Color nonlinear microscopy probes red blood cells and oxygenation
Published in Electrical & Electronic Engineering

Biological tissues and organisms rely on red blood cells (RBCs) for oxygen supply. Precise analyses of tissue metabolism require to map hemodynamics and blood oxygenation at high resolution. Reference microscopy methods for the purpose, based on two-photon excitation, require the injection of exogenous labels and are often limited in temporal resolution and imaging depth. In this work, we introduce a novel label-free imaging method which holds potential to address these limitations.
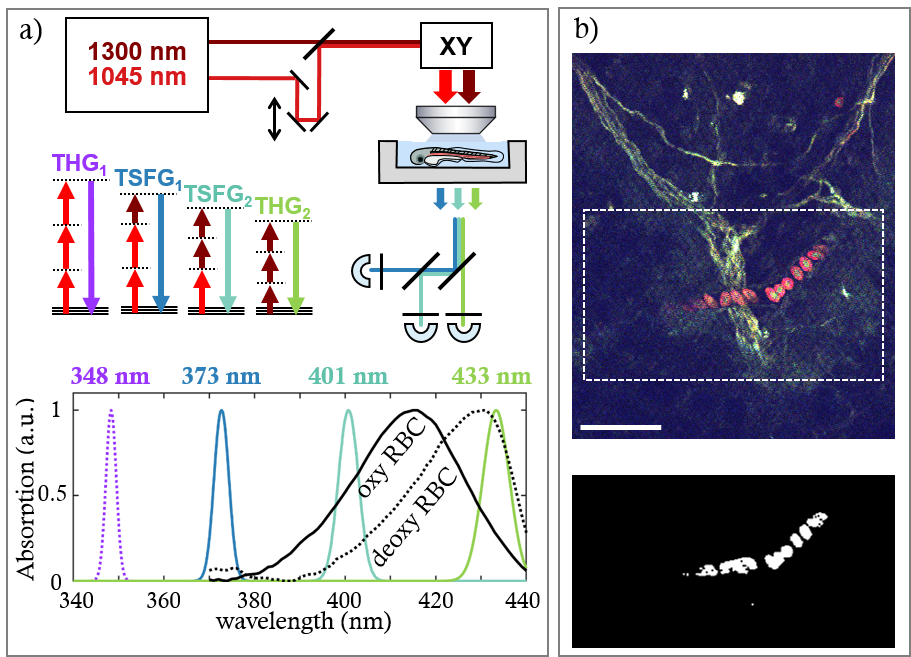
Our method, termed color third-order sum frequency generation (color TSFG), relies on the mixing of two infrared femtosecond laser pulse trains in the sample, and the simultaneous detection of several third-order signals resulting from third-harmonic generation (THG) and TSFG processes (Figure 1a).
THG microscopy highlights interfaces and optical heterogeneity in biological samples, and is in generally primarily used to reveal tissue morphology, in complement to fluorescence imaging of specifically labelled structures. However, a less used property of THG imaging is that the signal can be resonantly enhanced by the presence of strong absorbers such as hemoglobin, the oxygen-carrying molecule in RBCs.
In our study, we first characterized the spectral dependence of the THG and TSFG signals from isolated human and zebrafish RBCs. We confirmed that THG and TSFG signals exhibit a strong resonance enhancement when emitted near the absorption peak of hemoglobin in the 415-430 nm region, known as the Soret band. Moreover, this three-photon resonance is spectrally shifted depending on the oxygenation state of RBCs.
We then showed that the spectral properties of color TSFG images can be used to detect RBC in live zebrafish embryos with high specificity. Indeed, computing the ratio between a resonance-enhanced image and a simultaneously acquired non-resonant image selectively highlights RBCs, which can then be automatically detected (Figure 1b).
We also found that the ratio between two images obtained at different wavelengths within the hemoglobin-resonant part of the THG spectrum can be used as an oxygenation probe. We used this property to probe RBC oxygenation changes in live zebrafish embryos with pixel times in the microsecond range.
Finally, we demonstrated that color TSFG can easily be implemented in a three-photon (3P) microscope using an optical parametric amplifier (OPA) laser source operating at 1 MHz. 3P microscopy is a recently developed multiphoton method providing superior imaging depth than 2P microscopy. We showed that color TSFG can provide label-free RBC-specific contrast at depths exceeding 600 µm in a live adult zebrafish brain.
In conclusion, our work introduce a new multiphoton contrast modality capable of probing RBCs distribution and oxygenation with high spatial and temporal resolution. Moreover it is directly compatible with 3P imaging, thus extending the palette of observable parameters in deep tissue microscopy.
Follow the Topic
-
Light: Science & Applications
A peer-reviewed open access journal publishing highest-quality articles across the full spectrum of optics research. LSA promotes frontier research in all areas of optics and photonics, including basic, applied, scientific and engineering results.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in