Energy Storage Efficiency: The VRFB is a highly acclaimed large-scale energy storage system owing to its brilliant features, including decoupled capacity and power, long cycle life, and excellent safety.
- Reaction Kinetic Limitations: The insufficient active sites and weak hydrophilicity of carbon electrodes hinder the diffusion and charge transfer of vanadium ions during cell operation, limiting overall battery performance.
- Cost-Effective Solution: Electrode modification offers a relatively low-cost approach to enhancing battery performance compared with the significant expense of developing new electrolytes or membranes.
Innovative Design and Features
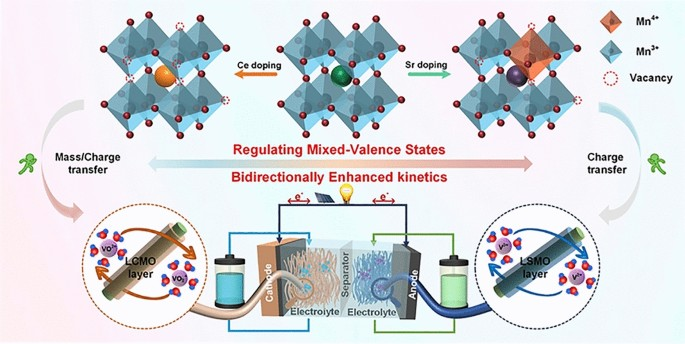
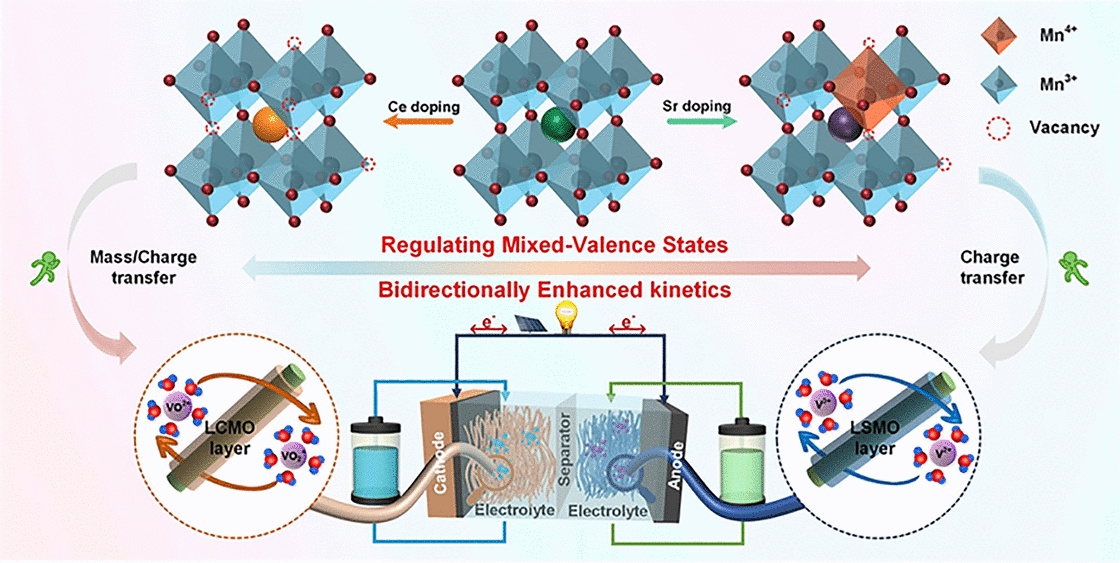
- Selective Doping Strategy: The study employs rational doping of Sr and Ce at the La site of LaMnO3 perovskite (LSMO and LCMO) to modulate chemical environments of Mn ion activity donors, thereby boosting vanadium redox reaction processes.
- Bidirectional Enhancement Mechanism: Sr doping increases the valence state of Mn ions, facilitating electron transfer to V3+ ions and lowering the reaction energy barrier of V3+/V2+ redox processes. Conversely, Ce doping decreases the Mn valence and increases oxygen vacancies, boosting charge transfer and mass transfer of VO2+/VO2+ redox processes.
- Structural Characterization: XRD, SEM, TEM, XPS, and Raman spectroscopy confirm the successful incorporation of Sr and Ce into the perovskite lattice, with uniform element distribution and tunable Mn3+/Mn4+ ratios.
Applications and Future Outlook
- Enhanced Electrochemical Performance: The VRFB with LSMO and LCMO-modified anode and cathode exhibits excellent energy efficiency (EE) of 67% at a high current density of 300 mA cm-2 and an increased EE of 15% at 150 mA cm-2 compared with pristine VRFB.
- Superior Power Density: The modified VRFB delivers a highest power of 233.3 mW cm-2 at 225 mA cm-2, significantly outperforming the pristine VRFB (172 mW cm-2 at 150 mA cm-2).
- Long-Term Stability: The LSMO/LCMO VRFB maintains stable performance over 500 cycles with a capacity decay rate of only 0.0216%, while the pristine VRFB fails after 126 cycles.
- Theoretical Validation: Density functional theory calculations demonstrate that doping Sr and Ce enhances the vanadium ion's ability for charge transfer and adsorption, corroborating the experimental findings.
- Challenges and Opportunities: The study highlights the importance of precise electronic structure control for designing bidirectional electrocatalysts. Future research will focus on scaling up the synthesis and exploring other perovskite compositions for broader flow battery applications.
This comprehensive study provides a roadmap for the development of high-performance metal-based electrocatalysts in VRFB through bidirectional electronic structure engineering. It highlights the importance of interdisciplinary research in materials science, electrochemistry, and computational modeling to drive innovation in energy storage technologies. Stay tuned for more groundbreaking work from Professor Zhangxing He at North China University of Science and Technology!
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in