Looking for who is the most glucose addicts in the bone marrow micro-environment in acute myeloid leukaemia
Published in Cancer


Note: An ancient Chinese legend that the dragon (metaphorically ‘hematopoiesis’) has nine sons (surrounding the dragon), with each of them having different qualities (‘cell subsets, including leukemia and non-leukemia cells’) and tastes (‘glucose preference’).
Background
Acute myeloid leukaemia (AML) is a blood cancer that originates from immature myeloid lineage cells. Metabolic re-programming is a hallmark of cancer cells which rely primarily on glucose metabolism for energy production and macromolecule synthesis (1, 2). Cell-intrinsic mechanisms of preferential glucose uptake is reported for bulk leukaemia samples, without precisely identifying which cell populations are involved (3). Benign immune cells infiltrating in the tumor micro-environment were reported to be in high demand of glucose to function (4, 5). So far, it remains unknown how glucose is partitioned between different cell subsets in the AML bone marrow micro-environment (BMME) under various disease burden or therapeutic interventions. And to what extent the differences of glucose uptake can be exploited to trace the AML blast cell numbers?
Glucose uptake can be measured using [18F] fluorodeoxyglucose (FDG) positron emission tomography (PET) imaging to detect cancer and monitor treatment responses in vivo (6, 7). Since both leukaemia cells and hematopoietic stem cells propagate in bone marrow, it’s difficult to distinguish AML cells from other non-leukaemia cells in BMME through PET imaging. Reinfeld et al. developed an interesting method to quantify the relative incorporation of metabolites in vivo on a cellular level (7). They reported that myeloid cells in the tumor micro-environment, but not cancer cells, have the greatest glucose uptake. This prompted us to investigate the cell-wise glucose partitioning in AML and BMME cells by a FDG-PET tracer-based method.
Results
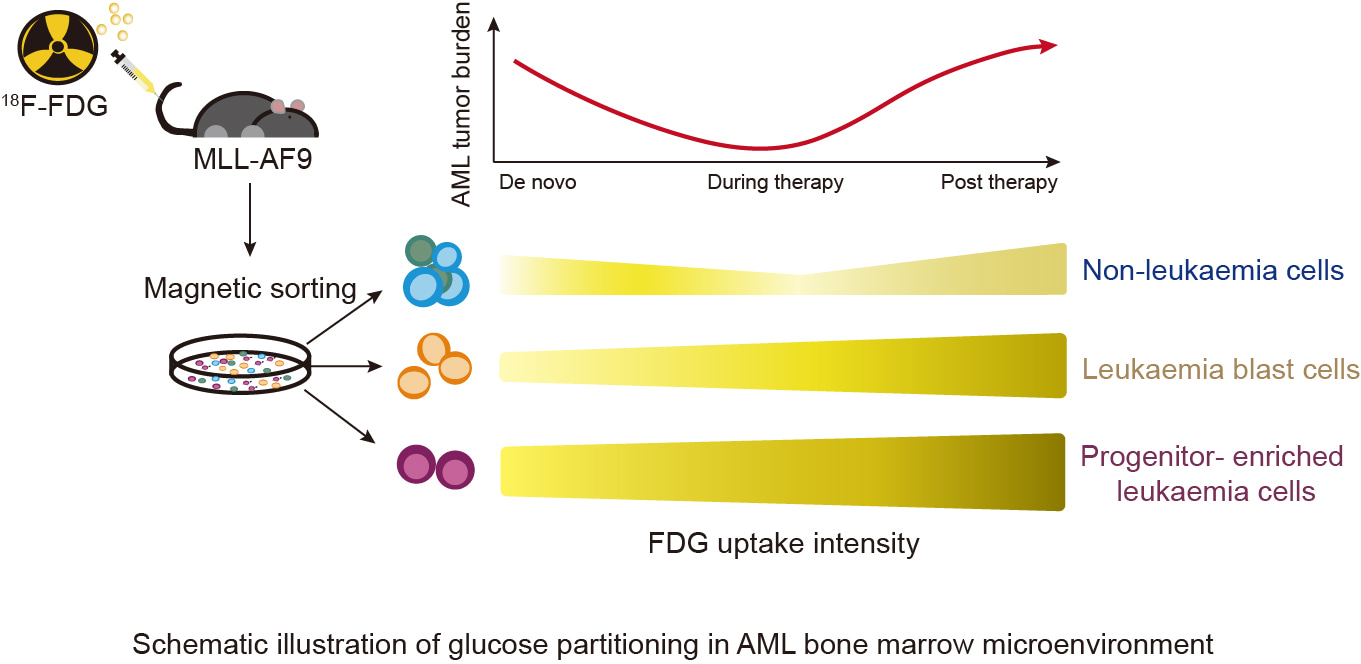
First, we studied glucose partitioning of leukaemia cells and non-leukaemia cells in the BMME. MLL-AF9 cells (GFP+CD45.2+) were tail-vein injected into CD45.1 mice to establish the MLL-AF9 AML model for [18F]-FDG-PET uptake assay. By magnetic bead sorting, leukaemia cells and non-leukaemia myeloid cells were obtained, and FDG uptake capacity was quantitatively measured, respectively. The results showed that leukaemia cells had the highest glucose uptake.
Then, we want to determine which leukaemia cells had the highest glucose uptake, so we took a closer look of glucose uptake in leukaemia cell subsets. ScRNA-sequencing data indicated that the glycolysis and glucose metabolism pathways were enriched in myeloid progenitor cells, especially granulocyte-macrophage progenitor cells (GMP-like cells). To validate these findings, we injected [18F]-FDG into MLL-AF9 mice and used magnetic beads to sort CD117+ and CD117- subsets of CD45.1- MLL-AF9 leukaemia cells. Our results confirmed that progenitor-enriched leukaemia cells (CD45.1-CD117+) absorbed glucose most efficiently within AML population.
We next sought to evaluate the effect of anti-leukaemia drugs on glucose uptake. We treated mice with anti-leukaemia drugs reminiscent of clinical regimen. Therapy reduced numbers of leukaemia cells without impairing their capacity to uptake glucose. Compared with untreated leukaemia mice, the leukaemia cells were still able to absorb glucose more efficiently than non-leukaemia cells upon drug treatment. Notably, after anti-leukaemia treatment, leukaemia stem progenitor cells significantly increased glucose uptake, and more differentiated leukaemia cells also enhanced glucose uptake capacity comparable to that of progenitor-enriched leukaemia cells.
Take home message and new directions
Based on a MLL-AF9 induced AML mouse model and cell-wise quantitative FDG-PET tracer, our work uncovers dynamic alterations in glucose uptake in AML cells and and non-leukaemia cells in the BMME before, during and after anti-leukaemia therapy. Growing numbers of studies suggested that resistant leukaemia cells shift glucose metabolism to OxPhos, but our work unravels another facet of glucose addiction within leukaemia cells, especially LSPC. After stopping therapy, glucose uptake by diverse cell sub-populations soared, leukaemia rebound simultaneously. If validated in humans, our data raise the possibility of therapeutic targeting glucose uptake by leukaemia stem/progenitor cells. Additionally, distinct partitioning of glucose with a detecting resolution of 10E-5, suggests a potential FDG-PET tracer-based strategy to monitor the measurable residual disease (MRD) in AML. Uptake of other metabolites such as amino acids, nucleotides and fatty acids also needs further investigation.
Reference
- Vander Heiden MG, DeBerardinis RJ. Understanding the Intersections between Metabolism and Cancer Biology. Cell. 2017;168(4):657-69.
- Fernandez-de-Cossio-Diaz J, Vazquez A. Limits of aerobic metabolism in cancer cells. Scientific reports. 2017;7(1):13488.
- Saito Y, Chapple RH, Lin A, Kitano A, Nakada D. AMPK Protects Leukemia-Initiating Cells in Myeloid Leukemias from Metabolic Stress in the Bone Marrow. Cell Stem Cell. 2015;17(5):585-96.
- Elia I, Rowe JH, Johnson S, Joshi S, Notarangelo G, Kurmi K, et al. Tumor cells dictate anti-tumor immune responses by altering pyruvate utilization and succinate signaling in CD8(+) T cells. Cell Metab. 2022;34(8):1137-50.e6.
- Ho PC, Bihuniak JD, Macintyre AN, Staron M, Liu X, Amezquita R, et al. Phosphoenolpyruvate Is a Metabolic Checkpoint of Anti-tumor T Cell Responses. Cell. 2015;162(6):1217-28.
- Ye H, Adane B, Khan N, Alexeev E, Nusbacher N, Minhajuddin M, et al. Subversion of Systemic Glucose Metabolism as a Mechanism to Support the Growth of Leukemia Cells. Cancer Cell. 2018;34(4):659-73 e6.
- Reinfeld BI, Madden MZ, Wolf MM, Chytil A, Bader JE, Patterson AR, et al. Cell-programmed nutrient partitioning in the tumour microenvironment. Nature. 2021;593(7858):282-8.
Follow the Topic
-
Leukemia
This journal publishes high quality, peer reviewed research that covers all aspects of the research and treatment of leukemia and allied diseases. Topics of interest include oncogenes, growth factors, stem cells, leukemia genomics, cell cycle, signal transduction and molecular targets for therapy.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in