Beyond Hospitals: What Wastewater Can Tell Us About Antimicrobial Resistance
Published in Healthcare & Nursing, Earth & Environment, and Ecology & Evolution
As a microbiology PhD researcher studying antibiotic resistance genes in wastewater treatment systems, I read the recent Nature Communications article, “Wastewater surveillance reveals patterns of antibiotic resistance across the United States,” with particular interest. The study addresses a question that is central to the future of antimicrobial resistance surveillance: can wastewater help us detect resistance patterns before they become fully visible through clinical surveillance?
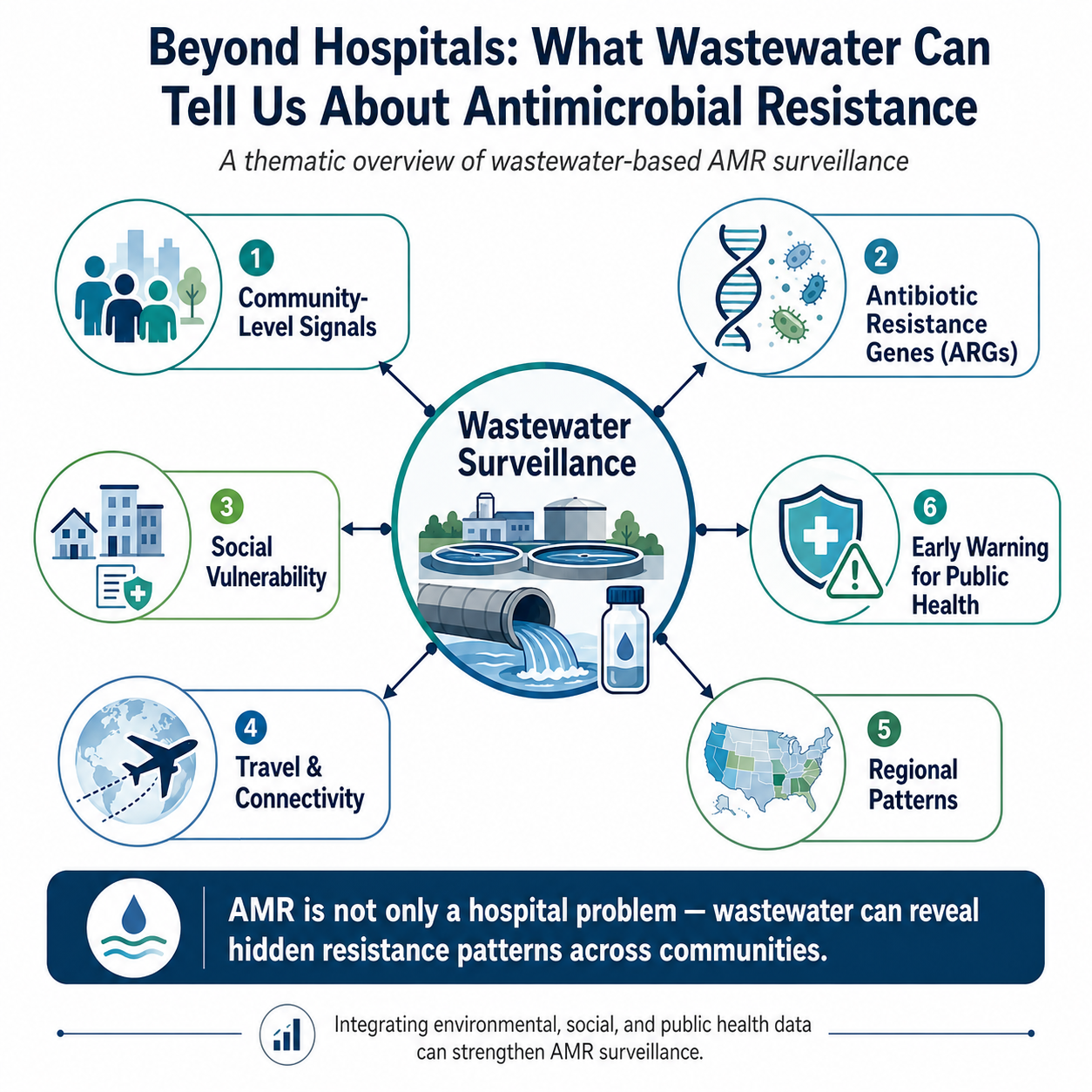
Antimicrobial resistance is often described as a clinical crisis, but resistant bacteria and resistance genes do not remain confined to hospitals, clinics, or laboratories. They move through communities, households, wastewater systems, animals, food systems, travel networks, and the environment. This makes AMR not only a medical challenge, but also a community, environmental, social equity, and One Health challenge.
The study by Kim and colleagues provides an important national-scale example of how wastewater-based surveillance can reveal hidden patterns of antibiotic resistance. The researchers measured 11 clinically relevant antibiotic resistance genes across 163 wastewater treatment plants in 40 U.S. states, creating a broad baseline of ARG concentrations in wastewater . This scale is important because it moves wastewater AMR surveillance beyond isolated local studies and toward a population-level public health tool.
Traditional AMR surveillance depends heavily on clinical samples from patients who seek healthcare. While this approach is essential, it does not fully capture the broader community burden of resistance. Many individuals carrying resistant bacteria may never visit a clinic, may not be tested, or may not have equal access to healthcare. Wastewater, by contrast, captures biological signals from entire communities. It can provide a wider lens into what is circulating at the population level. One of the strengths of this study is its use of droplet digital PCR to quantify resistance genes associated with beta-lactams, colistin, tetracycline, and vancomycin. Several genes, including blaCMY, blaCTX-M, blaKPC, blaOXA-48, blaTEM, and tetW, were consistently detected across wastewater samples . These are not abstract molecular signals. They represent genetic markers linked to resistance against clinically important antibiotics.
The study also found regional differences in resistance burden. The South had a higher overall antibiotic resistance gene burden compared with the Midwest . Such regional variation matters because it suggests that wastewater surveillance may help identify geographic patterns of resistance that could guide public health priorities, resource allocation, and future research.
However, the most thought-provoking finding is that antibiotic resistance patterns were not explained by antibiotic use alone. Antibiotic prescription rates showed only weak associations with wastewater ARG concentrations. Instead, several social vulnerability indicators, including housing burden, lack of health insurance, overcrowding, and limited English proficiency, were associated with higher concentrations of some resistance genes .
This finding deserves careful attention. Antibiotic stewardship remains essential, but this study reminds us that stewardship alone may not be enough. AMR is shaped not only by prescribing practices, but also by healthcare access, housing conditions, sanitation, mobility, infrastructure, social vulnerability, and environmental exposure. At the same time, these findings must be interpreted responsibly. The study reports associations, not causation . Social vulnerability indicators should not be used to stigmatize communities. Instead, they should prompt deeper investigation into structural conditions that may increase exposure to resistant organisms or limit access to timely diagnosis and treatment. AMR is not simply a problem of individual behavior; it is also a reflection of the systems in which people live.
Another important dimension is global mobility. The study identified associations between certain resistance genes and indicators related to international travel, including airport presence and limited English proficiency . This does not mean travel or immigrant communities should be blamed. Rather, it highlights how interconnected antimicrobial resistance has become. Resistant genes can move across borders through travel, healthcare systems, food systems, animals, and the environment. AMR surveillance therefore needs to be global, inclusive, and scientifically cautious.
For researchers working in wastewater-based epidemiology, this study offers a useful framework. It shows how wastewater data can be integrated with demographic, socioeconomic, healthcare, agricultural, and geospatial datasets. It also shows why gene-specific analysis matters. Different resistance genes may follow different patterns, and broad summary scores may miss important local or gene-specific signals .This is especially relevant for low- and middle-income countries, including Nepal, where clinical AMR surveillance may be limited by cost, infrastructure, laboratory capacity, and healthcare access. In such settings, wastewater surveillance could become a powerful complementary tool. It may help detect community-level resistance trends even when clinical data are incomplete or delayed.
For example, monitoring influent, effluent, and receiving river water from wastewater treatment plants could help identify how treatment processes influence the persistence and release of ARGs into the environment. Combining shotgun metagenomics, targeted PCR methods, and environmental metadata could provide a more complete picture of how resistance moves between humans, wastewater systems, and natural water bodies.
This study also raises a larger question for the field: How do we turn wastewater AMR data into action?
Generating surveillance data is only the first step. The next challenge is translating those data into public health decisions. Wastewater AMR surveillance could support early warning systems, identify emerging resistance hotspots, evaluate the impact of interventions, and inform policy. But this will require standardized methods, ethical data interpretation, interdisciplinary collaboration, and communication between scientists, wastewater utilities, clinicians, epidemiologists, policymakers, and communities.
The future of AMR surveillance should not be limited to hospitals. It should connect clinical microbiology, wastewater monitoring, environmental sampling, antibiotic use data, social vulnerability indicators, and One Health frameworks. This integrated approach can help public health systems move from a reactive model to a proactive one.
Wastewater does not replace clinical surveillance, but it can strengthen it. It can reveal resistance signals that may otherwise remain invisible. It can help us understand AMR as a community-level and environmental phenomenon. Most importantly, it reminds us that antimicrobial resistance is not only about microbes and medicines. It is also about people, infrastructure, inequality, mobility, and the environments we share.
As AMR continues to threaten global health, wastewater surveillance offers a powerful lens into the hidden movement of resistance across communities. The next frontier is not only detecting resistance genes, but building integrated surveillance systems that connect wastewater data with clinical evidence, social context, environmental monitoring, and policy action.
For me, this study reinforces a simple but important message: to understand antimicrobial resistance, we need to look beyond hospitals. Sometimes, the clearest signal of a community’s health is found in its wastewater.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in