Beyond the Organs: Why “Fuel Toxicity-Associated Spectrum (FTAS)” Could Unite T2DM, MASLD, and MASH
Published in General & Internal Medicine and Anatomy & Physiology
Beyond the Organs: Why “Fuel Toxicity-Associated Spectrum (FTAS)” Could Unite T2DM, MASLD, and MASH
By:Dr. P.Sureshkumar MD,PhD, FRCP
Date: 07/04/2026
If you work in metabolic research, you’ve felt the frustration. A patient presents with insulin resistance. A decade later, they develop non-alcoholic steatohepatitis (NASH). A colleague calls it “diabetic liver.” Another calls it “hepatic manifestation of metabolic syndrome.” The nomenclature is a Tower of Babel—T2DM, MASLD (formerly NAFLD), MASH, obesity, dyslipidemia. Each silo has its own journals, its own conferences, and its own pathogenesis narrative.
But what if we are looking at different weather patterns of the same storm?
A new article proposes a radical but elegant solution: 'The Fuel Toxicity-Associated Spectrum (FTAS)'. In this post, we’ll unpack why this unifying nomenclature might be the conceptual breakthrough metabolic medicine needs—and where it still needs to survive peer review.
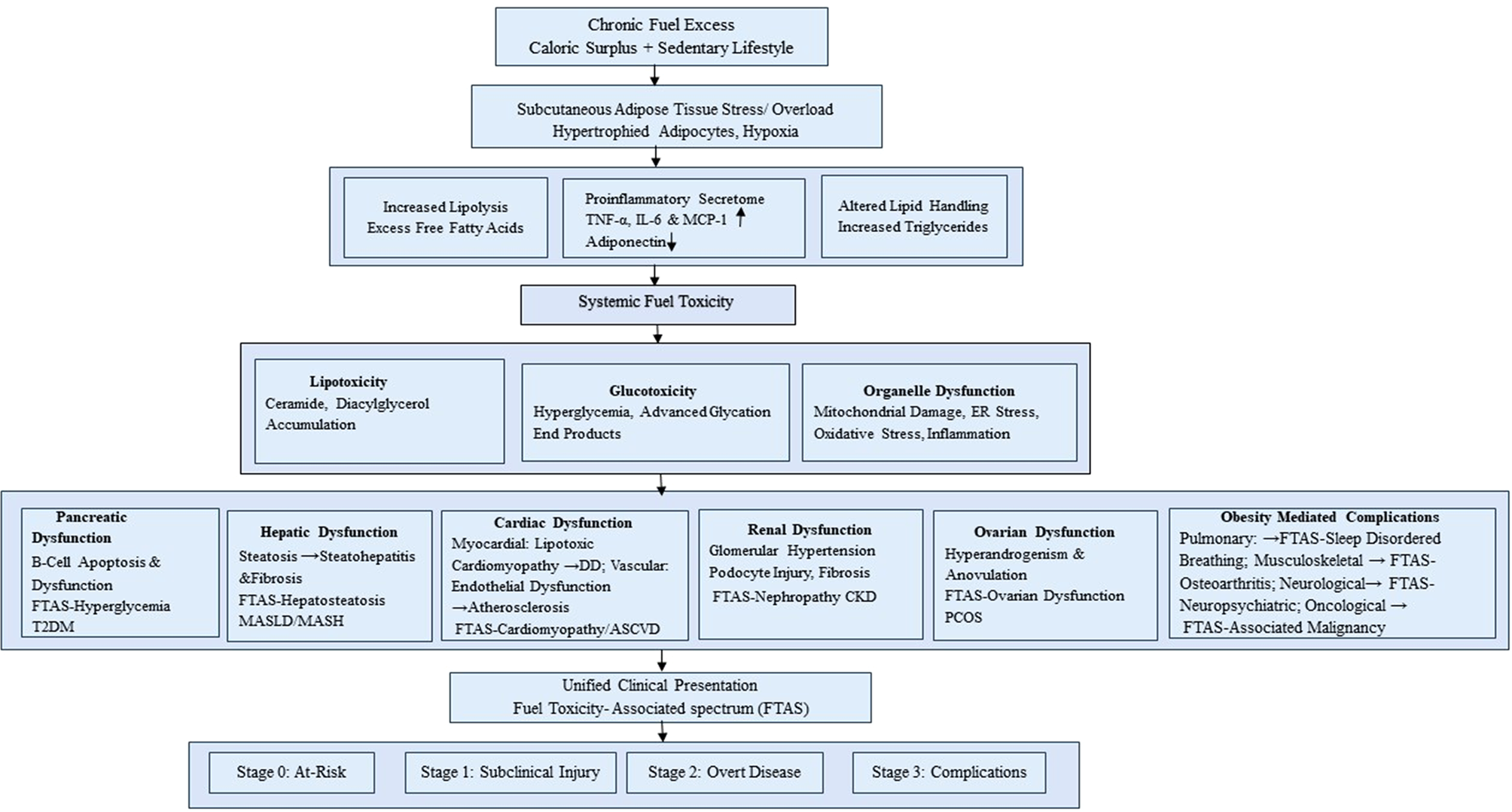
The Problem with 'Comorbidities' : The current lexicon treats Type 2 Diabetes Mellitus (T2DM) and Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD) as distinct diseases that frequently co-occur. This framework is misleading. It implies separate etiologies that merely overlap.But the evidence points to a shared root: chronic fuel overload. Whether the fuel is glucose, fructose, or saturated fatty acids, the cellular machinery doesn’t care. When the mitochondria are overwhelmed, the result is the same—lipotoxicity, glucotoxicity, ER stress, and inflammation. The author of our featured article argues that separating “diabetes” (pancreas) from “MASLD/MASH” (liver) is an anatomical relic, not a biological reality.
Enter FTAS: A Spectrum, Not a Set of Boxes
The proposed 'Fuel Toxicity-Associated Spectrum (FTAS)' collapses these categories into a single, continuous pathophysiological process. Here is the core logic:
- Stage 0 (Substrate Overload): Chronic caloric surplus exceeds adipose storage capacity.
Stage 1 (Ectopic Fuel Deposition):Lipids infiltrate the liver (MASLD) and pancreas (lipomatosis). - Stage 2 (Toxicity): Ceramides and reactive oxygen species trigger organelle dysfunction. This is the pivot point—the "toxicity" in FTAS.
- Stage 3 (Clinical Phenotypes): Depending on genetic background (e.g., PNPLA3 for liver vs. TCF7L2 for pancreas), the patient manifests predominantly as T2DM, MASH, or hypertriglyceridemia.
In the FTAS model, 'MASH with mild hyperglycemia' and 'T2DM with mild steatosis' are not different diseases. They are different coordinates on the same map.
Why This Matters for Drug Development : As a researcher, you know that a Phase 2 trial for a GLP-1 agonist often shows improvement in both HbA1c and liver fat. Under the old model, this is a 'pleiotropic effect'. Under FTAS, it is simply treating the fuel toxicity.
This nomenclature change has real-world implications:
- Clinical trials: Enrichment strategies would shift from organ-based inclusion criteria (e.g., biopsy-proven MASH) to mechanism-based criteria (e.g., evidence of fuel toxicity).
- Regulatory approval: A drug that reduces hepatic ceramides might simultaneously be a diabetes drug, a NASH drug, and a dyslipidemia drug.
- Patient communication: 'You have Fuel Toxicity Syndrome' is more honest and actionable than 'You have three chronic diseases'.
The Unanswered Questions
Here is what the author needs to defend:
1. Specificity vs. Sensitivity: Not every obese patient develops MASH or T2DM. What is the 'specific' switch that turns benign fuel storage into 'toxic' fuel storage? FTAS needs a molecular biomarker (e.g., a specific ceramide species or mitochondrial redox ratio) to define the 'toxicity' threshold.
2. The U-shaped paradox: Low fuel (starvation) and high fuel (obesity) both cause hepatic steatosis. Does FTAS include starvation-induced lipodystrophy? If not, the definition of 'toxicity' must explicitly exclude simple caloric surplus without cellular harm.
3. Therapeutic falsification: If FTAS is true, then any intervention that reduces fuel toxicity should improve all phenotypes equally. But we know metformin helps T2DM yet has minimal effect on MASH histology. Does that falsify FTAS, or merely indicate that metformin acts downstream of fuel toxicity (e.g., via GLP-1)?
4. The brain and heart: FTAS currently focuses on liver and pancreas. But fuel toxicity also drives diabetic cardiomyopathy and hypothalamic inflammation. Should the spectrum be broader?
The Verdict (So Far)
The 'Fuel Toxicity-Associated Spectrum' is not ready for the textbooks—yet. But it is precisely the kind of bold, reframing hypothesis that moves metabolic research beyond siloed thinking. For too long, we have named diseases by their victim organ rather than their perpetrator mechanism. FTAS flips that script. Whether it survives the next decade of genetic, imaging, and pharmacological testing will depend on our ability to define “toxicity” with molecular precision.
Over to you, SciPinion community. Is FTAS a unifying leap forward, or an oversimplification of organ-specific pathophysiology?
Read the full article: https://rdcu.be/e8Fst
About the author: Dr. P. Sureshkumar is a renowned consultant Diabetologist and researcher in the field
Keywords: FTAS, T2DM, MASLD, MASH, metabolic syndrome, lipotoxicity, nomenclature, pathophysiology, insulin resistance.
Follow the Topic
-
Cardiovascular Diabetology – Endocrinology Reports
This is an official companion journal to Cardiovascular Diabetology and is an open access journal publishing on clinical, translational and basic science aspects of endocrinology.
Related Collections
With Collections, you can get published faster and increase your visibility.
Pleiotropic Actions of Incretin-Based Therapies
Incretins play a crucial role in regulating glucose homeostasis and body energetics, and incretin based-therapies have fundamentally changed the management of people with diabetes and obesity. Importantly, recent evidence has revealed a wider profile of multi-organ beneficial effects for incretin therapies, including cardiovascular and renal protection, beyond glycaemic control. Whilst the full range of pleiotropic, organ-specific effects of incretins is still being elucidated, it should be noted that their use is limited by adverse events, such as gastrointestinal intolerance, and safety concerns regarding retinal disorders, muscle impairment, and pancreatitis.
Ongoing areas of interest include, but are not limited to:
- Tissue-specific effects of incretin-based medications
- Exploration of novel clinical indications for incretin therapies
- Safety concerns related to incretin therapies
- Next-generation incretin medications
- Combination of incretin treatment with lifestyle interventions
This Collection invites mini reviews, short reports and commentaries of clinical studies related to the pleiotropic effects of incretin-based therapies, but also welcomes original clinical, translation and basic research articles and reviews.
This Collection supports and amplifies research related to SDG 3, Good Health and Well-Being.
There is a related Collection "Incretin-Based Therapies in Type 2 Diabetes and Obesity: Current Evidence and the Evolving Cardiometabolic Landscape" in Cardiovascular Diabetology.
All submissions in this Collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Jan 14, 2027
Artificial intelligence in cardiovascular endocrinology: advancing precision in cardiometabolic care
In recent years, artificial intelligence (AI) has begun to permeate the clinical landscape—not as a distant promise, but as a transformative force in real-time decision-making. Nowhere is this transformation more compelling than at the crossroads of cardiovascular endocrinology, where hormonal disorders and metabolic dysregulation drive some of the world’s most pressing cardiovascular challenges.
Endocrine disorders such as diabetes, thyroid dysfunction, and adrenal abnormalities are major contributors to cardiovascular morbidity and mortality. Yet the tools traditionally used to assess cardiovascular risk often fail to capture the nuanced and dynamic nature of these diseases. AI is poised to fill this gap.
Emerging machine learning models can now detect early cardiovascular complications in patients with metabolic disorders, identify risk trajectories that are invisible to conventional clinical tools, and integrate vast amounts of data—from electronic health records and laboratory results to imaging and genomics—into clinically meaningful insights. Moreover, AI-powered wearable technologies are facilitating continuous, real-time monitoring, offering clinicians unprecedented opportunities for early intervention and personalized care outside traditional healthcare settings.
Despite these advances, key challenges remain. Algorithmic bias, lack of transparency in AI decision-making, and disparities in access to digital technologies are serious concerns that demand thoughtful and interdisciplinary solutions. Clinicians and researchers must be actively involved in shaping how AI is developed, validated, and implemented to ensure its safe and equitable use across diverse patient populations.
In response to the rapid developments in this space, we invite clinicians, researchers, and healthcare innovators to contribute to this emerging field through original research, reviews, perspectives, and clinical case studies.
We welcome submissions addressing, but not limited to:
- AI-based prediction and prevention of cardiovascular events in endocrine disorders
- Integration of AI with imaging, laboratory, and wearable data in clinical practice
- Development of clinical decision support systems for cardiometabolic management
- Applications of AI in diabetic cardiomyopathy and other endocrine-related heart diseases
- Ethical, legal, and health equity considerations in the deployment of AI technologies
This is a pivotal moment for the evolution of precision medicine in cardiometabolic care. By harnessing the power of AI, we have the opportunity to redefine standards of care, close diagnostic gaps, and improve outcomes for high-risk patient populations.
We encourage you to submit your work and join us in advancing this important and timely field. Together, let us shape a future where intelligent systems complement clinical expertise for the benefit of all.
This Collection supports and amplifies research related to SDG 3, Good Health and Well-Being.
All submissions in this collection undergo the journal’s standard peer review process. Similarly, all manuscripts authored by a Guest Editor(s) will be handled by the Editor-in-Chief. As an open access publication, this journal levies an article processing fee (details here). We recognize that many key stakeholders may not have access to such resources and are committed to supporting participation in this issue wherever resources are a barrier. For more information about what support may be available, please visit OA funding and support, or email OAfundingpolicy@springernature.com or the Editor-in-Chief.
Publishing Model: Open Access
Deadline: Jun 30, 2026

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
Please go through the article and give your valuable feedback
Please comment