From a Solubility Project to a Dual-Targeted Breakthrough
In the battle against colorectal cancer (CRC), chemoresistance remains a formidable barrier that continues to challenge treatment success. The frontline agent irinotecan (CPT-11), a topoisomerase I (TOP1) inhibitor, carries fundamental pharmacokinetic limitations including inefficient hepatic activation, poor solubility, and significant dose-limiting toxicity. Moreover, cancer cells often adapt by enhancing DNA repair and sustaining telomere maintenance, allowing tumors to ultimately resist therapy.
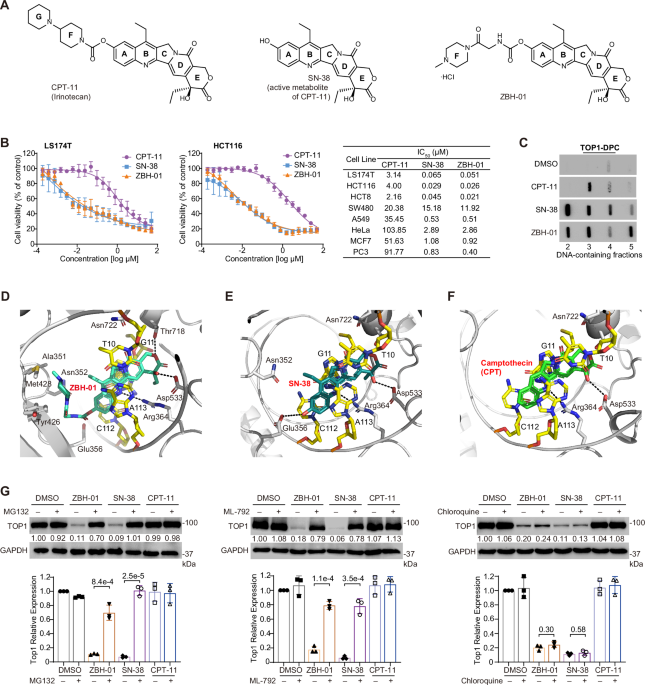
Our project began in 2014 with a clear pharmacological objective: to improve the aqueous solubility of SN-38, the active metabolite of CPT-11. Through structure-guided modification, we synthesized ZBH-01. The initial results were promising, showing a remarkable 2,600-fold increase in aqueous solubility while maintaining potent cytotoxicity across diverse cancer cell lines.
The pivotal moment, however, came when we tested ZBH-01 in chemoresistant models. In cisplatin-resistant CRC cells, it exhibited 14-fold greater efficacy than CPT-11 and 7-fold greater than SN-38. In 5-FU-resistant models, it was 61-fold more potent than CPT-11 and 2.4-fold stronger than SN-38. These dramatic results signaled something beyond solubility enhancement. ZBH-01 was engaging a fundamentally different biological mechanism.
Connecting the Dots: A Hypothesis Emerges
Returning to the literature, we recognized two converging threads. First, DNA G-quadruplexes, non-canonical nucleic acid secondary structures enriched in promoters of oncogenes such as hTERT and MYC, act as transcriptional switches. Second, both TOP1 and G-quadruplexes are concentrated in transcriptionally active genomic regions. This led to a compelling question: could the structural modifications that enhanced solubility have also granted ZBH-01 the ability to stabilize G-quadruplexes, effectively creating a dual-targeting molecule?
Validating the Dual-Target Mechanism
Subsequent experiments validated our hypothesis beyond expectations. Nuclear magnetic resonance and structural studies indicated that ZBH-01’s distinct crescent-shaped scaffold intercalates into the hTERT promoter G-quadruplex via π–π stacking. This binding stabilizes the structure and physically displaces transcription factors like SP1 and MYC, leading to strong suppression of telomerase (hTERT) expression. ZBH-01 was no longer just an optimized TOP1 inhibitor. It had revealed its true identity as a dual-targeted agent capable of simultaneously trapping TOP1-DNA complexes and stabilizing oncogenic G-quadruplexes.
Why Dual Targeting Represents a Therapeutic Advantage
Our comprehensive data demonstrate that ZBH-01 offers distinct advantages over conventional therapies. First, it delivers a synergistic mechanism of action by simultaneously inducing DNA damage through TOP1 inhibition while disrupting telomere maintenance and oncogenic transcription through the stabilization of G-quadruplexes. This combined genomic attack and epigenetic silencing leaves cancer cells with few escape routes, driving them toward senescence or death. Remarkably, its efficacy matched that of combining SN-38 with the classical G4 ligand TMPyP4, all within a single molecule.
Second, its exceptional potency against therapy-resistant cells likely arises from this dual mechanism, which simultaneously impairs DNA repair and transcriptional adaptation, effectively shutting down multiple survival pathways and leaving resistant cells with nowhere to run.
Third, its remarkable solubility addresses a key limitation of SN-38, enabling excipient-free formulation and potentially mitigating severe side effects like irinotecan-induced diarrhea. As a single agent, it also avoids the pharmacokinetic mismatches often seen in combination therapies.
Beyond hTERT: The Road Ahead
A critical question raised by reviewers was whether ZBH-01’s effect is limited to hTERT. Our data show that it also binds to G-quadruplexes in promoters of other oncogenes such as KRAS and MYC. The extraordinary efficacy in resistant models likely reflects this multi-gene targeting, which simultaneously disrupts complementary survival networks.
Current efforts are now focused on mapping ZBH-01’s full gene regulatory network using ChIP-seq and transcriptomics in resistant models, to elucidate the contribution of each target to its overall therapeutic effect. In parallel, we are designing next-generation analogs based on the solved complex structures, for instance, by stabilizing the linker to prevent hydrolysis to SN-38, to further refine and optimize its dual-targeted profile.
Looking back, this journey from solubility optimization to dual-target discovery reminds us that the most transformative scientific insights often emerge not from a grand initial plan, but from remaining open to unexpected data and pursuing curiosity-driven questions. ZBH-01 provides a compelling proof-of-concept that integrating two powerful anti-cancer mechanisms into a single, optimized molecule offers a promising new pathway to overcoming chemoresistance. We hope this strategy opens new avenues for the future of oncology drug discovery.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
From RNA Detection to Molecular Mechanisms
Publishing Model: Open Access
Deadline: May 05, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in