Nanosecond Bacteria Inactivation Realized by Locally Enhanced Electric Field Treatment (LEEFT)
Published in Electrical & Electronic Engineering

Public health is threatened by pathogenic bacteria in water, so water disinfection is necessary in many cases, and how fast a disinfection method can kill bacteria is critical. The commonly used disinfectants, such as chlorine and ozone, can kill bacteria within a few seconds. Nevertheless, these chemical-based methods may induce antimicrobial resistance and generate disinfection by-products (DBPs). Electric field treatment (EFT) is a physical method relying on cell electroporation, which is a phenomenon that pores are form on cell membrane when a cell is placed in strong electric field. Electric pulses with micro to millisecond durations are capable to induce cell inactivation in conventional EFT, but shorter pulses, such as nanosecond pulses, are considered too short to charge the cell membrane and kill the bacteria.
In this work, we achieved nanosecond bacteria inactivation using LEEFT, pushing the minimal time required for bacteria inactivation to nanosecond region. We used lab-on-a-chip devices with gold nanowedges on chip to conduct an operando investigation on the ultrafast bacteria inactivation process and mechanism. The results showed that after 20 nanosecond (ns) pulses at 40 kV/cm were applied for 10 times (totally 200 ns), 95% nanowedges achieved bacteria inactivation at their tips. Compared with conventional EFT without nanowedges, LEEFT can lower the applied eclectic field strength by 8 times or shorten the treatment time by a million times, thus greatly reducing the energy comsumption.
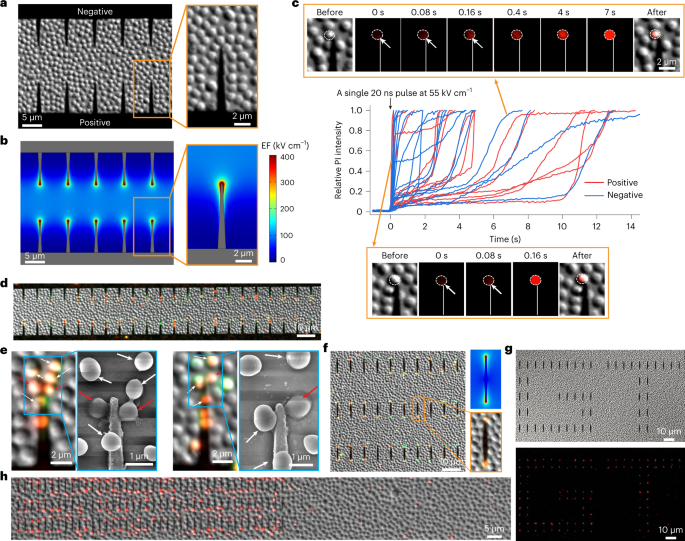
To enable the operando investigation of the ultrafast bacteria inactivation, we used e-beam lithography to fabricate gold nanowedges at the electrode edge on chip. The model bacteria Staphylococcus epidermidis were immobilized on the chip surface (Figure 1a). When electric pulses were applied to the electrodes, the electric field were enhanced at the nanowedge tips due to the lightning rod effect (Figure 1b). The cell damage/inactivation were indicated by fluorescence markers (Figure 1c). The nanowedges not connected to but distributed between the two electrodes could also enhance the electric field and induce ultrafast bacteria inactivation (Figure 1d).
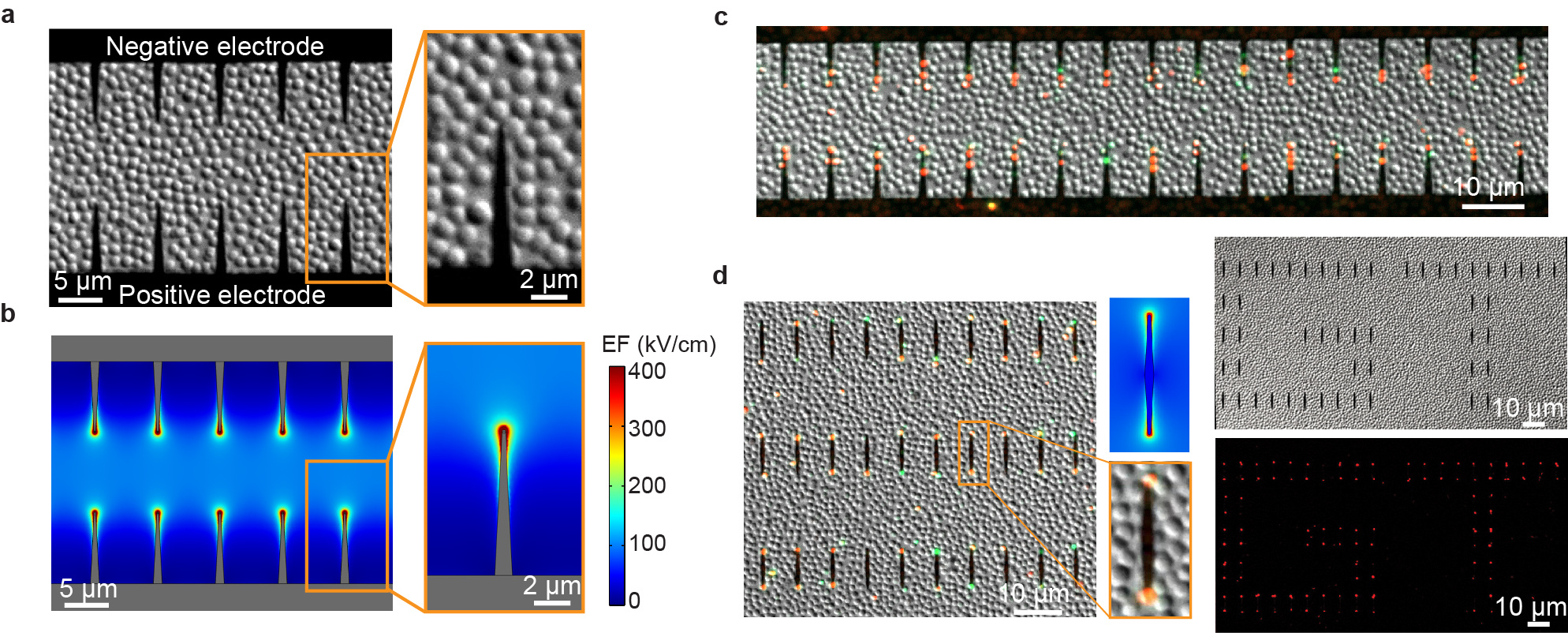
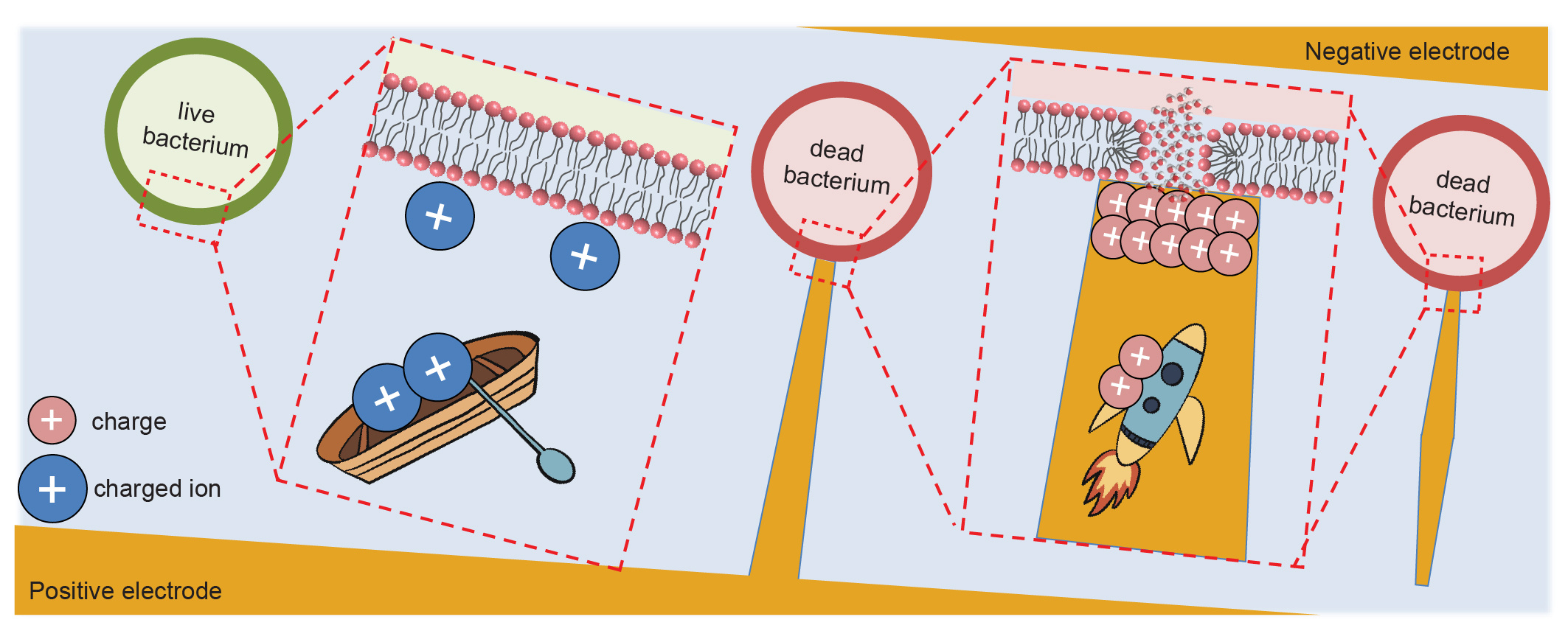
We have analyzed the cell membrane charging process using theoretical simulation and proposed the mechanism for this ultrafast bacteria inactivation (Figure 2). When exposed to an electric field, the cell membrane is like a capacitor in a circuit, which can be charged by the charged ions in water. Since the water conductivity is relatively low and the cell membrane is charged slowly, nanosecond pulses are too short to fully charge the membrane and induce electroporation. Nevertheless, in LEEFT, the cell located at the nanowedge tip can be directly charged by the charges concentrated at the nanowedge tip. Since the charges in the gold nanowedge travel much faster than the charged ions in water, the cell membrane is charged instantly, causing ultrafast electroporation and bacteria inactivation.
LEFFT has many future applications. Since it is less likely to induce undesired electrochemical oxidation and byproducts, it can be used to treat high quality samples, such as liquid food, drinking water, or biomedical sample. The ultrafast nanosecond bacteria inactivation can potentially lead to high water throughput and disinfection efficiency. LEEFT can be operated with ultrashort pulses and at a moderate electric field strength, making it safer and more energy efficient than conventional EFT.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in