Optimizing functional connectivity scanning conditions for predicting autistic traits
Published in Behavioural Sciences & Psychology
How do we best study brain-behavior relationships in autism?
Autism spectrum disorder (‘autism’ hereafter) affects approximately 1% of children around the world1 and is characterized by difficulties with social communication and interaction, restricted and repetitive behaviors, and sensory atypicalities2. Researchers and clinicians need to better understand the neurobiological underpinnings of autistic traits in youth. A better appreciation of how the brain gives rise to autistic features will help improve understanding of the condition and might lead to better ways to support autistic individuals and their families.
There are various methods to characterize the brain-based correlates of autism traits using functional magnetic resonance imaging (fMRI) connectivity data, in which measures of similarity of the blood-oxygen-level-dependent (BOLD) signal are computed between different regions of interest3. Prediction-based studies (using functional connectivity data to predict a phenotype) are one approach that has shown promise. In dimensional prediction studies, a continuous measure of a phenotype is predicted. One method of dimensional prediction is connectome-based predictive modelling (CPM)4,5, which seeks to identify the functional connections most strongly predictive of a given phenotype. Using CPM, there have been many studies of individuals with autism describing the brain correlates of clinician-rated autism symptoms6,7. Other phenotypes have also been studied using the approach in autism, including behavioral inhibition8, social responsiveness6,9, and attentional states10.
Can attention be probed to understand autism?
Which scanning conditions yield the best predictive modelling performance in autism is unknown. Most studies in the autism field have focused on resting-state fMRI, in which participants rest quietly while being scanned. Nevertheless, in participants without autism, the importance of scanning condition (e.g., ‘brain state’) is being recognized11-14. These studies suggest that predicting out-of-scanner phenotypes using connectivity measured during an in-scanner task tends to increase prediction accuracy, particularly when the task probes some aspect of the out-of-scanner item of interest (e.g., memory tasks in the scanner tend to result in higher prediction of memory performance outside the scanner15).
In addition, there are a number of studies showing that in-scanner attention tasks can be used to inform the neurobiological organization of autism16-20. There are also other brain imaging studies suggesting an overlap between the functional networks important in ADHD and autism6,21. At a behavioral level, the co-occurrence of autism and attention-deficit/hyperactivity disorder (ADHD) symptoms has long been acknowledged22-25.
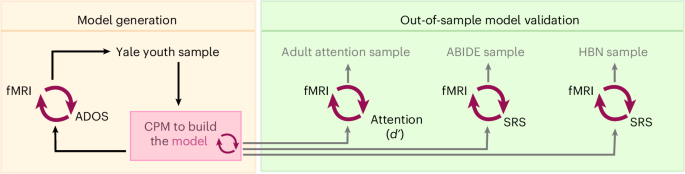
Based on the importance of tasks, as well as the importance of attention in autism, we studied brain-state-associated improvements in prediction performance in a sample of youth with autism and neurotypical participants26. Using data from three different scanning conditions—a task requiring sustained attention, a task requiring selective social attention (SSA), and a resting-state condition—we applied CPM to probe brain–behavior relationships. Specifically, the gradual onset continuous performance task (gradCPT)27 tests the ability to sustain attention to constantly changing stimuli. The SSA task captures the ability to process dynamic, multimodal faces presented one by one within a complex visual scene28.
We hypothesized that consistent with the social features of autism, prediction performance of autistic traits would be highest in the SSA task and would increase with the presence of increased social cues. We hypothesized the next highest prediction performance would result from the sustained attention task, due to the restricted and repetitive behaviors observed in autism, and that both tasks would outperform resting-state data. To determine whether results were robust, we used three other datasets to determine whether successful models can generalize to external samples. One of the datasets was used to assess the model’s generalizability in predicting performance on an attention task; the other two datasets were used to assess prediction of other autistic features.
What we found
In dataset one, a sample of youth scanned at Yale with autism and neurotypical participants (n = 63), we found that gradCPT resulted in high prediction performance of autistic traits. Predictions using the SSA task and the resting-state data were much lower, in some cases worse than chance level. In dataset two (n = 25), we observed the predictive network model of autistic traits generated from the sustained attention task generalized to predict measures of attention in neurotypical adults. In datasets three and four, we determined the same predictive network model further generalized to predict measures of social responsiveness in the Autism Brain Imaging Data Exchange (ABIDE29; n = 229) and the Healthy Brain Network (HBN30; n = 643). The predictive network model was neurobiologically complex, but aligned well with previous models of autism31. Specifically, we observed many connections in the model involved heteromodal association cortex—the parts of the brain involved in mediating higher-order thought.
Why this matters
The majority of fMRI predictive modelling studies have tended to rely on resting-state data. Our results suggest that by optimizing the brain state under which functional data are acquired, more accurate brain-behavior relationships can be determined11. Improved brain-behavior mapping increases the potential clinical utility of neuroimaging approaches32 and might help obtain a more accurate picture of brain circuits underlying the complex phenotypic landscape of autism. Moreover, our data suggest that not all tasks are equal. Our original hypothesis regarding prediction performance was that due to the social difficulties observed in autism, the SSA tasks would result in the highest prediction accuracy. Our data indicate that simply because a scanning condition is ostensibly related to a given phenotype does not mean that a brain-behavior relationship can be found in neurodevelopmental conditions.
Looking ahead
There are a number of exciting future directions to consider from our study. The participants in the first sample used from Yale all had a fairly high IQ. More work should be conducted using participants with a broad range of IQ scores to determine which scanning conditions are optimal for prediction performance. In addition, including participants with moderate autism symptoms to tease apart the brain correlates of more subtle autistic phenotypes will be important. Additional studies should also be conducted in younger samples, such as toddlers and young children.
Also, the Yale sample comprised mostly males. It is well-known that there are sex-based differences in autism involving brain circuitry33,34. Follow-up work with females with autism should be conducted. It is also possible that a more active social fMRI task condition requiring participant engagement could improve prediction. A few of the samples used here were quite small compared to other datasets of adolescents35. Our study should be replicated in larger samples. Finally, the similarity of the predictive model identified here should be more rigorously compared to other autism models. We have begun this work and have determined in preliminary analyses that autism models are similar to models obtained from participants with attention issues36.
Conclusion
We determined in a preliminary study that sustained attention tasks, such as gradCPT, can improve the prediction of autistic traits. We generated a robust brain-based marker that generalized to predict attention and social phenotypes in independent samples. Overall, our results highlight the need to continue investigating the optimal brain states for modelling phenotypes in autism and related neurodevelopmental conditions.
References
1 Zeidan, J. et al. Global prevalence of autism: A systematic review update. Autism Res 15, 778-790, doi:10.1002/aur.2696 (2022).
2 Diagnostic and statistical manual of mental disorders: DSM-5., (American Psychiatric Association, 2013).
3 Biswal, B., Yetkin, F. Z., Haughton, V. M. & Hyde, J. S. Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34, 537-541, doi:10.1002/mrm.1910340409 (1995).
4 Finn, E. S. et al. Functional connectome fingerprinting: identifying individuals using patterns of brain connectivity. Nat Neurosci 18, 1664-1671, doi:10.1038/nn.4135 (2015).
5 Shen, X. L. et al. Using connectome-based predictive modeling to predict individual behavior from brain connectivity. Nat Protoc 12, 506-518, doi:10.1038/nprot.2016.178 (2017).
6 Lake, E. M. R. et al. The Functional Brain Organization of an Individual Allows Prediction of Measures of Social Abilities Transdiagnostically in Autism and Attention-Deficit/Hyperactivity Disorder. Biol Psychiatry 86, 315-326, doi:10.1016/j.biopsych.2019.02.019 (2019).
7 Ma, X. et al. Connectome-based prediction of the severity of autism spectrum disorder. Psychoradiology 3, kkad027, doi:10.1093/psyrad/kkad027 (2023).
8 Rohr, C. S., Kamal, S. & Bray, S. Building functional connectivity neuromarkers of behavioral self-regulation across children with and without Autism Spectrum Disorder. Dev Cogn Neuros-Neth 41, doi:ARTN 100747
10.1016/j.dcn.2019.100747 (2020).
9 Dufford, A., Kimble, V., Tejavibulya, L., Dadashkarimi, J. & Scheinost, D. Predicting Transdiagnostic Social Impairments in Childhood Using Connectome-Based Predictive Modeling. Biol Psychiat 91, S87, doi:10.1016/j.biopsych.2022.02.234 (2022).
10 Horien, C. et al. A generalizable connectome-based marker of in-scan sustained attention in neurodiverse youth. Cereb Cortex 33, 6320-6334, doi:10.1093/cercor/bhac506 (2023).
11 Finn, E. S. Is it time to put rest to rest? Trends Cogn Sci 25, 1021-1032, doi:10.1016/j.tics.2021.09.005 (2021).
12 O’Connor, D., Horien, C, Mandino, F, Constable, RT. Identifying dynamic reproducible brain states using a predictive modelling approach. bioRxiv, doi:https://doi.org/10.1101/2022.10.14.512147 (2022).
13 Zhang, X., Hulvershorn, LA, Constable, RT, Zhao, Y, Wang, S. Cost efficiency of fMRI studies using resting-state vs task-based functional connectivity. arXiv, doi:
https://doi.org/10.48550/arXiv.2411.01092 (2024).
14 Ramduny, J. & Kelly, C. Connectome-based fingerprinting: reproducibility, precision, and behavioral prediction. Neuropsychopharmacology 50, 114-123, doi:10.1038/s41386-024-01962-8 (2024).
15 Ju, S. et al. Connectome-based predictive modeling shows sex differences in brain-based predictors of memory performance. Front Dement 2, 1126016, doi:10.3389/frdem.2023.1126016 (2023).
16 Keehn, B., Nair, A., Lincoln, A. J., Townsend, J. & Muller, R. A. Under-reactive but easily distracted: An fMRI investigation of attentional capture in autism spectrum disorder. Dev Cogn Neurosci 17, 46-56, doi:10.1016/j.dcn.2015.12.002 (2016).
17 Keehn, B., Shih, P., Brenner, L. A., Townsend, J. & Muller, R. A. Functional connectivity for an "Island of sparing" in autism spectrum disorder: An fMRI study of visual search. Hum Brain Mapp 34, 2524-2537, doi:10.1002/hbm.22084 (2013).
18 Vaidya, C. J. et al. Controlling attention to gaze and arrows in childhood: an fMRI study of typical development and Autism Spectrum Disorders. Dev Sci 14, 911-924, doi:10.1111/j.1467-7687.2011.01041.x (2011).
19 Rahko, J. S. et al. Attention and Working Memory in Adolescents with Autism Spectrum Disorder: A Functional MRI Study. Child Psychiatry Hum Dev 47, 503-517, doi:10.1007/s10578-015-0583-6 (2016).
20 Fitzgerald, J. et al. Disrupted functional connectivity in dorsal and ventral attention networks during attention orienting in autism spectrum disorders. Autism Res 8, 136-152, doi:10.1002/aur.1430 (2015).
21 Kernbach, J. M. et al. Shared endo-phenotypes of default mode dsfunction in attention deficit/hyperactivity disorder and autism spectrum disorder. Transl Psychiatry 8, 133, doi:10.1038/s41398-018-0179-6 (2018).
22 Stevens T, P. L., Barnard-Brak L. The comorbidity of ADHD in children diagnosed with autism spectrum disorder. Research in Autism Spectrum Disorders 31, 11-18 (2016).
23 Reiersen, A. M. & Todd, R. D. Co-occurrence of ADHD and autism spectrum disorders: phenomenology and treatment. Expert Rev Neurother 8, 657-669, doi:10.1586/14737175.8.4.657 (2008).
24 Antshel, K. M. & Russo, N. Autism Spectrum Disorders and ADHD: Overlapping Phenomenology, Diagnostic Issues, and Treatment Considerations. Curr Psychiatry Rep 21, 34, doi:10.1007/s11920-019-1020-5 (2019).
25 Alvarez-Fernandez, S. et al. Perceived social support in adults with autism spectrum disorder and attention-deficit/hyperactivity disorder. Autism Res 10, 866-877, doi:10.1002/aur.1735 (2017).
26 Horien, C. et al. Optimizing functional connectivity scanning conditions for predicting autistic traits. Nature Mental Health, doi:10.1038/s44220-026-00623-7 (2026).
27 Rosenberg, M. D. et al. A neuromarker of sustained attention from whole-brain functional connectivity. Nat Neurosci 19, 165-+, doi:10.1038/nn.4179 (2016).
28 Chawarska, K., Macari, S. & Shic, F. Context modulates attention to social scenes in toddlers with autism. J Child Psychol Psychiatry 53, 903-913, doi:10.1111/j.1469-7610.2012.02538.x (2012).
29 Di Martino, A. et al. Data Descriptor: Enhancing studies of the connectome in autism using the autism brain imaging data exchange II. Sci Data 4, doi:ARTN 170010
10.1038/sdata.2017.10 (2017).
30 Alexander, L. M. et al. An open resource for transdiagnostic research in pediatric mental health and learning disorders. Sci Data 4, 170181, doi:10.1038/sdata.2017.181 (2017).
31 Dufford, A. J. et al. Predicting Transdiagnostic Social Impairments in Childhood using Connectome-based Predictive Modeling. medRxiv, 2022.2004.2007.22273518, doi:10.1101/2022.04.07.22273518 (2022).
32 Finn, E. S. & Rosenberg, M. D. Beyond fingerprinting: Choosing predictive connectomes over reliable connectomes. Neuroimage 239, 118254, doi:10.1016/j.neuroimage.2021.118254 (2021).
33 Floris, D. L. et al. Towards robust and replicable sex differences in the intrinsic brain function of autism. Mol Autism 12, 19, doi:10.1186/s13229-021-00415-z (2021).
34 Lai, M. C., Lombardo, M. V., Auyeung, B., Chakrabarti, B. & Baron-Cohen, S. Sex/gender differences and autism: setting the scene for future research. J Am Acad Child Adolesc Psychiatry 54, 11-24, doi:10.1016/j.jaac.2014.10.003 (2015).
35 Casey, B. J. et al. The Adolescent Brain Cognitive Development (ABCD) study: Imaging acquisition across 21 sites. Dev Cogn Neurosci 32, 43-54, doi:10.1016/j.dcn.2018.03.001 (2018).
36 Horien C, Mandino F, Corriveau A, Greene AS, O'Connor D, Shen X, Keller AS, Baller EB, Chun MM, Finn ES, Chawarska K, Lake EMR, Scheinost D, Satterthwaite TD, Rosenberg MD, Constable RT. Feature consistency in transdiagnostic connectome-based models of sustained attention and autism symptoms. medRxiv [Preprint]. 2026 Apr 7:2026.04.01.26349372. doi: 10.64898/2026.04.01.26349372.
Follow the Topic
-
Nature Mental Health
This journal takes an expansive view of the relationship between mental health and human health. It brings together innovative investigation of the neurobiological and psychological factors that underpin psychiatric disorders to contemporary work examining the effects of public health crises.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Health outcomes of adversity in early life
Publishing Model: Hybrid
Deadline: Sep 30, 2026






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in