microRNAs as hidden regulators of testosterone disruption in e-cigarette exposure
Published in Biomedical Research and Paediatrics, Reproductive Medicine & Geriatrics
Explore the Research
 sciencedirect.com
sciencedirect.com
ScienceDirect
Please contact our support team for more information and provide the details below.
Review article
MicroRNA-mediated disruption of testosterone signaling associated with e-cigarette exposure
Why we wrote this paper
Most discussions about e-cigarettes and male reproductive health focus on oxidative stress, inflammation, or direct cellular toxicity. Those mechanisms matter, but they do not fully explain how exposure signals could be translated into coordinated endocrine effects across the HPG axis. That gap led us to look more closely at microRNAs.
Why microRNAs changed the question
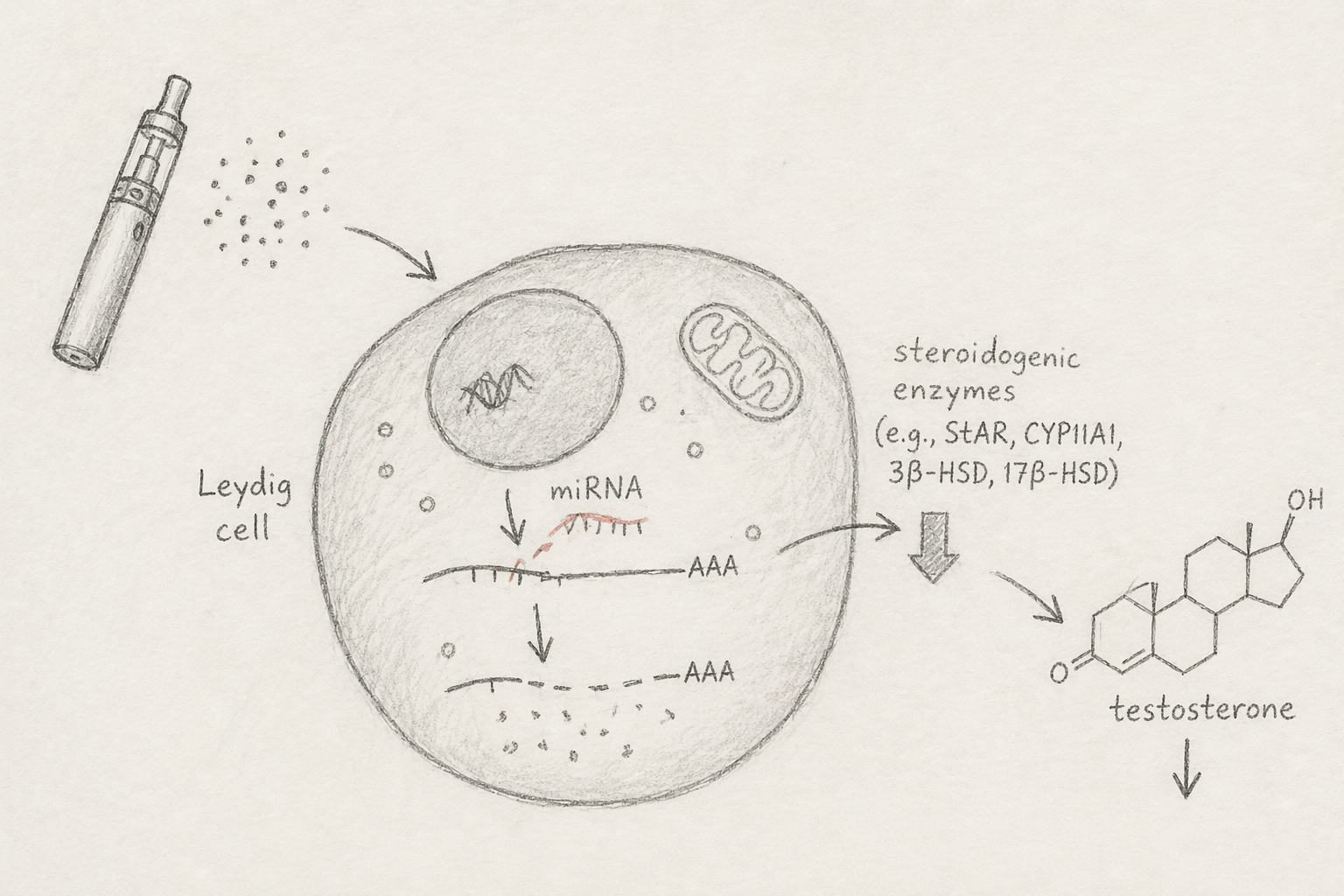
MicroRNAs are small regulators, but their effects are not small. They can fine-tune GnRH signaling, gonadotropin output, Leydig cell function, cholesterol transport, steroidogenic enzyme expression, and androgen signaling. This makes them plausible integrators of e-cigarette exposure and testosterone dysregulation.
Instead of asking only whether e-cigarettes damage the testis, we began asking whether they may reprogram the regulatory networks that control testosterone synthesis.
What stood out in the evidence
Direct evidence in e-cigarette users is still limited, but the surrounding biology is compelling. Multiple aerosol-related constituents, including nicotine, aldehydes, metals, and particulates, are already known to perturb stress-responsive miRNA pathways. At the same time, many miRNAs have established roles in steroidogenesis and HPG axis regulation.
What emerged was not a single linear mechanism, but a converging pattern. The same exposure classes present in e-cigarette aerosols can affect the same regulatory layer that controls testosterone-related pathways.
A broader framework
One of the central ideas in this paper is that miRNAs may act as an intermediate regulatory layer between inhaled exposure and endocrine outcome. In that sense, the effect is not only local. Lung injury, systemic inflammation, circulating extracellular vesicles, and direct testicular exposure may all converge on miRNA networks that shape steroidogenic function.
This offers a more integrated model of how vaping-related signals could move from the airway to the testis and eventually alter testosterone biology.
Why this matters
Hormone levels do not always change early, even when biological stress is already present. That is why miRNAs are interesting. They may capture disruption earlier than conventional endocrine endpoints and may help explain why human findings can appear muted or inconsistent despite clear mechanistic plausibility.
In that sense, miRNAs are not just mechanistic candidates. They may also become useful biomarkers.
What remains unresolved
This paper is hypothesis-generating by design. We still lack direct human evidence linking e-cigarette exposure, altered testicular or circulating miRNA profiles, and measurable testosterone disruption. That is the key translational gap.
But the framework is now testable. The candidate pathways exist. The candidate miRNAs exist. The exposure biology exists. What is missing is direct validation.
Where this leads next
Future work needs to connect exposure, miRNA signatures, steroidogenic targets, and endocrine outcomes in the same study design. That includes biomarker-based exposure assessment, functional validation of candidate miRNAs, and longitudinal human work that can distinguish early regulatory disruption from later hormonal decline.
Closing thought
This paper began with a simple idea: perhaps e-cigarettes do not influence testosterone only by injuring cells, but also by altering the small RNA networks that tell those cells how to respond.
If that is true, then miRNAs may be one of the missing links in understanding vaping-associated male reproductive toxicity.
Reference
Sailis AB, Mat Noh MA, Leo BF, Faruqu FN, Yee A, Sim MS. MicroRNA-mediated disruption of testosterone signaling associated with e-cigarette exposure. Environmental Toxicology and Pharmacology. 2026. https://doi.org/10.1016/j.etap.2026.104994
Full text available here (until 2 May 2026): link




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in