24 hours later: using systems vaccinology to understand responses to self-amplifying RNA
Published in Bioengineering & Biotechnology and Immunology
Vaccination works by training the immune system to recognise parts of infectious micro-organisms, so that when we get infected with them we can better fight them off. This builds upon the immune system’s ability to remember what it has seen before. Training this memory takes some time from the initial encounter with the vaccine. Importantly, the events immediately after vaccination can shape the quality and quantity of the response.
These early responses to immunisation have been explored across a range of different vaccines. In our recent study ‘Systems vaccinology analysis of saRNA immunization identifies an acute innate immune signature correlated with adaptive immunity’ we measured the immune response at 24 hours to an RNA vaccine to explore how this might predict responses later on.
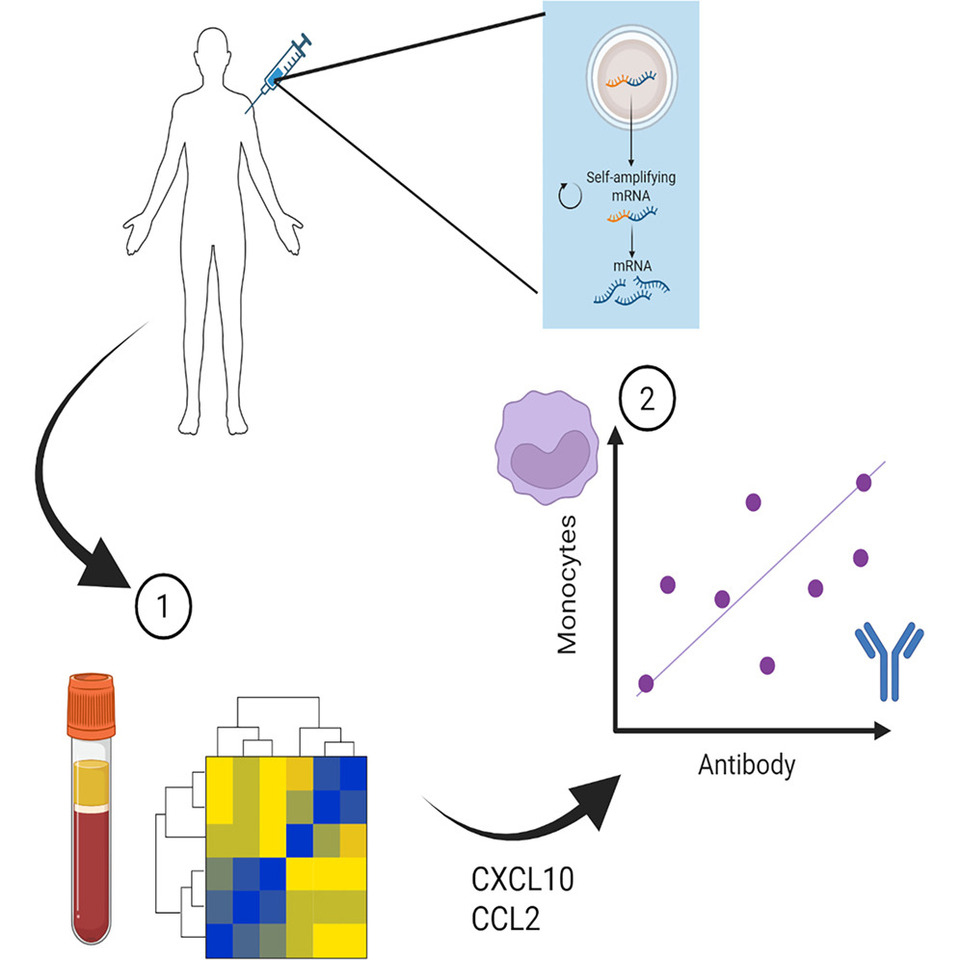
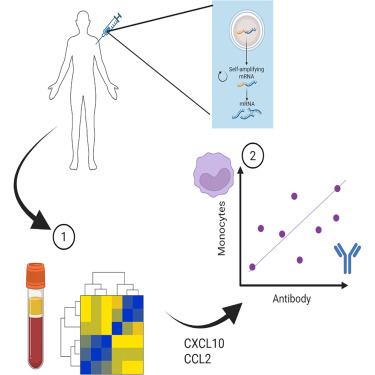
The vaccination part of the study took place as part of early trials of a self-amplifying RNA vaccine during the COVID-19 pandemic. This vaccine approach has potential advantages over the mRNA vaccines because the ability to amplify itself means the dose use can be much smaller. The study was performed towards the end of 2020 when the licensed vaccines were beginning to be offered widely to people. This reduced the number of people eligible for the study – because we wanted to evaluate responses in people who had not been vaccinated before. But we were able to enrol a small number of volunteers and undertook the study.
We used an approach called systems vaccinology. Which is a sciencey way of saying we measured lots of stuff and then looked for associations in the data. Blood was collected 24 hours after the initial injection. And three categories of immune markers were measured: RNA, protein and cells. The RNA gives a global picture of everything that has changed, the proteins focuses in on the way that immune cells communicate with one another and the cells gives a snapshot of what immune cells are moving from one place to another. These measurements because they are taken very soon after immunisation tell us about the short-term reaction to the vaccination, but the important thing is then to link them to the outcomes of vaccination. To do this we took a second blood sample several weeks later to measure whether the vaccine had indeed trained a memory response that could help prevent future infections.
We observed that immunisation led to significant changes in immune signalling in the blood. When the changes in RNA were profiled, there was a significant increase in genes linked to the immune response. Many of these fell in a family called the type I interferon pathway. This was not entirely surprising, RNA vaccines are made of RNA (#spoiler), so are many viruses; the type I interferon pathway is a programmed package of genes that fight viruses. We also saw an increase in genes that encode signalling molecules – particularly those that instruct immune cells to move from one place to another. These are called chemokines.
Having looked at the broad RNA picture, we focused down on proteins (as a quick reminder, RNA encodes proteins, proteins do the functional stuff). We saw similar increases in signalling molecules at the protein level in the blood, with increases in a chemokine called CCL2. One of the important functions of the immune system is to direct white blood cells (immune cells) to where the infection is occurring. CCL2 encourages the movement of a family of cells called monocytes, which have the ability to carry vaccine from one place to another and then engage with the cells that form the immune memory (called lymphocytes). Matching the increase in CCL2, we also saw an increase in the cells they recruit (monocytes) in the blood.
This demonstrated that the vaccine was somehow engaging the immune response, but did it mean anything with regards the training response? Well of course it did – otherwise I wouldn’t be writing about it. When we compared the magnitude of the CCL2 response in the blood with the magnitude of the antibodies targeting SARS-CoV-2 spike protein (the vaccine payload). People who had more CCL2 had more antibody.
So why is this important? It allows us to make predictions of how well a vaccine will work as soon as 24 hours after immunisation. It also enables us to design vaccines – if we can learn how to increase the induction of CCL2, we might improve the strength of the response. Of course this work was a massive team effort – and we owe a great debt to the volunteers, who during a time of considerable uncertainty undertook a study to help move us closer to having a working COVID vaccine.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in