Crystalline Ni3S2 Nanorods Tuned by Low‑Crystalline NiCoSx with Built‑In Electric Field for Efficient Overall Water Splitting
Introduction: The Hydrogen Economy and Electrocatalytic Challenges
The transition toward a sustainable hydrogen economy relies heavily on the efficiency of water electrolysis. Electrolytic water splitting consists of two half-reactions: the Hydrogen Evolution Reaction (HER) and the Oxygen Evolution Reaction (OER). However, the large-scale deployment of this technology is hindered by the sluggish kinetics of these reactions, particularly the OER, and the high cost of noble-metal catalysts like Platinum and Iridium.
To overcome these barriers, researchers have focused on transition metal sulfides due to their earth-abundance and metallic conductivity. However, traditional sulfides often suffer from poor structural stability and limited active sites. A recent breakthrough published in Nano-Micro Letters by Professor Xiaojun He and Professor Jieshan Qiu presents a novel strategy: armoring crystalline Ni3S2 nanorods with low-crystalline NiCoS to create a high-performance, bifunctional electrocatalyst.
Architecture Design: The Amorphous-Crystalline Heterostructure
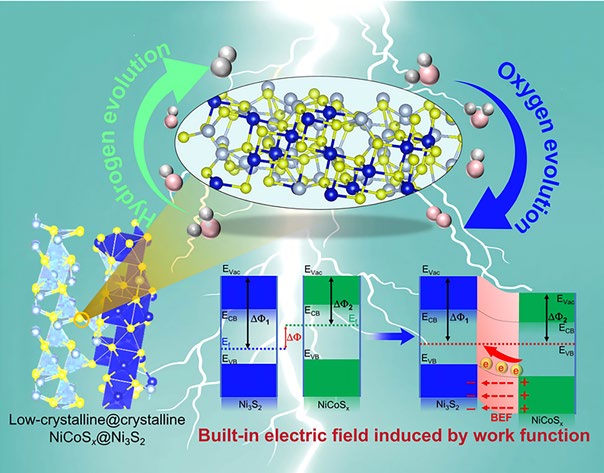
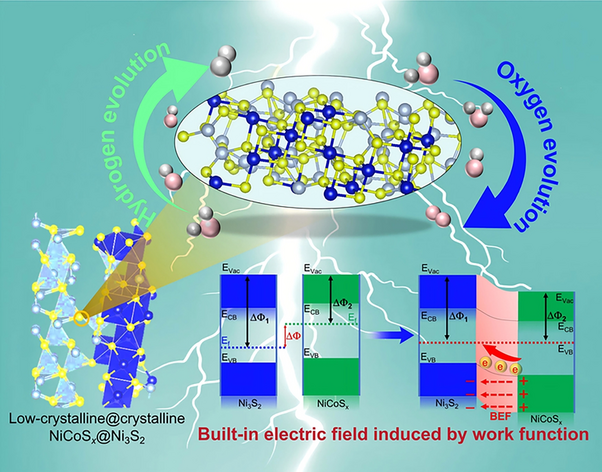
The core innovation of this research is the construction of a unique heterostructure on nickel foam (NF). The catalyst features a crystalline Ni3S2 nanorod core "armored" by a low-crystalline (amorphous-like) NiCoSx shell. This specific design addresses two fundamental issues in electrocatalysis:
- Preventing Sulfide Loss: One of the primary failure mechanisms of transition metal sulfides during the OER is the leaching of sulfur atoms, which leads to structural collapse. The low-crystalline NiCoSx layer acts as a protective shield, effectively inhibiting the loss of sulfide from the crystalline core and significantly extending the catalyst's operational lifespan.
- Creating Active Sites: Unlike perfectly ordered crystals, low-crystalline or amorphous materials are rich in defects, such as unsaturated coordination sites and grain boundaries. These defects serve as highly active centers for the adsorption of water molecules and intermediate species, lowering the overall energy barrier for the reaction.
The Power of the Built-in Electric Field (BIEF)
The most significant scientific contribution of this study is the exploration of the Built-in Electric Field (BIEF) at the heterostructure interface. By coupling crystalline Ni3S2 with low-crystalline NiCoSx, the researchers created a junction with a distinct difference in work functions.
This difference triggers a spontaneous redistribution of electrons at the interface, establishing a BIEF. This internal field acts as a "molecular accelerator," promoting faster electron transfer between the catalyst surface and the reactants. DFT calculations confirmed that the BIEF optimizes the adsorption energies of hydrogen and oxygen intermediates (such as H*, OOH*, and OH*), ensuring that they are neither too tightly nor too loosely bound to the surface—a condition known as the Sabatier principle.
Bifunctional Excellence in Water Splitting
The NiCoSx@Ni3S2/NF catalyst demonstrates exceptional bifunctionality, meaning it can efficiently catalyze both the HER and the OER in the same alkaline environment.
In the HER process, the heterostructure facilitates rapid water dissociation and hydrogen recombination. During the OER, the synergistic effect between Nickel and Cobalt sites within the NiCoSx shell enhances the formation of oxyhydroxide species, which are the true active phases for oxygen evolution. When integrated into a full-cell electrolyzer, this material allows for overall water splitting at low cell voltages, rivaling the performance of precious metal benchmarks.
Structural Stability and Durability
Beyond its activity, the catalyst exhibits remarkable durability. Electrocatalysts in industrial water splitting must withstand harsh alkaline conditions and high current densities for hundreds of hours. The Ni3S2 nanorods provide a stable conductive pathway, while the NiCoS "armor" maintains chemical integrity. Testing showed that the heterostructure maintains its morphology and electrochemical surface area even after prolonged periods of gas evolution, proving its potential for long-term industrial use.
Conclusion and Future Outlook
The development of the NiCoSx@Ni3S2 nanorod heterostructure provides a powerful blueprint for designing the next generation of water-splitting catalysts. By combining crystalline and low-crystalline phases, researchers can leverage the stability of the former and the reactivity of the latter.
The successful implementation of the Built-in Electric Field effect to modulate electronic structures opens new avenues for other energy-related reactions, such as the CO2 reduction reaction and nitrogen fixation. As the world pushes for "Green Hydrogen," these sophisticated, non-precious metal heterostructures will play a vital role in making sustainable energy affordable and accessible.
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in