Interfacial Evolution and Accelerated Aging Mechanism for LiFePO4/Graphite Pouch Batteries Under Multi-Step Indirect Activation
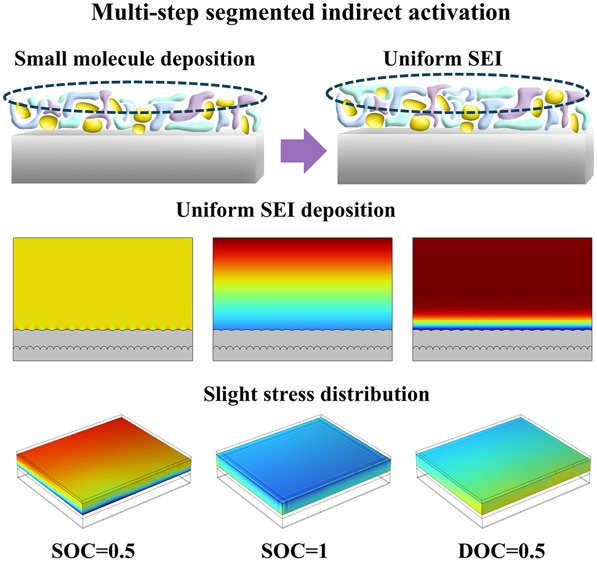
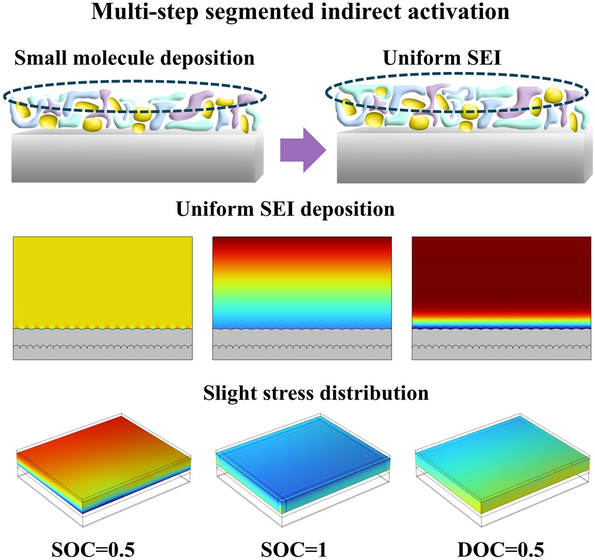
The dissolution of iron from the cathode and electrode/electrolyte interface (EEI) during long cycles significantly accelerates the aging process of LiFePO4 (LFP)/graphite batteries; there is a lack of systematic understanding of the spatial distribution of the EEI interface layer and the dissolve of Fe ions, especially in terms of the mechanism of the cathode–electrolyte interphase (CEI), solid electrolyte interphase (SEI), and iron dissolution. In this study, aged cells were subjected to continuous activation with constant current and multi-step segmented indirect activation (IA) and analyzed for capacity fade, impedance growth, and active Li+ mass loss at the EEI and nanoscale levels. The interaction between dissolved Fe2+ and the EEI in LFP/graphite pouch batteries was proposed and verified. The findings indicate that during IA process, the electric field facilitates the migration of solvated ions toward the electrodes, while simultaneously inhibiting the formation of organic species such as ROCO2Li. The SEI primarily consists of a mixture of organic and inorganic small molecules, forming a continuous and uniform film on the electrode surface. This study demonstrates that IA favors the formation of a uniform EEI and offers constructive insights for advancing accelerated lifetime prediction strategies in lithium-ion batteries.
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in