Behind the paper: Single atoms of indium enable superior green methanol synthesis
Published in Chemistry

The big picture
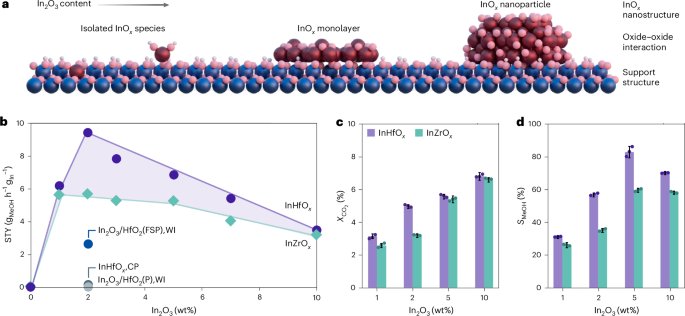
Thermocatalytic CO2 hydrogenation to methanol, a high-pressure gas-phase reaction that transforms greenhouse gases into useful platform chemicals, plays a crucial role in decarbonizing the hard-to-abate chemical sector. Although industrial copper-based catalysts in syngas-to-methanol production demonstrate high activity, they typically exhibit low methanol selectivity and suffer from severe water-induced deactivation under pure CO2 feed. Consequently, the interest in CO2 valorisation has propelled the quest for superior catalytic systems with high methanol selectivity and stability under CO2 hydrogenation conditions. In this context, indium oxide (In2O3)-based systems, in particular mixed indium-zirconium oxide (InZrOx), have emerged as the leading candidate since its discovery in 2015.[1] This is because the strong oxide-support interaction stabilizes the active In2O3 phase, driving selective and stable methanol production.[2,3] Crucially, ZrO2 enhanced methanol productivity of the active indium phase by more than an order of magnitude with augmented interfacial oxygen vacancies, which have been suggested to be the active sites for this reaction.

A decade-long challenge in the field
Significant efforts worldwide have been dedicated to deepening the understanding of this exceptional oxide-oxide interfacial behaviour over the last decade, with the ultimate goal of improving the catalytic performance. Although different strategies such as catalyst design and reaction engineering have been reported to improve the performance of InZrOx catalysts;[4,5] fundamentally, no other oxides have showcased such a unique support promotional effect. Furthermore, various In2O3 architectures, such as monolayers and clusters, have been proposed to be the optimal active nanostructures. Indium single atoms have generally been considered less active than extended In2O3 nanostructures for methanol synthesis, precluding better utilization of the expensive element. Overall, two questions persist: is there an alternative support comparable or even superior to ZrO2? And how can we maximize the atom efficiency by optimizing the catalytic performance of indium single atoms?
Breaking the long-standing limitation by coupling single atom catalysis and wide bandgap dielectrics
Writing in Nature Nanotechnology, we present the discovery of a new catalytic system that genuinely addresses these two long-standing challenges.[6] We show that single atoms of indium on hafnia overcomes these hurdles by coupling the concepts of wideband gap dielectrics and single-atom catalysis. Hafnium oxide, or hafnia (HfO2), is a metal oxide rarely explored in heterogeneous catalysis owing to its presumable chemical and electronic inertness. Notably, it has been widely applied in scalable electronics and ferroelectrics due to its wide bandgap, simultaneously minimizing current leakage and maximizing charge transport efficiency. We transferred this concept and revealed that a dielectric traditionally considered physiochemically inert can actively participate in interfacial catalysis, enabling efficient reactant activation and facilitating intermediates hydrogenation around the isolated indium sites. Using flame spray pyrolysis, we precisely controlled the indium dispersion on HfO2, stabilizing isolated single-atom sites, and demonstrating that they are the most efficient hydrogen splitters.
Outlook and scientific impact
Extending this concept beyond In2O3, we uncovered that HfO2 support also enhances the CO2 hydrogenation performance of other families of reducible oxides, such as zinc- and gallium single atoms, by more than two orders of magnitude. Overall, these results show the broad application of HfO2, justifying its role as an efficient and robust support for advancing sustainable chemical transformation and single-atom catalysis. For more details, please refer to the article[6].
References
[1] Martin, O. et al. Indium oxide as a superior catalyst for methanol synthesis by CO2 hydrogenation. Angew. Chem. Int. Ed. 55, 6261-6265 (2016).
[2] Frei, M. S. et al. Role of zirconia in indium oxide-catalyzed CO2 hydrogenation to methanol. ACS Catal. 10, 1133-1145 (2019).
[3] Yang, C. et al. Strong electronic oxide–support interaction over In2O3/ZrO2 for highly selective CO2 hydrogenation to methanol. J. Am. Chem. Soc. 142, 19523-19531 (2020).
[4] Pinheiro Araújo, T. et al. Flame-made ternary Pd-In2O3-ZrO2 catalyst with enhanced oxygen vacancy generation for CO₂ hydrogenation to methanol. Nat. Commun. 13, 5610 (2022).
[5] Gao, F. et al. Redox-mediated interfacial restructuring of supported In2O3 to drive CO2 hydrogenation to methanol. ACS Catal. 15, 2785–2795 (2025).
[6] Chiang, Y.-T. et al. Single atoms of indium on hafnia enable superior CO2-based methanol synthesis. Nat. Nanotechnol. doi:10.1038/s41565-026-02135-y (2026).
Follow the Topic
-
Nature Nanotechnology
An interdisciplinary journal that publishes papers of the highest quality and significance in all areas of nanoscience and nanotechnology.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in