Alcohol Detoxification with Food Amyloid Fibrils and Iron
Published in Materials and Agricultural & Food Science
Alcohol-related chronic diseases -and health impact in general- cause an average of more than 3 million deaths per year and more than 280 million individuals with alcohol-use disorders, according to the 2018 WHO Global status report on alcohol and health. Beyond these numbers, non-communicable diseases and other indirect issues related to alcohol consumption further worsen the full picture. With such an alarming global impact on society and health, strategies to: i) decreasing the effects of alcohol via prophylactic strategies in the short term and, ii) alleviating the chronic pathologies associated with excessive alcohol consumption remain a pressing need. Most of the approaches followed so far relate to the use of natural enzymes or bacteria to accelerate the metabolism of alcohol in the body; yet, these methodologies often result in the accumulation of toxic acetaldehyde, or, in the best case, user-dependent efficiencies in the metabolization of alcohol into less toxic by-products.
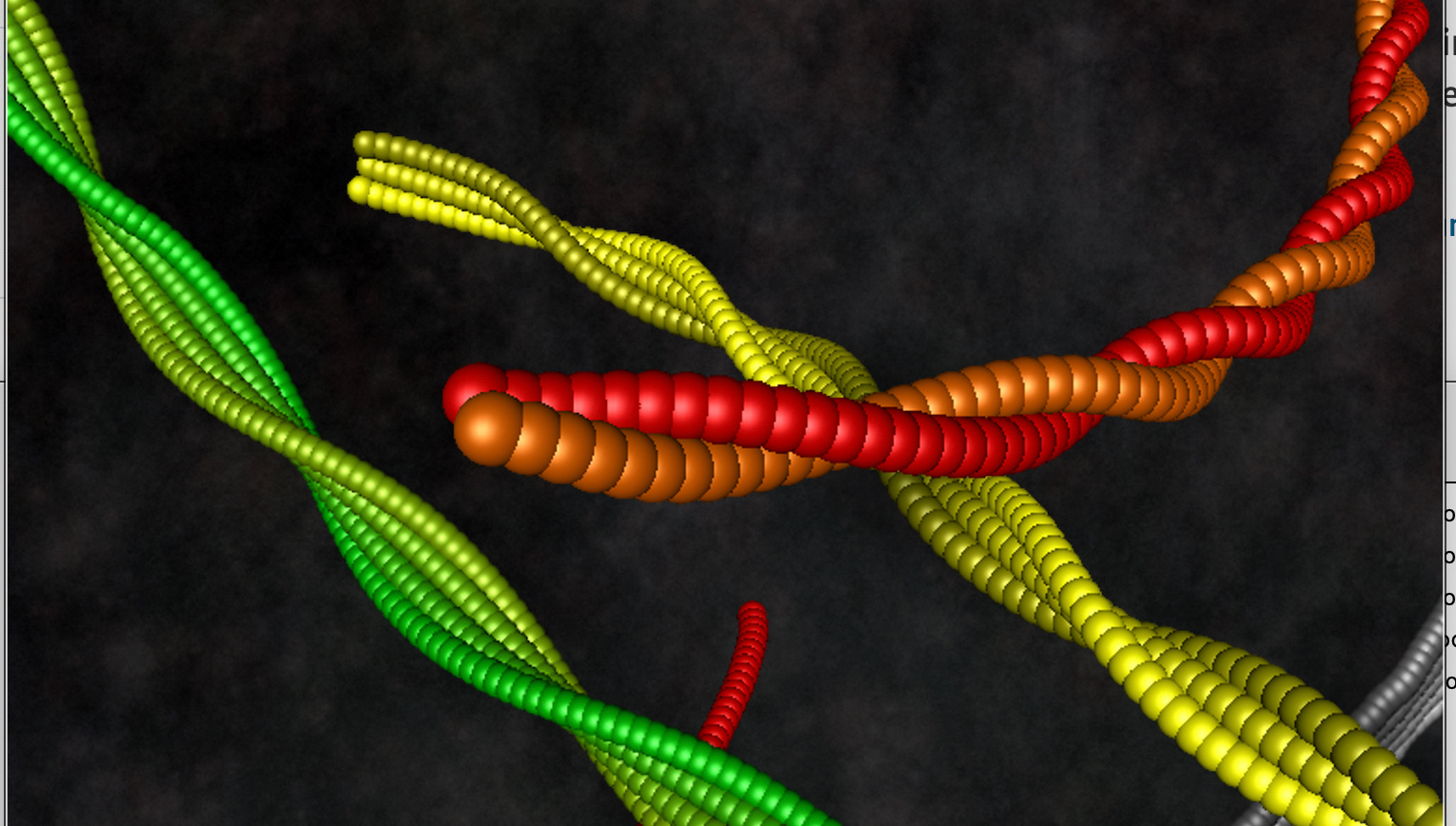
In our recent work1 we introduce a simple, safe, and very effective formulation for the metabolization of alcohol via heterogeneous catalysis performed by food amyloid fibril hydrogels combined with single-atom iron, acting as main catalyst. Our work builds on a previous technology developed at ETH Zurich, where food amyloid fibrils were combined with iron nanoparticles for iron fortification2. In that work, the iron nanoparticles decorating the food amyloid fibrils were digested down to the ionic form by the acidic pH present in the stomach, while enzymes in the gastro-intestinal (GI) tract would digest the food amyloid fibrils. That work, which served as an inspiration for our alcohol detoxification technology, provided iron in the form of nanoparticles, which although feature catalytic activity for alcohol detoxification, do not have the necessary turnover needed to metabolize alcohol at a speed sufficiently high to avoid its crossing into the bloodstream.
What we have now done1, is to switch to single atom iron-anchored food amyloids on one hand to increase the catalytic efficiency and, on the other hand, use amyloid hydrogels rather than single amyloid fibrils to delay their digestion over the GI tract. By doing so, we demonstrate both in-vitro and in-vivo the successful catalytic conversion of ethanol into non-toxic acetic acid at a speed sufficiently high to avoid most of the negative effects of alcohol ingestion, with surprisingly good results.
To start, we have benchmarked the superior turnover of this enzymatic reaction against robust protein-based controls and found an outstanding performance of our iron-anchored amyloid hydrogels, which is understood to stem from the fine atomic distribution of iron on the surface of the food amyloids fibrils, several times more efficient than the iron in form of nanoparticles onto the food amyloid fibrils. This design provides exceptionally high surface/volume ratio for both the iron atoms (compared to nanoparticles), and amyloid fibrils (compared to other proteinaceous aggregates or native proteins). Additionally, the hydrogel formulation itself slows down the digestion of the complex within the gastro-intestinal tract, thus extending the effect over time.
Most importantly, we show that this same formulation has a long-lasting therapeutic effect in-vivo, restoring body weight, blood composition and tissue histology back to normal standards, up to the point that tissues from healthy mice and mice having consumed alcohol together with our iron-based amyloid gel are nearly undistinguishable. These findings, together with the recent conclusion that food amyloid fibrils are safe nutrition ingredients3, may have far reaching consequences and open new avenues in fighting worldwide the long-lasting battle against alcoholism and alcohol-related diseases.
Although our findings are -so far- demonstrated in-vivo only in mice models, these results provide already compelling evidence in both therapeutic and prophylactic applications. As an illustrative case, it may be brought forward the behavior of mice swimming in a pool, under the effect of alcohol on one group, and alcohol plus our iron-amyloid hydrogel on the other group. As Figure 1 shows, under the effect of alcohol alone, mice are lost in their swimming paths, following long, random trajectories before exiting the pool. On the contrary, mice having consumed alcohol in combination with our hydrogel, manage to find the exit in a rapid and efficient way, similar to fully sober mice, opening the door even to recreational use of the present technology.
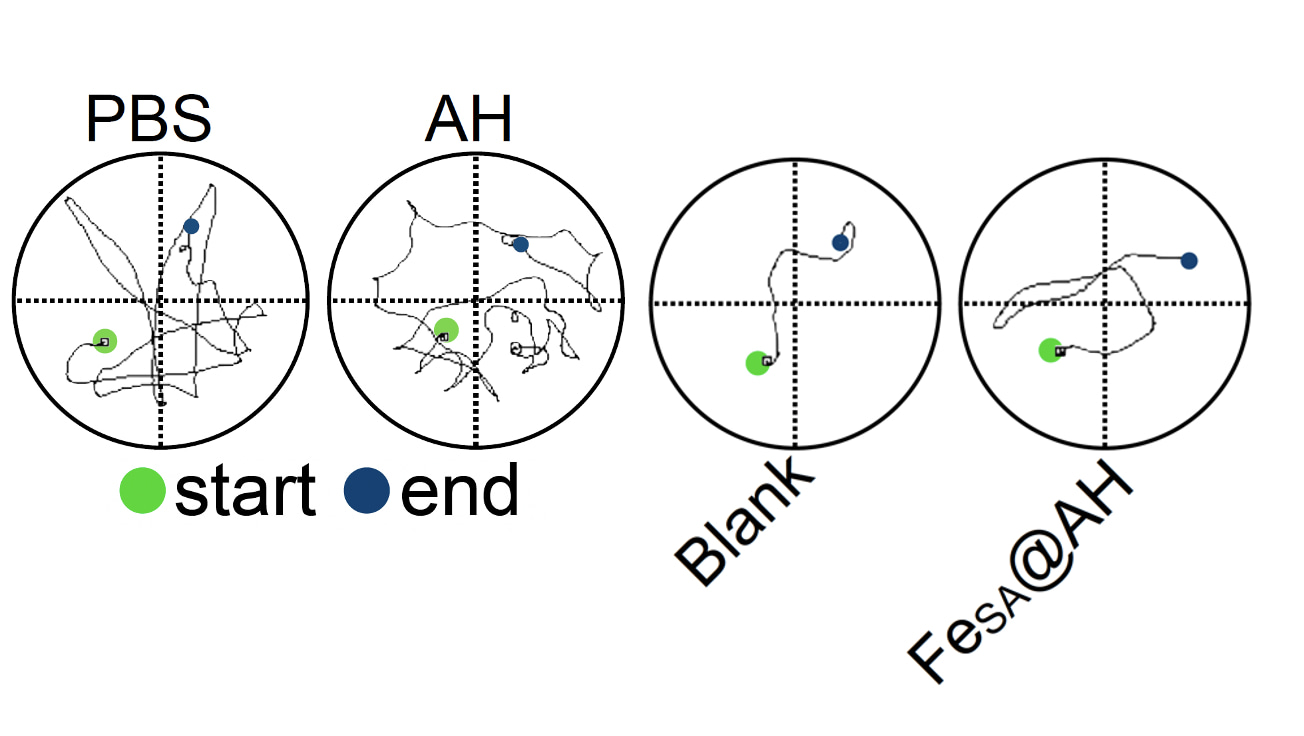
Figure 1: Representative trajectories in Morris water maze experiments for mice having followed different treatments: PBS buffer is the group of mice with acute ethanol exposure; AH is the group of mice having ingested ethanol plus the amyloid hydrogel, but without iron; blank is the sober mice group and FeSA@AH is the mice group having consumed alcohol and our iron-decorated hydrogel.
What is more, our technology may have also important implications for treating other diseases. For example, the hypothesis that the gut microbiota of people with non-alcoholic fatty liver disease (NAFLD) may produce enough ethanol to trigger this disease, is gaining growing consensus4. If this is confirmed further, our iron-anchored food amyloid hydrogels could have a therapeutic role even in treating this pathology which affect nearly 5% of human population5.
We envision a recreational, therapeutic, and prophylactic use of our iron-amyloid gel and we are looking forward to translating soon these results to human studies, in order to make this technology accessible to millions of people around the globe.
References:
- Su et al. Single-site iron-anchored amyloid hydrogels as catalytic platforms for alcohol detoxification, Nature Nanotechnology, (2024)
- Shen et al. Amyloid fibril systems reduce, stabilize and deliver bioavailable nanosized iron, Nature Nanotechnology, 12, 642–647 (2017), https://doi.org/10.1038/nnano.2017.58
- Xu et al. Food amyloid fibrils are safe nutrition ingredients based on in-vitro and in-vivo assessment, Nature Communications, 14, 6806 (2023), https://doi.org/10.1038/s41467-023-42486-x
- Meijnikman et al. Microbiome-derived ethanol in nonalcoholic fatty liver disease, Nature Medicine, 28, 2100, (2022), https://doi.org/10.1038/s41591-022-02016-6
- Riazi et al. The prevalence and incidence of NAFLD worldwide: a systematic review and meta-analysis. Lancet Gastroenterol Hepatol, 7, 851 (2022), https://doi.org/10.1016/S2468-1253(22)00165-0
Follow the Topic
-
Nature Nanotechnology
An interdisciplinary journal that publishes papers of the highest quality and significance in all areas of nanoscience and nanotechnology.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in