Decoupled water electrolysis: Reshaping a century-old technology to scale up green hydrogen production
Published in Materials
Green hydrogen produced by water decomposition using renewable energies such as solar and wind is essential to reduce greenhouse gas emissions, especially in hard-to-abate industrial sectors such as steel, cement and ammonia production. However, commercial technologies for green hydrogen production such as alkaline and PEM electrolysis are uncompetitive with the low-cost production of grey hydrogen from fossil fuels that result in substantial greenhouse gas emissions. Reducing the electrolyzers' cost and improving their efficiency are essential for wide deployment of green hydrogen as part of the transition to Net Zero.
Conventional water electrolysis technologies present challenges to increase their performance, economic competitiveness, and deployment. These challenges result from the co-production of hydrogen and oxygen in the same electrolytic cell, as well as from substantial energy losses. To prevent hydrogen and oxygen mixing, and the resultant risk of explosion, the electrolytic cell is divided by a membrane or diaphragm into cathodic and anodic compartments (Figure 1a). This membrane architecture complicates the electrolyzers construction by adding gaskets and compression plates to prevent gas crossover across the anodic and cathodic compartments (Figure 1b). These components add substantial cost and weight and limits operation at high pressures and low-purity water that may foul the membranes. In addition, substantial energy losses, mostly due to the difficult oxygen evolution reaction that requires the exchange of four electrons and four protons in a single catalytic site to generate an O2 molecule, increase the cost of energy in this energy intensive technology.
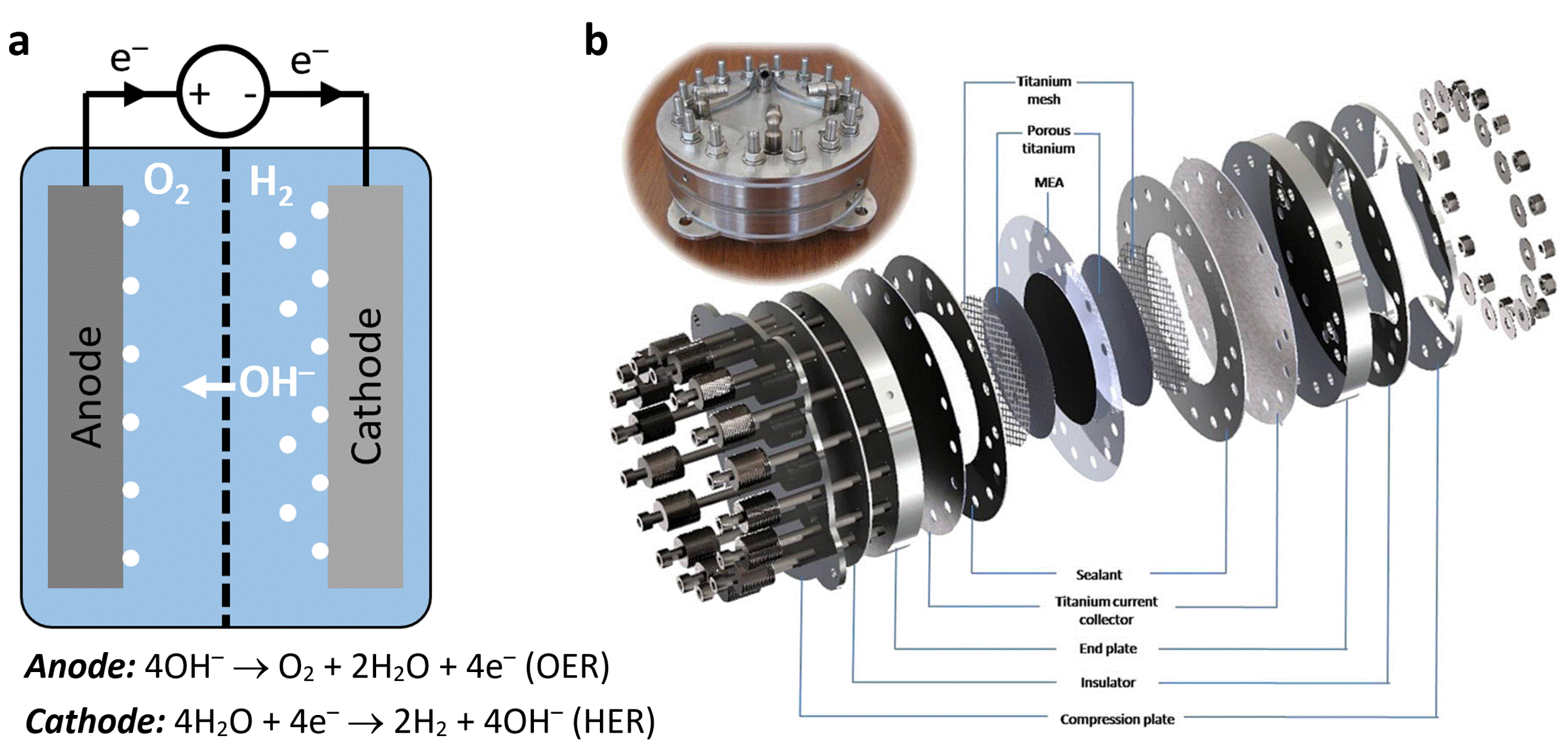
Figure 1: Conventional electrolyzer design. (a) Schematic illustration of the anodic and cathodic compartments in an alkaline electrolysis cell. The dashed line represents a membrane or diaphragm that divides the cell into two compartments to prevent H2/O2 mixing. (b) CAD drawing and photograph of a PEM electrolyzer. Reprinted from International Journal of Hydrogen Energy, Vol. 36, Selamet, Ö. F., Becerikli, F., Mat, M. D. and Kaplan, Y., Development and testing of a highly efficient proton exchange membrane (PEM) electrolyzer stack, Pages No. 11480-11487, Copyright (2011), with permission from Elsevier.
To overcome these challenges, decoupled water electrolysis wherein the hydrogen and oxygen evolution reactions (HER and OER, respectively) are decoupled in time and/or space offers disruptive opportunities for cost reduction by avoiding the need for membranes and compression sealing and increasing energy efficiency.[1] The first decoupling approach was reported by Symes and Cronin in 2013, introducing phosphomolybdic acid as a soluble redox couple (SRC) that functions as a proton buffer and mediates the proton exchange between the anodic OER and cathodic HER in acidic electrolyte.[2] Despite generating oxygen and hydrogen in different stages, or co-generation in separated oxygen and hydrogen cells (Figure 2a), membranes are still required to prevent redox shuttling of the SRC between the primary (anode, cathode) and auxiliary electrodes in each cell, and the efficiency is lower than in conventional water electrolysis since the oxidation and reduction overpotentials of the SRC add up to those of the OER and HER, thus necessitating a larger voltage than in water electrolysis without redox couples.
The second approach was reported by our group, using nickel (oxy)hydroxide auxiliary electrodes to mediate the hydroxide ion exchange between the anodic OER and cathodic HER in alkaline electrolyte.[3] Replacing the soluble redox complexes by solid redox auxiliary electrodes leads the way to membraneless electrolysis (Figure 2b) that promises potential advantages in simplifying the electrolyzer's structure and construction, eliminating the need for expensive membranes and sealing components, and enabling high pressure operation. However, these advantages come with the disadvantage of batch operation due to the finite capacity of the auxiliary electrodes that need to be swapped periodically. In addition, the efficiency is lower than in conventional water electrolysis for the same reason as in the first approach.
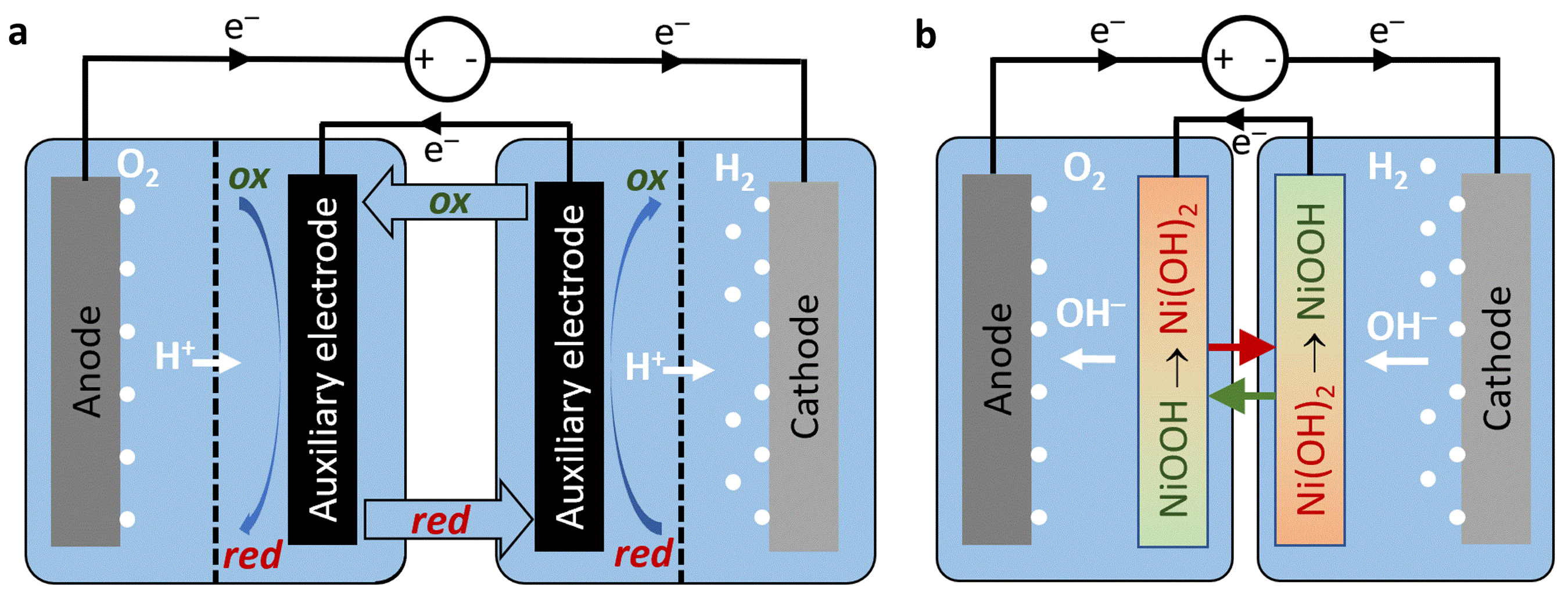
Figure 2: Decoupled water electrolysis. Schematic illustrations of separated anodic and cathodic cells in decoupled water electrolysis using a soluble redox couple in acidic electrolyte (a) or solid redox auxiliary electrodes in alkaline electrolyte (b). The dashed lines in (a) represent membranes. The anodic and cathodic reactions are the OER and HER in PEM (a) or alkaline (b) water electrolysis, respectively.
The third approach was reported by Rausch et al., introducing an electrochemical and chemical cycle whereby silicotungstic acid (ox) was reduced electrochemically at the cathode alongside oxygen evolution at the anode, and then the reduced electrolyte (red) was transferred to another cell where it was oxidized chemically and released hydrogen upon contact with a platinum catalyst.[4] This approach divides the HER into electrolytic and catalytic sub-reactions, enabling oxygen and hydrogen generation in different cells (Figure 3). However, a membrane is still needed to prevent shuttling of the soluble red and ox species in the electrolytic cell, and the efficiency is lower than in conventional electrolysis.
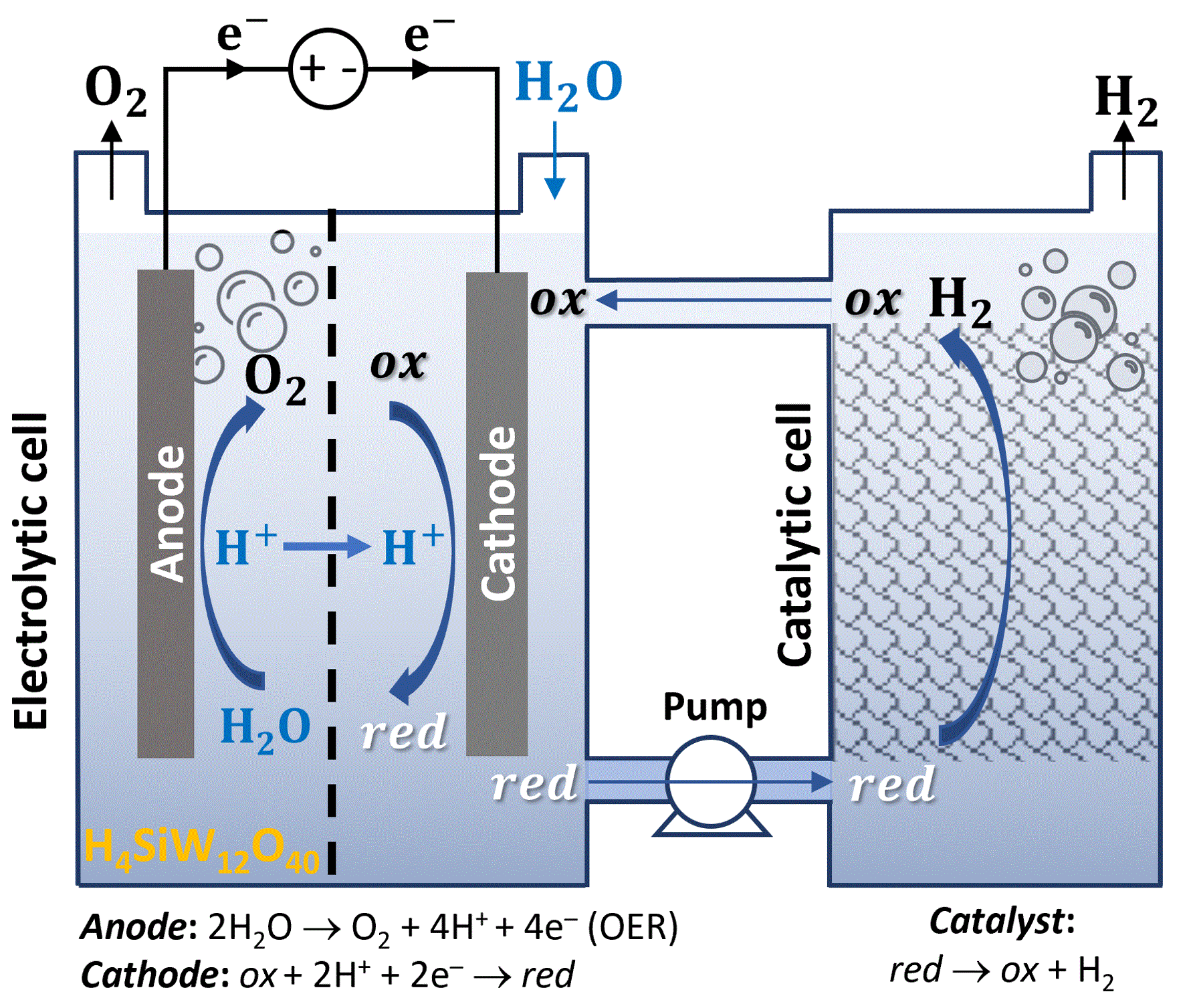 Figure 3: Electrochemical and chemical cycle for decoupled water electrolysis in silicotungstic acid. Schematic illustration of an electrochemical and chemical cycle using a soluble redox complex (H4SiW12O40) that absorbs protons in one cell and releases hydrogen in another cell. The dashed line in the electrolytic cell (left) represents a membrane that prevents shuttling of the red and ox species from one electrode to another.
Figure 3: Electrochemical and chemical cycle for decoupled water electrolysis in silicotungstic acid. Schematic illustration of an electrochemical and chemical cycle using a soluble redox complex (H4SiW12O40) that absorbs protons in one cell and releases hydrogen in another cell. The dashed line in the electrolytic cell (left) represents a membrane that prevents shuttling of the red and ox species from one electrode to another.
The fourth approach was reported by our group, introducing an electrochemical and thermally-activated chemical (ETAC) cycle that charges a nickel hydroxide (Ni(OH)2) anode to nickel oxyhydroxide (NiOOH) concurrent with hydrogen evolution at the cathode in one stage, and then reduces it back to nickel hydroxide by a chemical reaction with water that evolves oxygen without applying electricity in another stage (Figure 4).[5] The chemical reaction that evolves oxygen is slow at ambient temperature and is accelerated at high temperature (above 70°C, ideally close to 100°C). This provides a handle to control the oxygen evolution off and on in turns by circulating cold or hot electrolyte in the cell during the electrochemical and chemical stages, respectively, enabling operation without membranes and compression sealing. Furthermore, the anodic reactions divide the OER into electrochemical and chemical sub-reactions that are spread over four nickel sites to generate an O2 molecule, as opposed to four consequent proton-coupled electron transfer reactions occurring in one site at the surface of the anode in the conventional OER. This relaxes the strain on the chemical bonds that form and break between the reaction site and adsorbed reaction intermediates, resulting in a lower overpotential for oxygen evolution, lower cell voltage and higher electrolytic efficiency than in conventional water electrolysis. Indeed, a remarkable efficiency of 98.7% (based on the higher heating value of hydrogen) was reported, at cell level.[5]
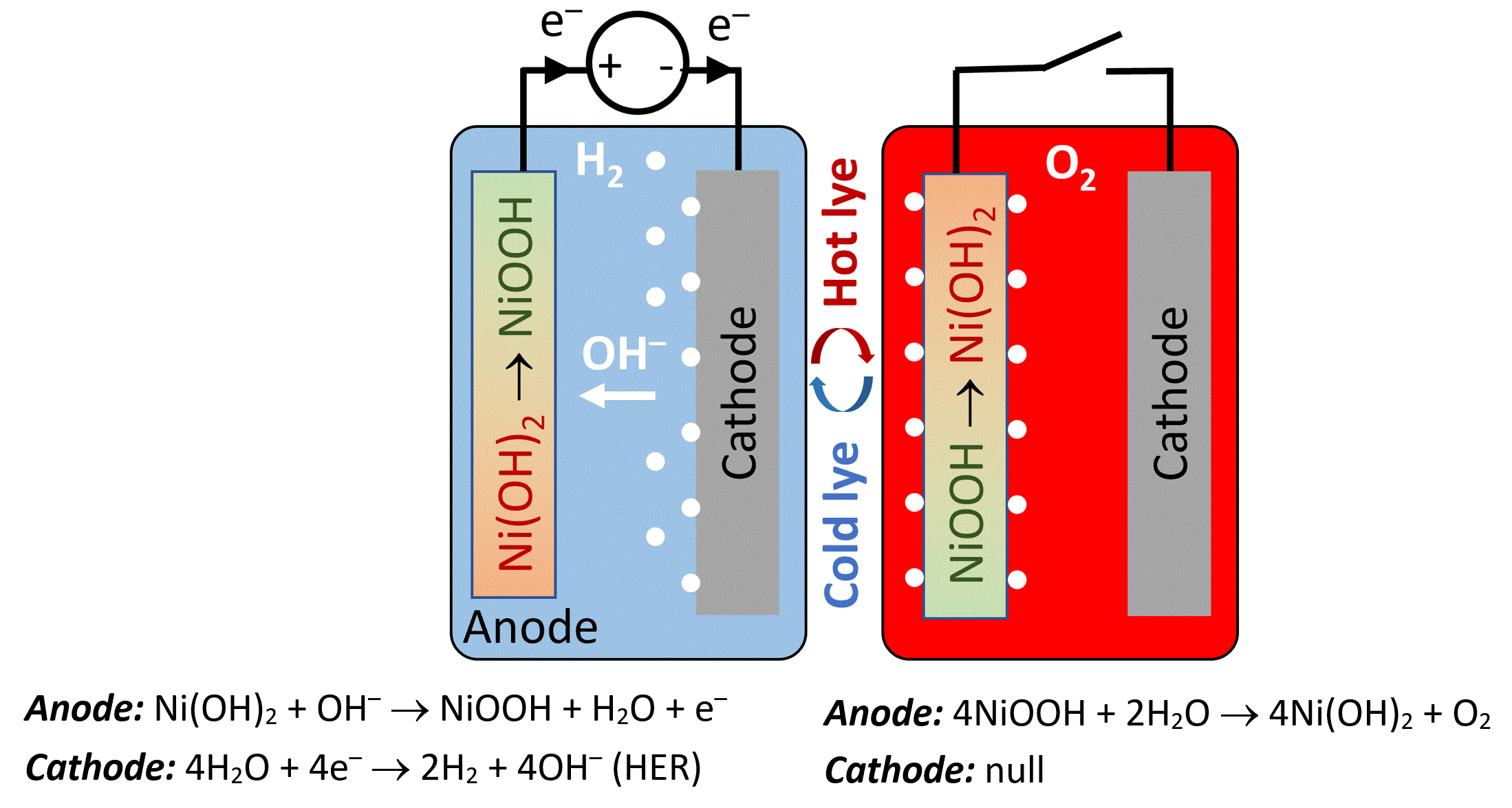 Figure 4: ETAC water electrolysis. Schematic illustration of the ETAC cycle comprising an electrochemical stage that evolves hydrogen at the cathode and charges the anode (left), and a chemical stage that evolves oxygen and regenerates (reduces) the anode without applying electricity (right).
Figure 4: ETAC water electrolysis. Schematic illustration of the ETAC cycle comprising an electrochemical stage that evolves hydrogen at the cathode and charges the anode (left), and a chemical stage that evolves oxygen and regenerates (reduces) the anode without applying electricity (right).
The ETAC cycle leads the way to high efficiency membraneless electrolysis. These advantages are pursued by H2Pro, an Israeli startup company that develops transformative electrolyzers based on the ETAC cycle. However, the ETAC cycle introduces operational challenges that arise from batch operation with thermal swings between the cold (ambient temperature) electrolyte in the electrolytic stage and the hot (close to 100°C) electrolyte in the chemical stage. Other challenges arise from capacity and rate limitations of the nickel (oxy)hydroxide anode that undergoes electro-oxidation and chemical reduction cycles that involve solid-state diffusion and phase transformation, similarly to battery electrodes.[6]
To overcome these challenges, our article[7] introduces a new electrochemical and chemical cycle that leverages the advantages of the ETAC cycle while avoiding its operational and material challenges. To this end, we use bromide (Br–) / bromate (BrO3–) as a SRC that stores and releases oxygen by turns in a near-neutral electrolyte of sodium bromide (NaBr) in water. The bromide anions are electro-oxidized to bromate concurrent with hydrogen evolution in one cell, while bromate anions are chemically reduced to bromide anions in a catalytic reaction that evolves oxygen in another cell (Figure 5). This electrochemical and chemical cycle occurs in a liquid electrolyte rather than in a solid electrode (i.e., the nickel (oxy)hydroxide anode in the ETAC cycle), avoiding the solid-state diffusion and phase transformation limitations of the ETAC cycle. In addition, the oxidized and reduced anions flow with the electrolyte from one cell to another, enabling continuous operation instead of batch operation in the ETAC cycle. Oxygen evolution is induced by a special catalyst (RuO2 Adams) rather than by a hot electrolyte, resulting in an isothermal process without the thermal swings in the ETAC cycle. Our new process presents a leap forward in decoupled water electrolysis and it may lead the way to high-efficiency membraneless water electrolysis that overcomes the limitations of century-old membrane electrolysis. The key challenges on the way to further development of this promising process to a viable technology for green hydrogen production at scale are discussed at the end of the article.
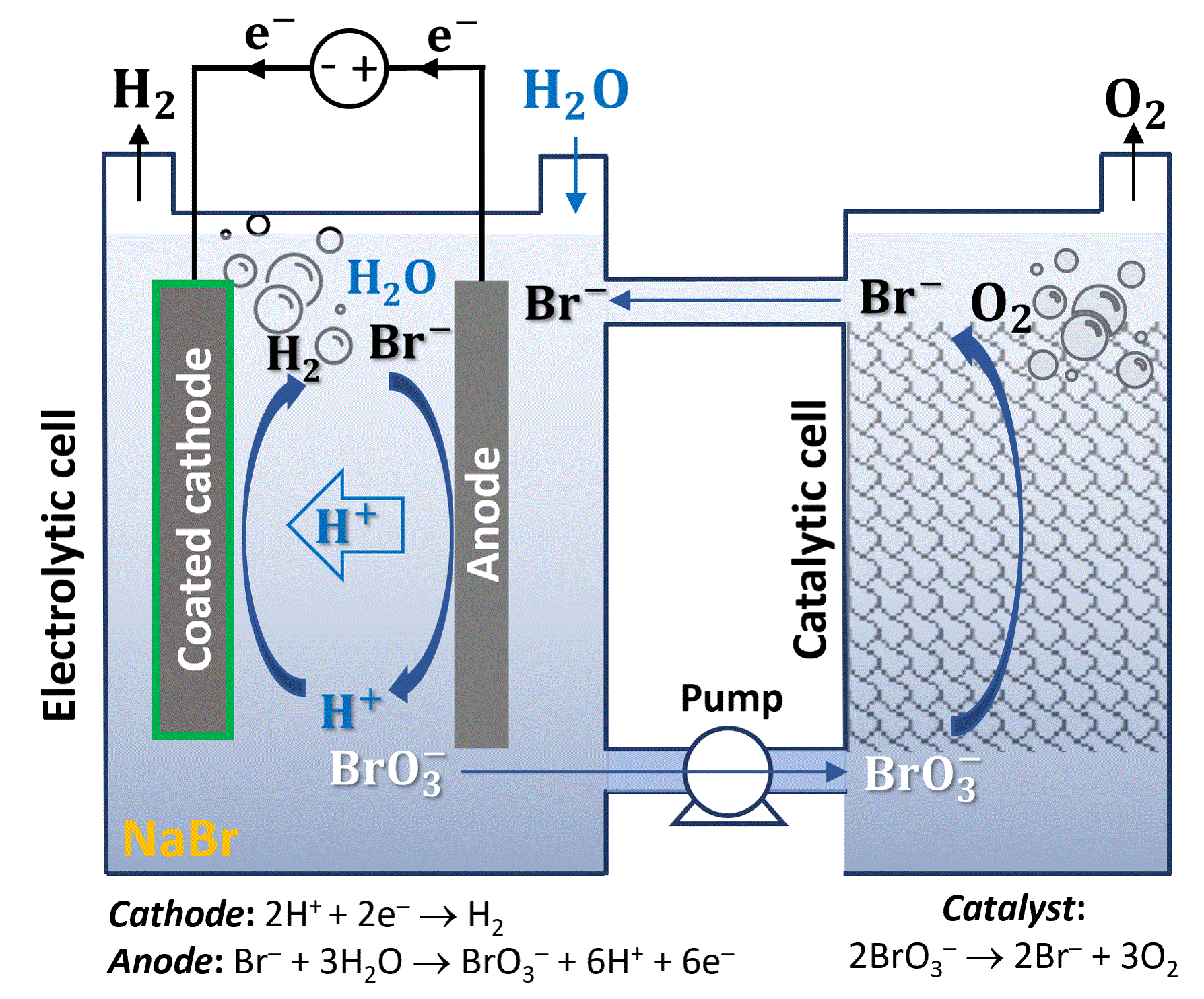 Figure 5: Electrochemical and chemical cycle for decoupled water electrolysis in NaBr aqueous electrolyte. Schematic illustration of an electrochemical and chemical cycle using bromide (Br–) / bromate (BrO3–) as a SRC that divides the OER into electrochemical and chemical sub-reactions. The bromide anions are oxidized to bromate concurrent with hydrogen evolution in one cell (left) and reduce back to bromide while evolving oxygen upon contact with a RuO2 catalyst in another cell (right).
Figure 5: Electrochemical and chemical cycle for decoupled water electrolysis in NaBr aqueous electrolyte. Schematic illustration of an electrochemical and chemical cycle using bromide (Br–) / bromate (BrO3–) as a SRC that divides the OER into electrochemical and chemical sub-reactions. The bromide anions are oxidized to bromate concurrent with hydrogen evolution in one cell (left) and reduce back to bromide while evolving oxygen upon contact with a RuO2 catalyst in another cell (right).
References:
[1] Landman, A., Rothschild, A. and Grader, G., New electrolyzer principles: Decoupled water splitting, in: Hydrogen Production by Water Electrolysis, edited by T. Smolinka and J. Gärche (Elsevier, 2021).
[2] Symes, M. D. and Cronin, L., Decoupling hydrogen and oxygen evolution during electrolytic water splitting using an electron-coupled-proton buffer. Nat. Chem. 5, 403–409 (2013).
[3] Landman, A. et al. Photoelectrochemical water splitting in separate oxygen and hydrogen cells. Nat. Mater. 16, 646–651 (2017).
[4] Rausch, B., Symes, M. D., Chisholm, G. & Cronin, L. Decoupled catalytic hydrogen evolution from a molecular metal oxide redox mediator in water splitting. Science 345, 1326–1330 (2014).
[5] Dotan, H. et al. Decoupled hydrogen and oxygen evolution by a two-step electrochemical–chemical cycle for efficient overall water splitting. Nat. Energy 4, 786–795 (2019).
[6] Bazant, M. Z. Theory of chemical kinetics and charge transfer based on nonequilibrium thermodynamics. Acc. Chem. Res. 46, 1144–1160 (2013).
[7] Slobodkin, I., Davydova, E., Sananis, M., Breytus, A. and Rothschild, A., Electrochemical and chemical cycle for high-efficiency decoupled water splitting in a near-neutral electrolyte, Nat. Mater. (DOI: 10.1038/s41563-023-01767-y).
Follow the Topic
-
Nature Materials
A monthly multi-disciplinary journal that brings together cutting-edge research across the entire spectrum of materials science and engineering, including applied and fundamental aspects of the synthesis/processing, structure/composition, properties and performance of materials.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in