Biohybrid Nanocatalysts Unlock Efficient C–C Bond Chemistry
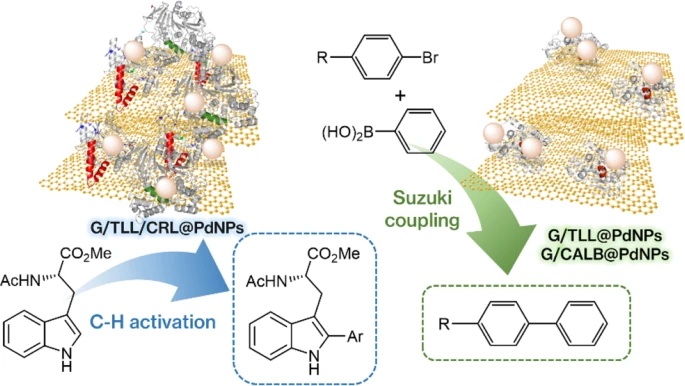
We report heterogeneous enzyme–metal nanoparticle biohybrids prepared by in‑situ growth of Pd and Cu nanoparticles on lipases immobilized over few‑layer graphene, enabling selective and efficient C–H activation and Suzuki–Miyaura coupling under mild, green conditions. By leveraging enzyme scaffolds to control nanoparticle size (≈2–3 nm) and metal speciation (Pd⁰/PdO), we achieve high activity and selectivity in aqueous and organic media at or near room temperature.
Key Highlights
- Hybrid catalyst platform using graphene‑immobilized lipases (CALB, TLL, CRL) to direct the formation of Pd and Pd/Cu nanoparticles.
- Controlled nanoparticle size and oxidation state through enzyme identity and immobilization strategy.
- Dual‑enzyme Pd hybrid achieves >95% yield in selective C‑2 C–H arylation at room temperature, with TOF up to 5000 h⁻¹ (surface‑normalized).
- Pd⁰‑rich single‑enzyme systems show optimal performance in Suzuki–Miyaura cross‑coupling in aqueous media.
- Catalysts are stable, recyclable, and accessible via a simple one‑pot aqueous synthesis.
Significance
This work establishes a scalable and versatile strategy for designing artificial metalloenzyme‑like catalysts in which enzymes function as scaffolds, reductants, and stabilizers of metal nanoparticles. Fixation of enzymes in an open conformation on graphene enables full accessibility of metal active sites and overcomes long‑standing compatibility challenges between biocatalysis and metal catalysis. The demonstrated ability to rationally tune nanoparticle size and oxidation state provides a powerful framework for sustainable and selective C–C bond formation relevant to pharmaceutical and fine‑chemical synthesis.
Authors
Noelia Losada‑Garcia; Clara Ortega‑Nieto; Jose M. Palomo*
Instituto de Catálisis y Petroleoquímica (ICP), CSIC, Madrid, Spain
Corresponding author email: josempalomo@icp.csic.es
Follow the Topic
-
Catal
Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in