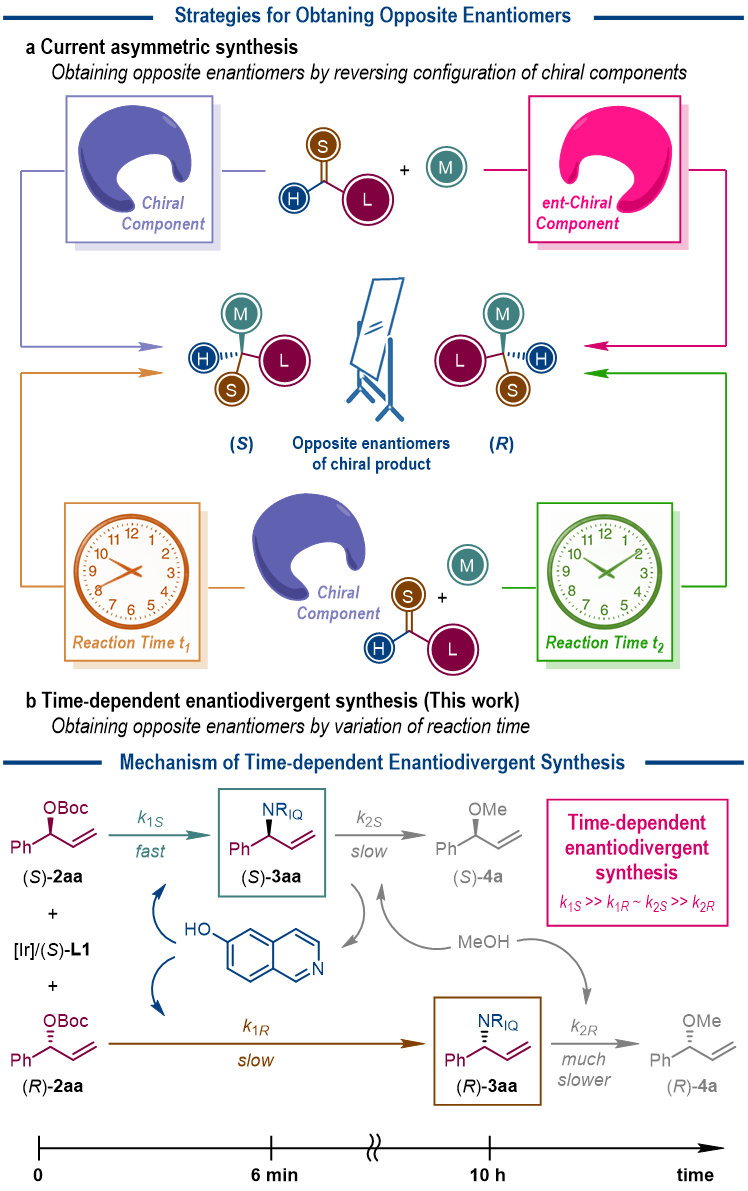
The homochirality is a fundamental phenomenon of Nature. It is well known that a pair of enantiomers of given chiral molecules will probably exert different influences in living organism. People have learned a lesson from the thalidomide tragedy on the importance of evaluating both enantiomers of any chiral drug candidate before it comes into the market. In this regard, the preparation of both enantiomers of chiral molecules is highly demanding and thus a prominent task in asymmetric synthesis.
In asymmetric synthesis, certain chiral component in the reaction system, in any form of chiral substrate, auxiliary, reagent, catalyst or ligand, is required for the chirality transfer or amplification process. It is the common strategy to switch the absolute configuration of the chiral component if the opposite enantiomer of the target chiral molecules is expected. However, the situation will be complicated when these chiral components are derived from natural sources with only one configuration available.
Recently, the research group led by Prof. Shu-Li You at Shanghai Institute of Organic Chemistry, Chinese Academy of Sciences disclosed an unusual phenomenon in asymmetric catalysis, where the reaction time is found as the controlling factor to determine the absolute configuration of the chiral products. More specifically, in the Ir-catalyzed asymmetric allylic amination reactions with the Carreira phosphoramidite ligand [in (S) configuration] as the only chirality source, the target allylic amination products are isolated as the (R)-isomers when the reactions are quenched at 5~10 min, but as the (S)-isomers when the reactions are quenched at 8~11 h. The compatible amine nucleophiles include 6- or 8-hydroxylisoquinolines and a series of aniline derivatives.
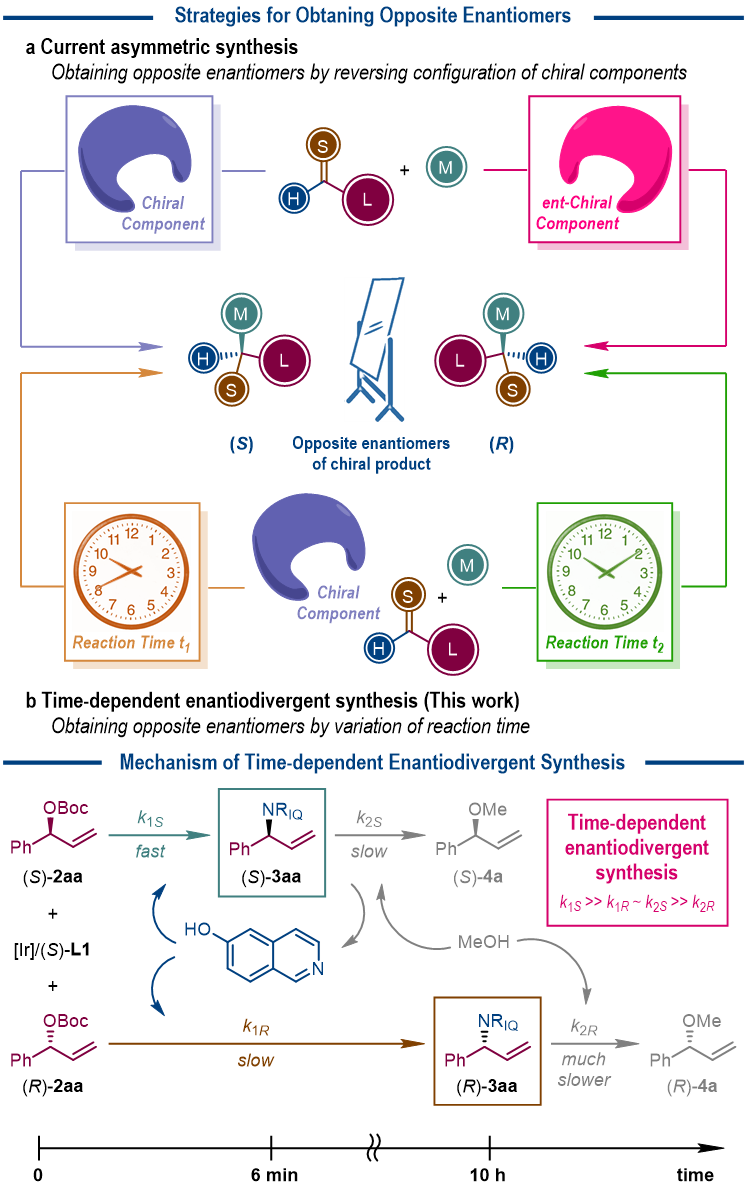
Comprehensive mechanistic studies reveal that the reason behind this unique time-dependent enantiodivergent synthesis phenomenon is that the same chiral Ir-catalyst promotes sequential kinetic resolution of allylic precursors and amination products with amine nucleophiles and the solvent methanol as the reaction partner, respectively. The appropriate permutation of the four individual reaction rates (those of two enantiomers of the allylic precursors and two enantiomers of the allylic amination products) guarantees the isolation of each enantiomer of the allylic amination products in a highly enantioenriched form at different reaction time.
“This is an unprecedented phenomenon in asymmetric catalysis,” said Prof. Shu-Li You when asked about the significance of this finding, “it alerts our community the critical role of reaction time when we are doing asymmetric catalytic reactions. We are currently working on expanding the scope of this reactivity pattern, and checking whether it will contribute to the fast generation of useful chiral molecule libraries for medicinal chemistry.”
The results of this study was just published on Nature Chemistry under the title of “Time-dependent enantiodivergent synthesis via sequential kinetic resolution”. (https://www.nature.com/articles/s41557-020-0489-1)
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in