Pisiform Homojunction with Energy Band Bending Induced via Co‑Implantation Design Enabling Fast‑Charging Sodium‑Sulfur Battery
Published in Bioengineering & Biotechnology and Materials
Introduction: The Sodium-Sulfur Frontier and the Shuttle Challenge
As the global energy landscape transitions toward renewable sources, the demand for high-performance and cost-effective energy storage systems has intensified. Sodium-sulfur (Na-S) batteries are considered one of the most promising candidates for grid-scale storage due to the high theoretical specific capacity of sulfur and the abundant, low-cost nature of sodium resources. However, the commercial realization of room-temperature Na-S batteries is severely hindered by the "shuttle effect" of soluble sodium polysulfides (NaPSs) and the sluggish conversion kinetics during the charge-discharge process. These issues lead to rapid capacity decay and poor rate performance, particularly under fast-charging conditions.
To address these fundamental hurdles, a research team led by Professor Qiyao Yu and Professor Jianguo Zhang has proposed a revolutionary "induced-homojunction" concept. Their work, published in Nano-Micro Letters, demonstrates how a p-n Mo2C homojunction/carbon composite can dramatically accelerate polysulfide conversion and stabilize battery performance.
The Innovation: Induced-Homojunction via Co-Implantation
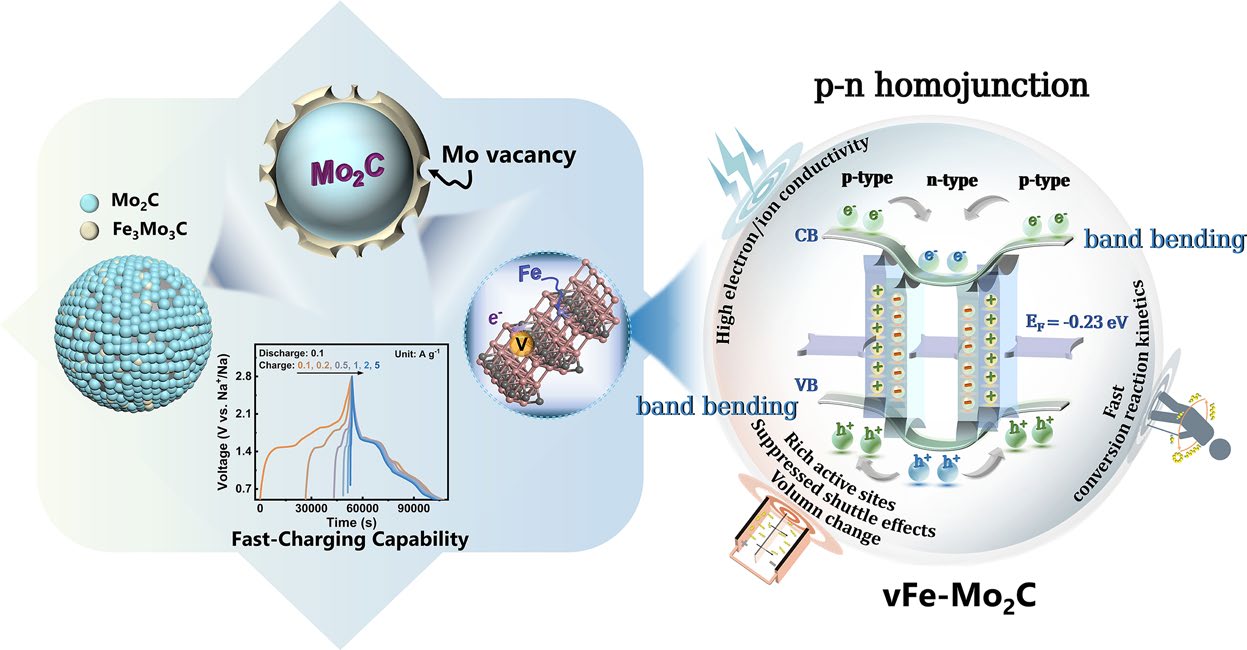
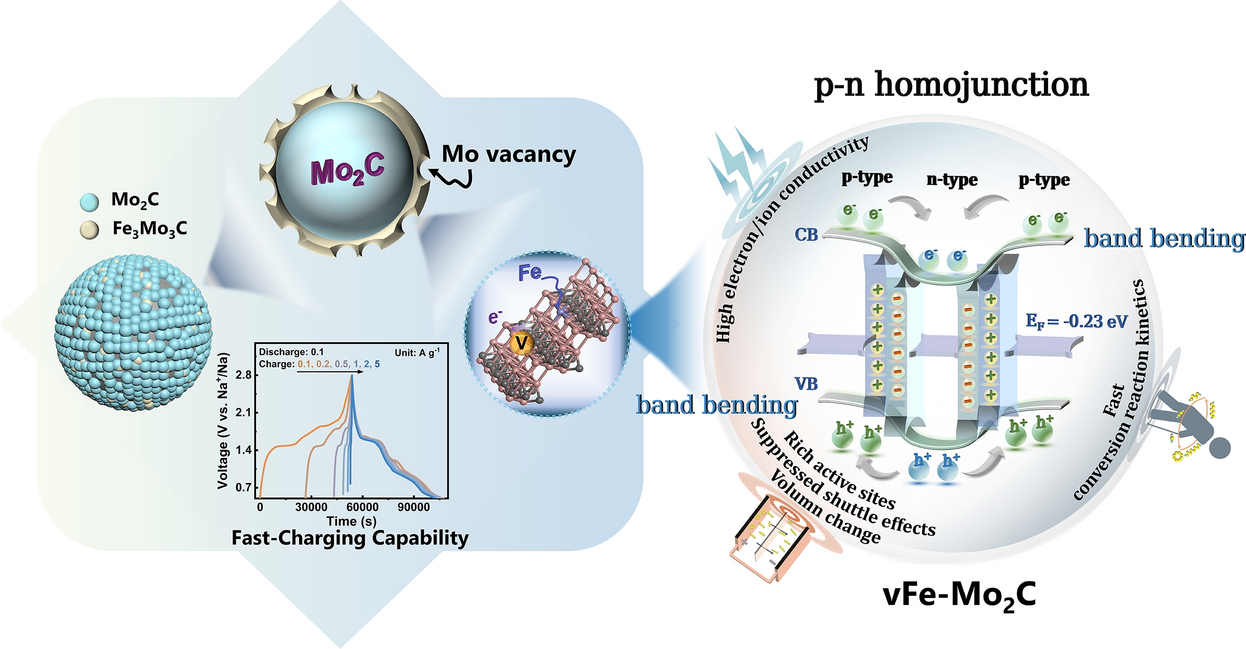
The core innovation of this study lies in the creation of a "pisiform" (pea-shaped) homojunction through a sophisticated co-implantation design involving iron (Fe) and molybdenum vacancies (Mo-vacancy).
Unlike traditional heterojunctions that involve two different chemical compounds, this "induced-homojunction" is created within a single material—Mo2C. By precisely controlling the co-implantation of Fe atoms and the creation of vacancies, the researchers induced a local electronic phase transition, forming a p-n junction interface within the Mo2C nanoparticles. This homojunction design offers a more coherent interface with lower lattice strain compared to heterojunctions, facilitating more efficient charge carrier transport.
Mechanism: Energy Band Bending and Polysulfide Capture
The scientific secret to the performance of this material lies in the energy band bending induced at the p-n homojunction interface.
- Built-in Electric Field and Charge Transfer: The alignment of the Fermi levels between the p-type and n-type regions of the Mo2C creates a built-in electric field. This field acts as a powerful "driving force" for the transfer of electrons and sodium ions. In the context of Na-S batteries, this accelerated charge transfer is crucial for the rapid reduction of long-chain polysulfides into short-chain solid sulfides, which effectively suppresses the shuttle effect.
- Enhanced Adsorption and Conversion: The homojunction interface serves as a "chemisorption center." The redistribution of electron density at the junction provides optimized binding sites for NaPSs. This ensures that the soluble polysulfides are not only captured at the cathode but are also immediately converted through fast redox kinetics. DFT calculations supported these findings, showing a significant reduction in the activation energy for the conversion of Na2S4 to Na2
Fast-Charging Capability and Rate Performance
A standout feature of this induced-homojunction catalyst is its ability to support fast-charging. One of the primary weaknesses of standard Na-S batteries is that their discharge capacity drops significantly when the charging rate is increased.
The Mo2C homojunction breaks this limitation. Because the built-in electric field significantly accelerates the sluggish sulfur redox reactions, the battery can be charged at high current densities without compromising the subsequent discharge capacity. This "charging-rate independent" behavior is a major step toward practical applications where rapid refueling or charging is required, such as in heavy-duty electric transport or grid stabilization.
Structural Integrity and Long-term Stability
The "pisiform" structure, where the homojunction particles are embedded within a protective carbon matrix, provides excellent structural stability. The carbon network not only enhances the overall conductivity of the cathode but also acts as a secondary physical barrier against polysulfide dissolution.
Furthermore, the homojunction interface itself is more resistant to the mechanical stress induced by the volume expansion of sulfur during sodiation. This structural robustness allows the battery to maintain ultra-high capacity retention even after hundreds of intensive cycles, proving its viability for long-duration energy storage.
Conclusion and Future Outlook
The introduction of the "induced-homojunction" concept marks a significant advancement in the field of battery sulfur cathodes. By moving beyond simple catalytic additives to sophisticated electronic-level interface engineering, the researchers have provided a powerful tool to overcome the kinetic and stability issues of Na-S systems.
This design philosophy—modulating energy bands through vacancy and dopant co-implantation—is highly generalizable. It could potentially be applied to other "shuttle-prone" systems, such as lithium-sulfur or lithium-selenium batteries. As researchers continue to refine these atomic-level designs, the goal of safe, low-cost, and fast-charging sulfur-based energy storage moves closer to reality.
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in