Development and validation of an integrative pan-solid tumor predictor of PD-1/PD-L1 blockade benefit
Published in Cancer
Expanding and optimizing the use of immunotherapy
Immune checkpoint inhibition with monoclonal antibodies targeting PD-1 and PD-L1 has revolutionized the care of patients with advanced cancer, with approvals in multiple tumor types and pan-tumor indications.1-3
There are several biomarkers used to guide anti-PD-1/PD-L1 therapy. Microsatellite instability (MSI) and tumor mutational burden (TMB) are the two biomarkers used for pan-tumor indications, but studies have shown they fail to identify most patients who benefit from anti-PD-1/PD-L1 therapy.4,5
Likewise, although PD-L1 immunohistochemistry (IHC) is another important anti-PD-1/PD-L1 therapy biomarker, in practice it does not represent a pan-tumor biomarker, but rather a series of tumor type-specific biomarkers with various antibodies, staining platforms, PD-L1 expressing cells included in scoring algorithms, and cutoffs.6-16
Improved biomarkers capable of predicting anti-PD-1/PD-L1 therapy benefit have the potential to expand immune checkpoint inhibitors to additional patient populations outside of currently approved indications, and to focus their application more effectively on likely responsive patients when alternative therapies exist.
Additionally, immunotherapy is now often combined with chemotherapy, which means increased clinical and financial toxicity for patients. This is despite there being little evidence for additive or synergistic benefit between anti-PD-1/PD-L1 therapies and other agents in approved combinations.17 A biomarker that could guide monotherapy vs. combination therapy would be clinically useful.
In our recent publication in Communications Medicine we describe the development, validation and clinical utility of a new biomarker that fulfills these unmet medical needs – the Immunotherapy Response Score or “IRS”.
Developing innovative new cancer biomarkers using a large observational cohort
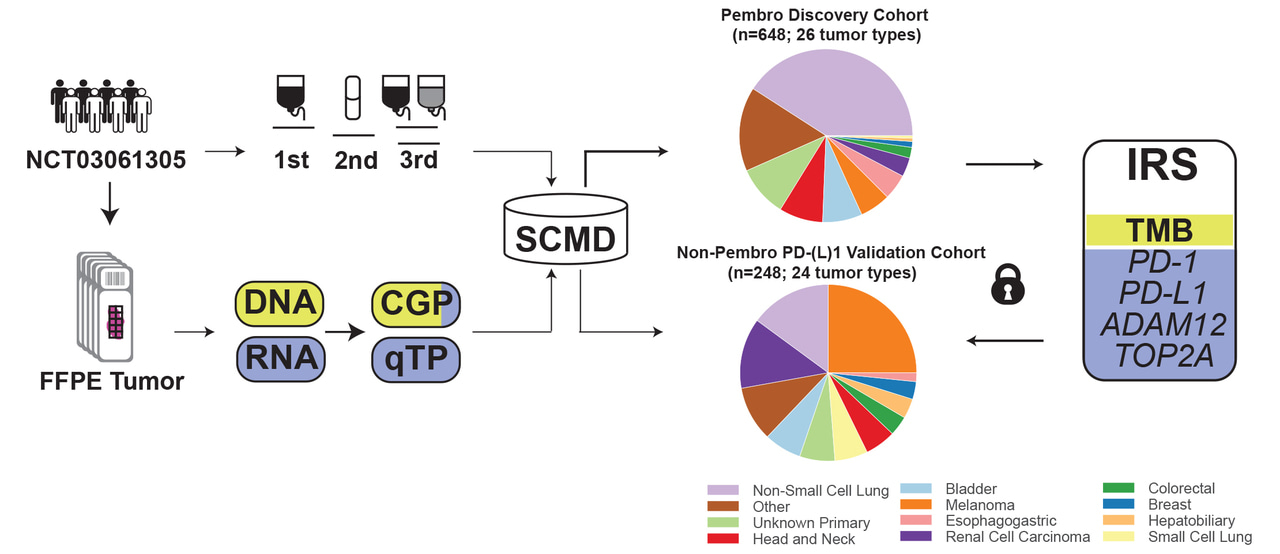
We have partnered with diverse health systems across the United States to provide routine clinical testing with our integrated molecular tumor profiling platform through an observational clinical trial protocol18 called the Strata Trial.
With data from tens of thousands of patients collected under this protocol that pairs clinically validated genomic and transcriptomic profiling of tumor tissue with outcomes data, we are discovering, validating and demonstrating clinical utility for multiple new biomarker algorithms.
For IRS, we used our clinical-molecular database to discover, and then validate in a separate cohort, a multivariate biomarker that captures the biology of the tumor and its microenvironment by combining TMB with quantitative expression of PD-L1, PD-1, ADAM12 and TOP2A.
Thanks to the longitudinal nature of our data collection in the Strata Trial (treatment data collected from all lines, going back to time of diagnosis of advanced disease), we are able to conduct within-subject comparative effectiveness studies (aka case-crossover) to assess the biomarker’s ability to predict benefit between therapies. We control by patient – every patient serves as their own control – as an effective alternative to randomization for testing many clinical associations.
For IRS, this allowed us to confirm the predictive (vs. prognostic) nature of the biomarker across tumor types by assessing patients who had received a previous line of non-immunotherapy systemic therapy prior to pembrolizumab monotherapy.
IRS predicts anti-PD-1/PD-L1 monotherapy benefit
We showed that across tumor types, IRS predicts real-world progression free survival and overall survival in anti-PD-1/PD-L1 monotherapy treated patients. IRS-high status predicts similar duration of benefit as tumor mutational burden (TMB)-high status across tumor types, but identifies twice as many patients who may benefit from checkpoint inhibitor treatment as TMB alone.
In an exploratory analysis we found that in non-small cell lung cancer (NSCLC) patients who were IRS-high there was no significant benefit of combination therapy (pembrolizumab + chemotherapy) compared to monotherapy (pembrolizumab).
Together, these data show that IRS meets the significant unmet medical need for an integrative diagnostic test that better predicts likelihood of benefit from anti-PD-1/PD-L1 checkpoint inhibitor monotherapy, across solid tumor types.
References:
- Sharma, P. et al. The next decade of immune checkpoint therapy. Cancer Discov. 11, 838–857 (2021).
- Chamoto, K., Hatae, R. & Honjo, T. Current issues and perspectives in PD-1 blockade cancer immunotherapy. Int. J. Clin. Oncol. 25, 790–800 (2020).
- Ribas, A. & Wolchok, J. D. Cancer immunotherapy using checkpoint blockade. Science 359, 1350–1355 (2018).
- Marabelle, A. et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. Lancet Oncol 21, 1353-1365 (2020).
- Cristescu, R. et al. Tumor mutational burden predicts the efficacy of pembrolizumab monotherapy: a pan-tumor retrospective analysis of participants with advanced solid tumors. J Immunother Cancer 10, e003091 (2022).
- Doroshow, D. B. et al. PD-L1 as a biomarker of response to immune checkpoint inhibitors. Nat. Rev. Clin. Oncol. 18, 345–362 (2021).
- Gavrielatou, N., Shafi, S., Gaule, P. & Rimm, D. L. PD-L1 expression scoring: non-interchangeable, non-interpretable, neither, or both. J. Natl Cancer Inst. 113, 1613–1614 (2021).
- Rimm, D. L. et al. A prospective, multi-institutional, pathologist-based assessment of 4 immunohistochemistry assays for PD-L1 expression in non-small cell lung cancer. JAMA Oncol. 3, 1051–1058 (2017).
- Salgado, R. et al. How current assay approval policies are leading to unintended imprecision medicine. Lancet Oncol. 21, 1399–1401 (2020).
- Herbst, R. S. et al. Atezolizumab for first-line treatment of PD-L1-selected patients with NSCLC. N. Engl. J. Med. 383, 1328–1339 (2020).
- Abdul Karim, L., Wang, P., Chahine, J. & Kallakury, B. Harmonization of PDL1 immunohistochemistry assays for lung cancer: a working progress. J. Thorac. Oncol. 12, e45 (2017).
- Dolled-Filhart, M. et al. Development of a prototype immunohistochemistry assay to measure programmed death ligand-1 expression in tumor tissue. Arch. Pathol. Lab. Med. 140, 1259–1266 (2016).
- Dolled-Filhart, M. et al. Development of a companion diagnostic for pembrolizumab in non-small cell lung cancer using immunohistochemistry for programmed death ligand-1. Arch. Pathol. Lab. Med. 140, 1243–1249 (2016).
- Hirsch, F. R. et al. PD-L1 immunohistochemistry assays for lung cancer: results from phase 1 of the blueprint PD-L1 IHC assay comparison project. J. Thorac. Oncol. 12, 208–222 (2017).
- Velcheti, V. et al. Real-world PD-L1 testing and distribution of PD-L1 tumor expression by immunohistochemistry assay type among patients with metastatic non-small cell lung cancer in the United States. PLoS ONE 13, e0206370 (2018).
- Grant, M. J., Herbst, R. S. & Goldberg, S. B. Selecting the optimal immunotherapy regimen in driver-negative metastatic NSCLC. Nat. Rev. Clin. Oncol. 18, 625–644 (2021).
- Palmer, A. C., Izar, B., Hwangbo, H. & Sorger, P. K. Predictable clinical benefits without evidence of synergy in trials of combination therapies with immune-checkpoint inhibitors. Clin. Cancer Res. 28, 368–377 (2022).
- Strata Trial: ClinicalTrials.gov Identifier: NCT03061305
Follow the Topic
-
Communications Medicine
A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Public health and health governance in China
Publishing Model: Open Access
Deadline: Jul 31, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in