Application of wrist-wearable accelerometry for objective motor diary assessment in fluctuating Parkinson’s disease
Published in Neuroscience
In advanced Parkinson's disease (PD), motor complications affect a significant percentage of patients, becoming prevalent in about 50% after 5 years and 90% after 10 years of the disease. With disease progression, the therapeutic window of optimal response of Parkinsonian symptoms to dopaminergic treatment narrows. Initially, an oral substitution of levodopa t.i.d. suffices to control symptoms throughout the day. A narrower therapeutic window will eventually result in a shorter response of dopaminergic medication on Parkinsonian symptoms. In addition, the effect of dopaminergic medication might also exceed the upper margin of the therapeutic window, resulting in dyskinesia, usually at the peak of the medication’s plasma concentration. These response fluctuations between “Off” - the state when the medication effect is absent, “On” – the state where the medication is at its optimal level to control Parkinson symptoms, and “Dyskinetic” - the state when patients experience involuntary movements induced by dopaminergic treatment, disrupt daily life and reduce quality of life (QoL).
Clinical assessment and drug development for response fluctuations rely heavily on rating instruments such as the PD home diary, the Unified PD Rating Scale (UPDRS), or the modified Abnormal Involuntary Movement Scale (mAIMS). These assessments, while standard, suffer from biases and errors as patients may forget to record their motor state. In addition, non-motor symptoms can influence the judgement on motor symptoms, resulting in an inaccurate description of the motor state at the time. Recent studies have shown that the PD home diary inadequately reflects actual motor states when compared to clinical observation, highlighting the need for continuous and objective monitoring of motor function in PD.
Advances in digital health technologies (DHTs) offer user-friendly wearables, including smartwatch-based applications with inertial measurement unit sensors (e.g., accelerometers, gyroscopes) and long battery life, suitable for continuous monitoring of motor symptom severity and fluctuations. While the feasibility of using DHTs for measuring motor function and fluctuations has been demonstrated, clinical evidence suggests that continuous, objective monitoring using wearable biosensors can improve clinical decision-making for and functional outcomes in PD patients.
In this context, we present the results of the VALIDATE-PD study, a prospective observational multi-centre cohort study on 63 PD patients with motor fluctuations. Data from wrist-worn accelerometers were compared to the patients’ PD home diary entries and ratings from an external rater (Fig. 1).
Our primary goal was to determine whether a wearable accelerometer-based digital Parkinson's Motor Diary (adPMD) using the Parkinson's Kinetigraph (PKG®) could accurately capture the clinical motor state when compared to simultaneous half-hourly clinical examinations by experienced raters, utilizing the PD home diary as a reference. The PD home diary entries from day 0 were used for adPMD calibration. On days 1 and 2, patients were additionally observed by an experienced PD rater in half-hour intervals, independently rating the patients’ motor states as Off, On, or dyskinetic. A subcohort performed the 7-meter Timed-Up-and-Go test (7m-TUGT).
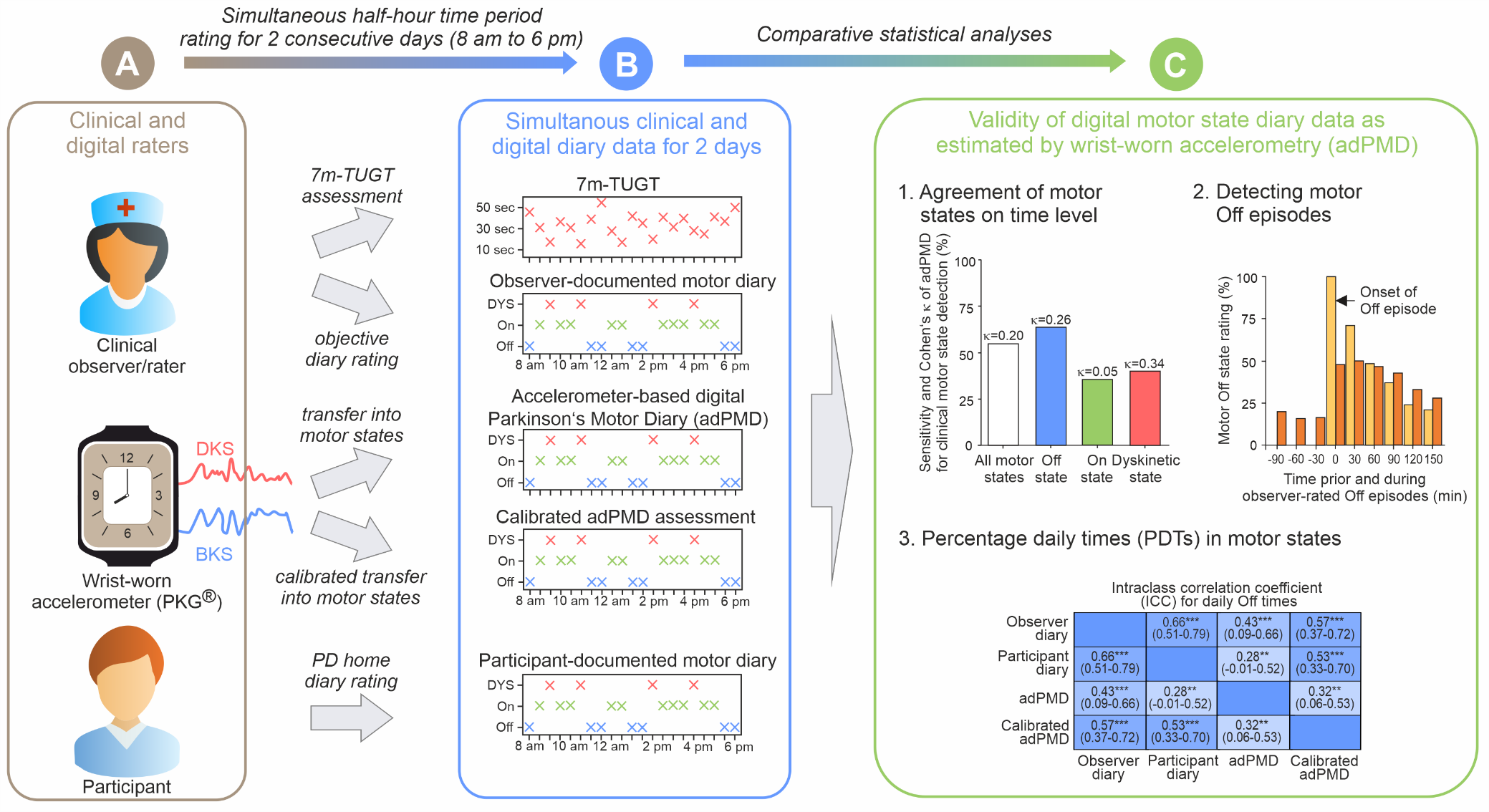
Figure 1. Graphical synopsis of application study of wrist-wearable accelerometry for objective motor diary assessment in fluctuating Parkinson’s disease.
Temporal agreement of adPMD motor state classification with clinical diary data
To assess the temporal agreement between adPMD (accelerometer-based digital Parkinson's Motor Diary) and clinical diary data, we examined the sensitivity of adPMD in recognizing motor states as rated by clinical observers at group level (Fig. 2a). Our findings indicate that adPMD ratings accurately identified 55% of all motor states. Specifically, it correctly recognized 64% of observed Off states, but only 36% of On states, and 40% of Dyskinetic states. The corresponding Cohen's κ values ranged from 0.05 for the On state to 0.34 for the Dyskinetic state. Notably, adPMD identified an On state in 21% of intervals when the clinical observer had noted an Off state (Fig. 2b).
Furthermore, when comparing adPMD data to the patients’ PD home diary entries (Fig. 2c), adPMD accurately recognized 40% of all motor states. Specifically, it correctly identified 57% of patient-rated Off states, 34% of On states, and 34% of dyskinetic states.
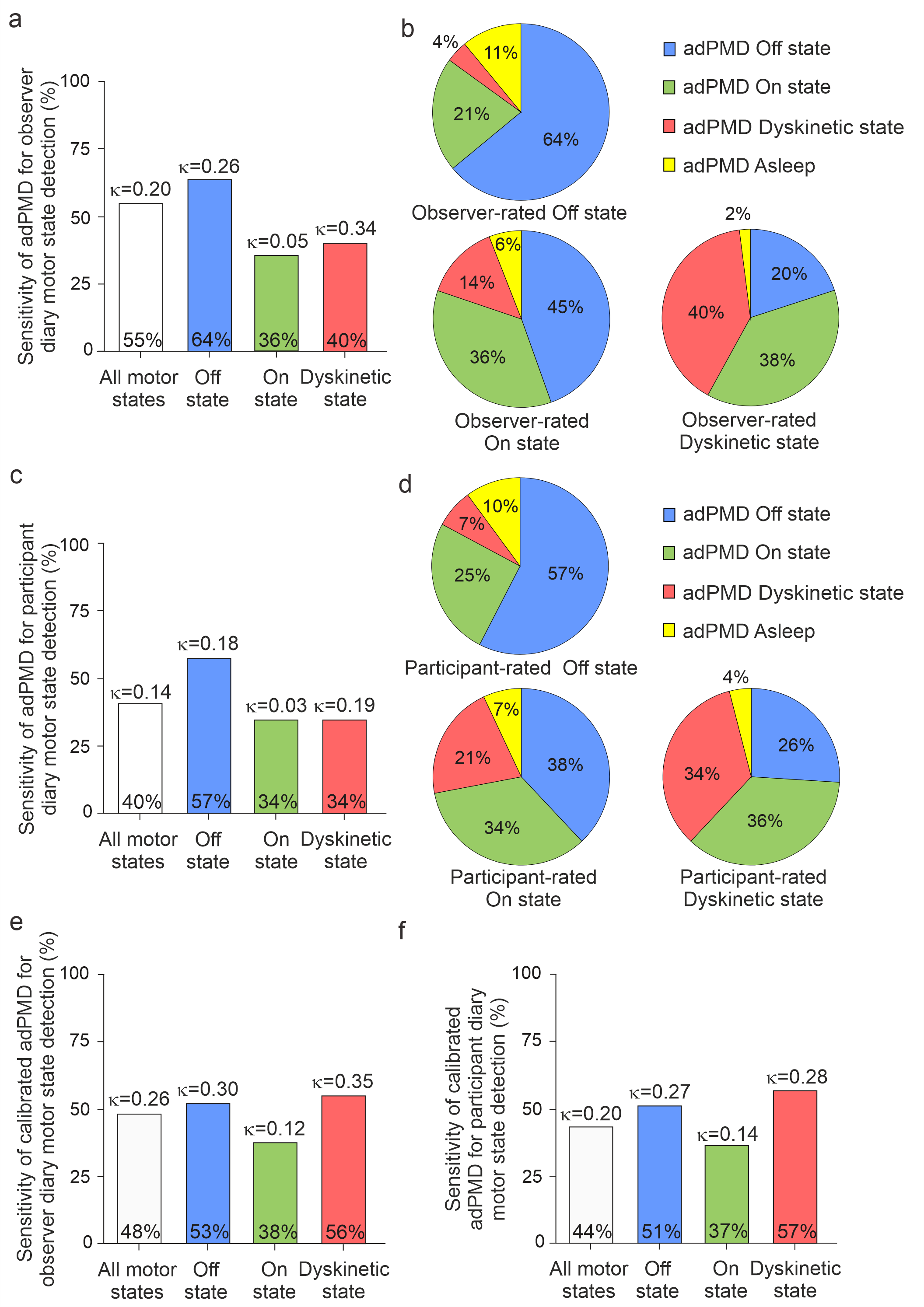
Figure 2. Temporal agreement of accelerometer-based digital Parkinson's Motor Diary (adPMD) data and observer- and participant-documented diary motor states on the half-hour time period level.
Agreement of adPMD classification with clinical ratings during Off episodes
Rather than comparing adPMD and diary scores from half-hour periods, the timing of adPMD scores were synchronized with Off episodes. Off episodes were defined as an Off state period of at least 0.5 hours following a motor On state period of at least 1.5 hours (Fig. 3). We did not observe relevant changes of median bradykinesia scores at the beginning of the Off episodes with an increase of 12% (IQR: -9 to 53%) for observer-reported Off episode (P=0.615; Mann-Whitney U-test). Consistently, there was no change in 7m-TUGT as a quantitative clinical bradykinesia outcome measure.
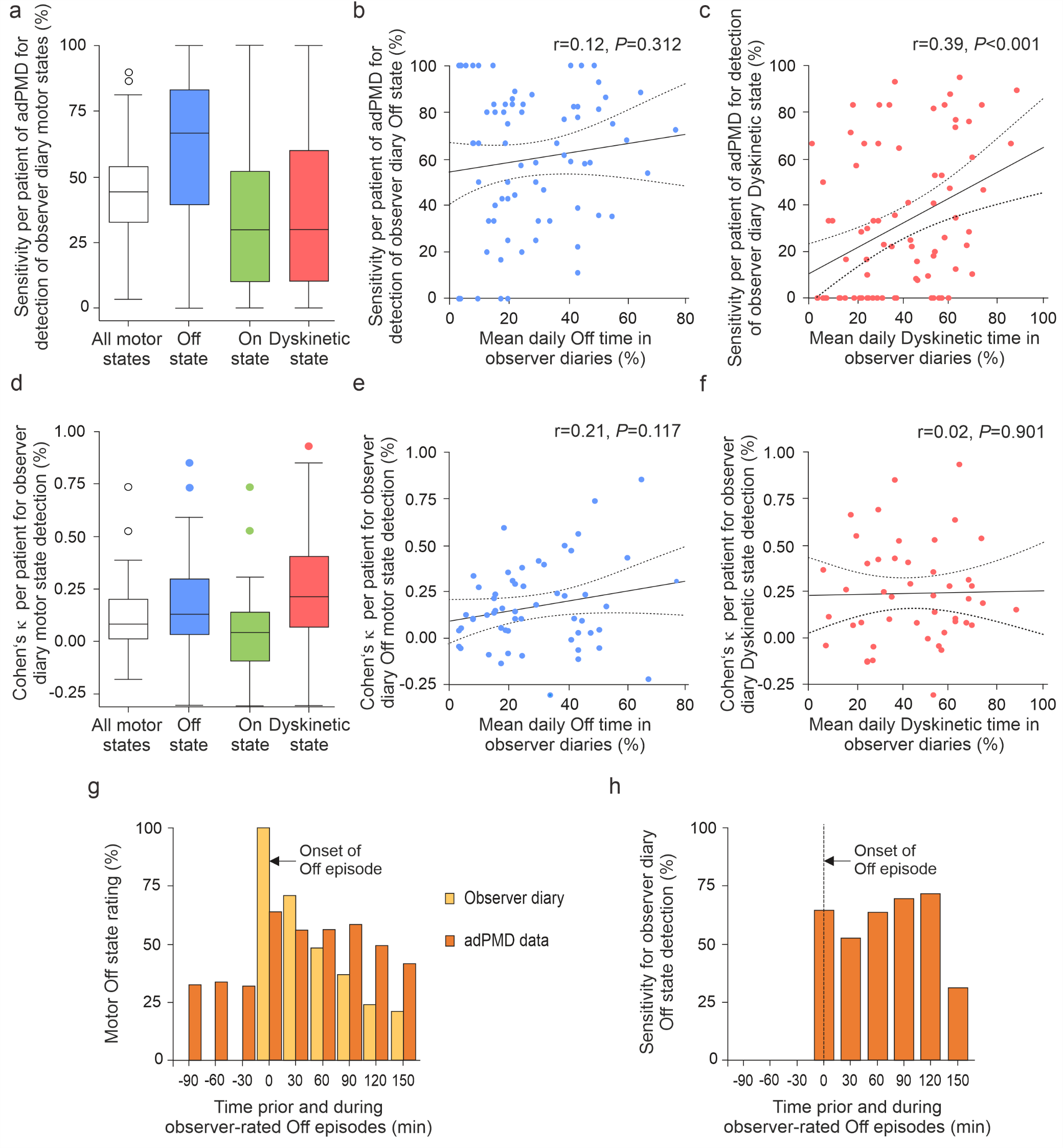
Figure 3. Temporal agreement between accelerometer-based digital Parkinson's Motor Diary (adPMD) ratings and observer-documented motor states on the participant level and during the time course of Off episodes.
Concluding remarks
In summary, our results show that the accuracy between adPMD and external observer ratings were higher than between adPMD and patients’ PD home diary ratings. Synchronizing adPMD data to the time of diary entries and applying an index day for calibrating the adPMD threshold to patients’ PD home diary ratings did not increase the concordance between these two. Calibration of adPMD data to the external observer ratings however increased the concordance on dyskinetic state detection to a moderate level. Future studies in larger cohorts are warranted to confirm these results on thresholding for PKG® data transfer into motor state diary data, and to provide evidence for the adPMD using the PKG® device as a suitable trial endpoint.
This post was originally drafted by co-authors Alexander Bremer and Florin Gandor and approved by all authors of our paper before publication.
Follow the Topic
-
npj Digital Medicine
An online open-access journal dedicated to publishing research in all aspects of digital medicine, including the clinical application and implementation of digital and mobile technologies, virtual healthcare, and novel applications of artificial intelligence and informatics.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
Evaluating the Real-World Clinical Performance of AI
Publishing Model: Open Access
Deadline: Jun 03, 2026
Impact of Agentic AI on Care Delivery
Publishing Model: Open Access
Deadline: Jul 12, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in