Reading the Cell Membrane’s "Molecular Mirror" with HSI and AI
Published in Bioengineering & Biotechnology, Materials, and Cell & Molecular Biology
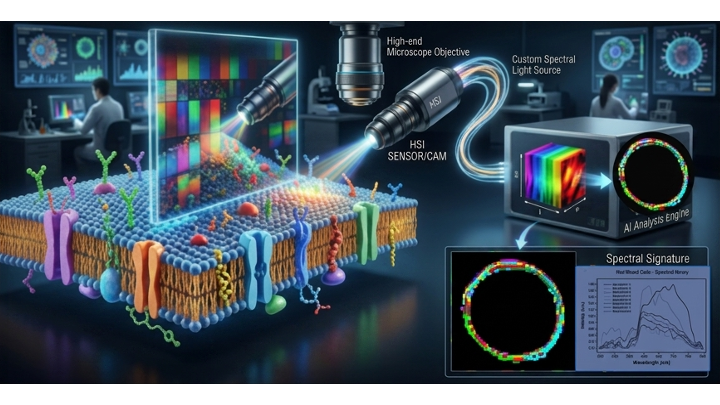 The story began almost a decade ago with a simple yet profound question: Could the physical light scattering of a cell tell us about its internal molecular health?
The story began almost a decade ago with a simple yet profound question: Could the physical light scattering of a cell tell us about its internal molecular health?
We started by using Hyperspectral Imaging (HSI) to analyze red blood cell (RBC) membranes under dark-field microscopy. The method was deceptively simple—requiring only a single drop of blood without labels or probes—yet it offered 2-nanometer precision. We observed that these discoid particles scattered light in a way that formed a consistent spectral fingerprint (composed of 8 spectral endmembers) in healthy subjects.
However, when we looked at RBCs from children with Autism Spectrum Disorder (ASD), the fingerprints changed. While we reported these initial findings in Scientific Reports (2017), the "why" remained elusive.
The turning point came when I began comparing the light interaction on the membrane to the behavior of light passing through the cell of a polarimeter. I realised that HSI wasn't just taking a picture; it was measuring the chiral organization of the membrane. To respect biophysical laws, this organization must be both flexible and constant. While the biochemistry of membrane homeostasis was well-understood, measuring it through light was entirely new.
With support from Cytoviva and an interdisciplinary team—ranging from neuropsychiatrists and biochemists to AI experts—we set out to prove this link. We created a "stress model" and at a low dose of hydrogen peroxide, changes in RBC membrane lipids and HSI signatures were not detectable. At 1.5% concentration, the RBCs remained structurally intact, yet we detected a distinct shift in the HSI signature. This was the "smoking gun": oxidative stress was reorganizing themolecular architecture of the membrane without destroying the cell, and the light was capturing it. When we applied this model to clinical samples, the results were striking. We identified "oxidative stress signatures" specifically in the ASD group. By feeding these complex HSI signatures into an AI-aided analysis platform, we achieved an identification of ASD subjects with accuracy of >93%.
This is the story of a successful transition from a "serendipitous discovery" to a "validated multidisciplinary breakthrough."
This journey proves that the membrane is more than a boundary;it is a sensitive biosensor. By combining chemical intuition with advanced photonics and machine learning, we have opened a new door to non-invasive diagnostics, with predictive power to be assayed in large population studies, for complex health conditions affected by oxidative stress, such as neurological conditions.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in