ASIC1a affects hypothalamic signaling and regulates the daily rhythm of body temperature in mice
Published in Neuroscience
The importance of the circadian rhythm
The circadian rhythm enables organisms to adjust their state to be best adapted to changes in the external environment along the day. The body's various behaviors and physiological functions show obvious circadian rhythms, such as sleep/wake, feeding, blood pressure, coagulation-fibrinolysis balance, heart rate, body temperature, hormone levels, cell metabolism, cell proliferation and immune regulation 1. Circadian rhythms are driven by an internal biological clock located in the brain, specifically in a region called the suprachiasmatic nucleus (SCN) of the hypothalamus. This clock receives input from external cues, primarily light and food intake, which help synchronize the internal rhythm with the external day-night cycle. Disruptions to circadian rhythms, such as those caused by shift work, jet lag, or irregular sleep schedules, can lead to various health issues.
Control of the body temperature and the metabolism
The hypothalamus senses the body temperature and controls the balance of heat production and loss. The latter are achieved by adjusting food intake, metabolism and energy expenditure through hormonal and neuronal mechanisms. Body temperature and metabolism are regulated by thyroid hormones, which increase the basal metabolic rate. They increase the oxygen consumption, respiration rate, and body temperature. The release of thyroid hormones is controlled by signaling from the hypothalamus via the pituitary to the thyroid, involving secretion of thyrotropin-releasing hormone from the hypothalamus and thyroid-stimulating hormone from the pituitary. The body temperature is also regulated by other mechanisms, as e.g., the adrenergic tone of the sympathetic nervous system.
ASIC1a affects daily rhythms
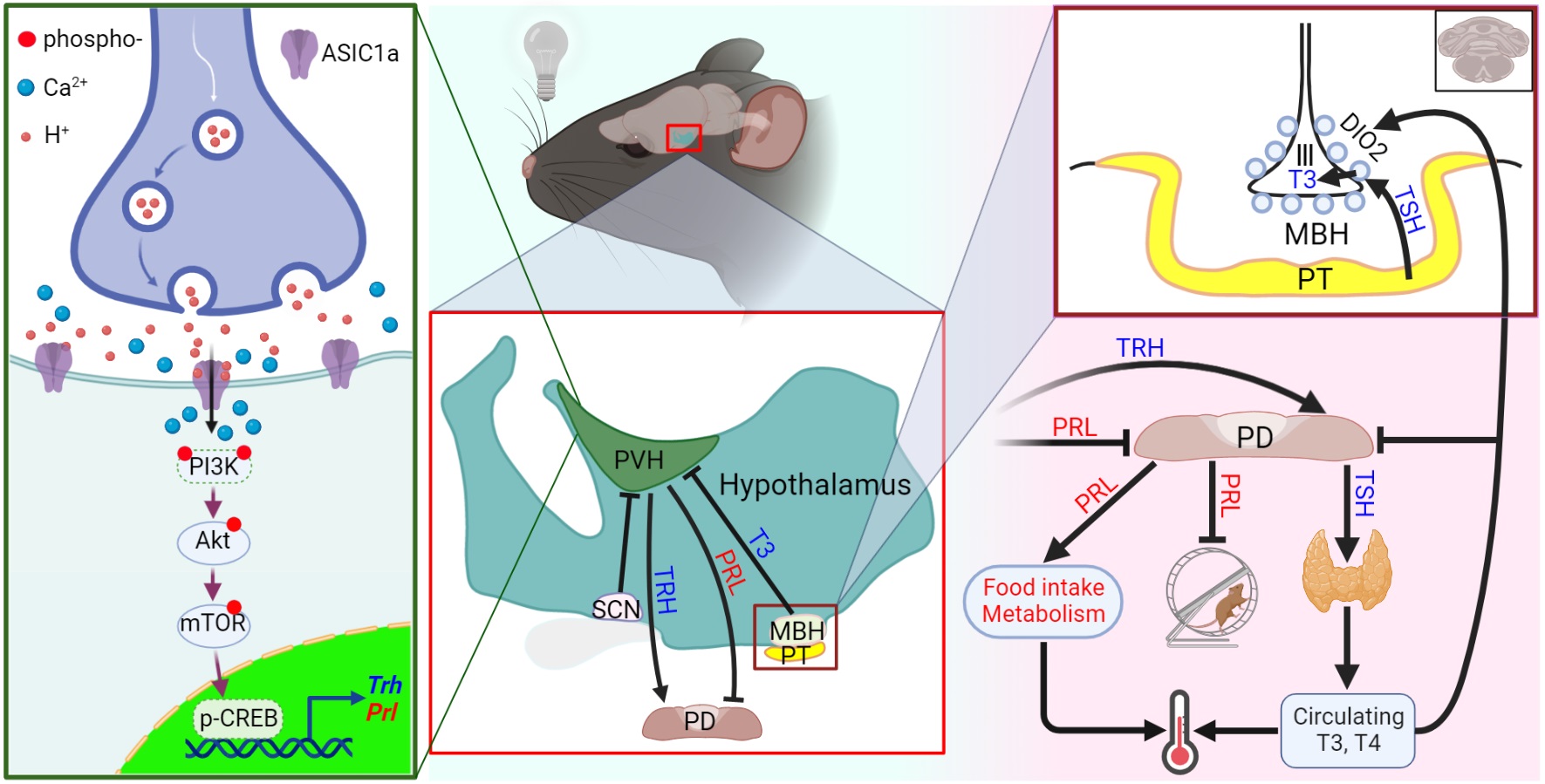
ASIC1a is a proton-gated Na+ channel whose activity affects neuronal signaling2. ASIC1a is expressed in many brain regions and shows abundant expression in the hypothalamus and pituitary. To determine whether ASIC1a influences the body temperature, we implanted temperature sensors in wild type (WT) mice and mice in which ASIC1a was deleted, to monitor their temperature rhythms. The measurements showed that the body temperature of the mice was increased during their active phase (Left panel in the figure; Zeitgeber Time 0 corresponds to the moment when the light is switched on in the morning). We observed a small, statistically significant difference between WT and ASIC1a-/- mice at the two time points ZT17 and ZT18. The spontaneous activity of these mice, measured with the implanted sensor, did not differ between the genotypes.
This prompted us to ask whether basic mechanisms of daily rhythms may be changed in ASIC1a-/- mice. To this end we conducted a wheel-running experiment, in which mice had free access to a running wheel in their cage while this activity was recorded.
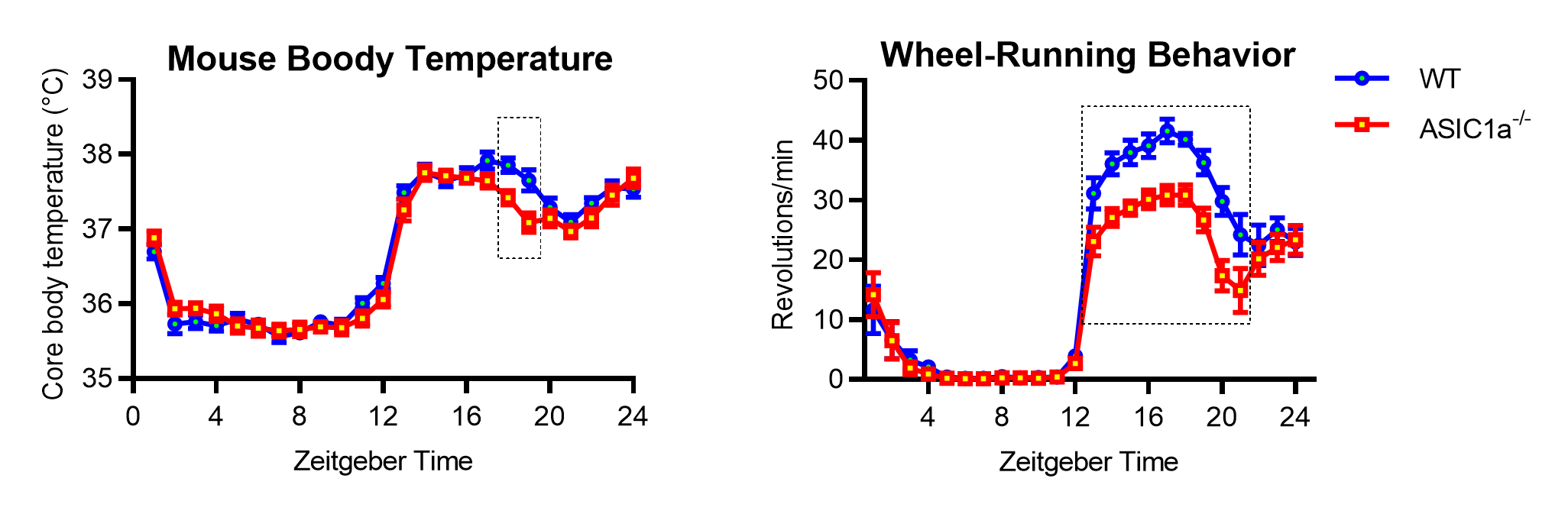
| Figure 1. Body temperature and wheel-running activity of mice. Mice were kept on a 12h light / 12h dark cycle. Left, the body temperature was measured with an implanted sensor and is shown as a function the Zeitgeber time (n=10-11). Right, wheel-running activity in separate experiments, showing the 7-day average activity (n=11). |
The duration of the daily cycle under constant darkness (the "free-running period") was slightly increased in ASIC1a-/- mice. Interestingly, ASIC1a-/- mice showed lower voluntary activity than WT mice throughout the active period (right panel on graph). The mechanisms underlying the daily rhythm regulation of body temperature and wheel-running behavior differ, with body temperature rhythms being jointly regulated by the signals from the central and the peripheral nervous system, while wheel-running behavior is primarily controlled by the signals from the central nervous system.
How does ASIC1a affect daily rhythms and body temperature?
Because we found that ASIC1a affects the daily rhythms of both body temperature and wheel-running behavior, we tested next whether ASIC1a itself follows a daily rhythm of expression and/or activity. This showed that ASIC1a expression in the hypothalamus and SCN follows a daily rhythm and that deletion of ASIC1a affects the daily rhythms of hypothalamic neuron activity.
We asked next how ASIC1a's daily rhythm regulates body temperature and wheel-running behavior. To answer this question, we conducted high-throughput RNA sequencing on the mouse hypothalamus. Deletion of ASIC1a only affected the expression of one functional gene at ZT1 and 13 functional genes at ZT13 in the hypothalamus. Intriguingly, the genes with altered expression were all regulated by the same signaling pathway, making the subsequent research considerably easier.
We validated that ASIC1a's daily rhythmic expression regulates the PI3K-Akt-mTOR signaling pathway to modulate the expression of prolactin and thyrotropin-releasing hormone (TRH). Prolactin can influence both body temperature regulation and mouse wheel-running behavior. On the other hand, TRH leads via the hypothalamic-pituitary-thyroid axis to the release of the thyroid hormones T3 and T4, which regulate body temperature and control hypothalamic activity by providing feedback to the central nervous system. We found that thyroid-stimulating hormone mRNA in the pituitary was also differently regulated in ASIC1a-/- mice, consistent with an involvement of ASIC1a in the signaling along the hypothalamic-pituitary-thyroid axis. However, the concentrations of the thyroid hormones T3 and T4 in the serum were not affected by ASIC1a deletion, indicating that despite the changed signaling in the hypothalamus and the pituitary, ASIC1a regulates the body temperature not via the thyroid hormones.
Concluding remarks
In conclusion, the present study demonstrates that the expression of ASIC1a in the hypothalamus follows a daily rhythm and that deletion of ASIC1a partially prevents the normal increase in body temperature during the active phase. The lower body temperature in ASIC1a-/- mice is likely due to altered signaling in the hypothalamus that leads to changes in metabolism and/or food intake by mechanism that warrant further characterization. Our study not only expanded the understanding of ASIC1a's physiological functions but has also provided deeper insights into temperature regulation. Additionally, it has introduced ASIC1a as a new player in temperature and metabolic control.
References
1. Potter, G. D. et al. Circadian Rhythm and Sleep Disruption: Causes, Metabolic Consequences, and Countermeasures. Endocr Rev 37, 584-608, doi:10.1210/er.2016-1083 (2016).
2. Kellenberger, S. & Schild, L. International Union of Basic and Clinical Pharmacology. XCI. Structure, Function, and Pharmacology of Acid-Sensing Ion Channels and the Epithelial Na+ Channel. Pharmacol Rev 67, 1-35, doi:10.1124/pr.114.009225 (2015).
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
From RNA Detection to Molecular Mechanisms
Publishing Model: Open Access
Deadline: May 05, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in