Adipose as a driver, not a bystander in obesity-related erectile dysfunction
Published in General & Internal Medicine and Anatomy & Physiology
Explore the Research
Just a moment...
dom-pubs.onlinelibrary.wiley.com
Review paper
Adipose as a Driver, Not a Bystander: A Modern Synthesis of Obesity-Related Erectile Dysfunction
Why we wrote this paper
Obesity and erectile dysfunction are often linked through familiar explanations: poor vascular health, insulin resistance, low testosterone, and inflammation.
All of those matter. But they still leave an important question: why does erectile dysfunction sometimes appear early, even before severe metabolic disease is obvious?
That question pushed us to look more closely at adipose tissue itself.
The main idea
Adipose tissue is not just fat storage. It is an active signaling organ.
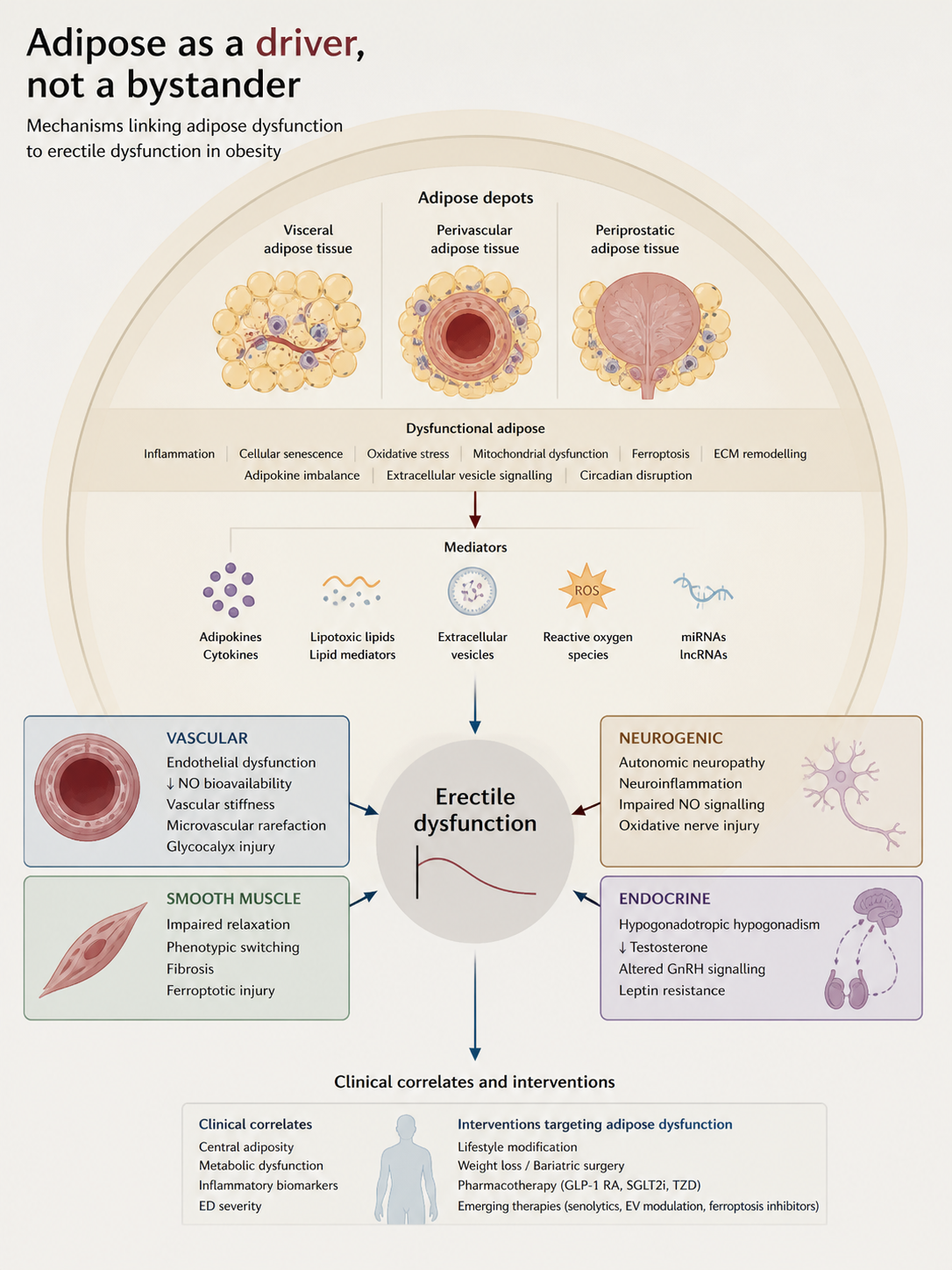
In obesity, visceral, perivascular, and periprostatic fat can release inflammatory cytokines, adipokines, oxidized lipids, and extracellular vesicles. These signals may reduce nitric oxide availability, increase vascular stiffness, disrupt testosterone regulation, and promote cavernosal fibrosis.
In simple terms, unhealthy adipose tissue can help create the conditions that make normal erectile function harder to maintain.
Why location matters
Not all fat behaves the same way.
Visceral fat can affect the whole body through inflammation, insulin resistance, and hormone disruption. Perivascular fat sits close to blood vessels and may directly influence vascular tone and endothelial function. Periprostatic fat may also contribute through local inflammatory and neurovascular signals.
This is why central adiposity may tell us more about erectile dysfunction risk than BMI alone.
What stood out
The evidence pointed to a network, not one single pathway.
Adipose dysfunction may connect several processes that are usually discussed separately: endothelial dysfunction, oxidative stress, testosterone disruption, vascular stiffness, smooth muscle remodeling, and fibrosis.
Newer mechanisms such as cellular senescence, extracellular vesicle signaling, ferroptosis, mitochondrial dysfunction, and glycocalyx injury may also help explain how obesity reshapes erectile biology.
Why this matters
Most ED treatments focus on improving the immediate erectile response. That is useful, but it does not always address the upstream biology.
If adipose dysfunction is part of the cause, then improving adipose health may help modify the disease process itself. Weight loss, exercise, bariatric surgery, GLP-1 receptor agonists, SGLT2 inhibitors, and future adipose-targeted approaches may all be relevant, not just for metabolism but also for erectile function.
What remains unclear
The biggest gap is direct evidence from penile and pelvic fat depots. Much of what we know about perivascular adipose tissue comes from other vascular beds, so penile-specific studies are still needed.
Future work should combine imaging, erectile function testing, adipose biomarkers, extracellular vesicle profiling, and molecular studies of inflammation, senescence, and lipid stress.
Closing thought
This paper started with a simple idea: adipose tissue may not just accompany obesity-related erectile dysfunction. It may help drive it.
Seeing adipose tissue as an active regulator gives us a broader way to study, detect, and eventually treat obesity-related erectile dysfunction.
Reference
Sailis AB, Mat Noh MA. Adipose as a Driver, Not a Bystander: A Modern Synthesis of Obesity-Related Erectile Dysfunction. Diabetes, Obesity and Metabolism. 2026;0:1-19. https://doi.org/10.1111/dom.70818
Full text available here: link





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in