Bivalent HPV vaccine performance against cross-protected HPV genotypes: Implications of differential vaccine efficacy across viral variants
Published in Cancer, Biomedical Research, and Immunology
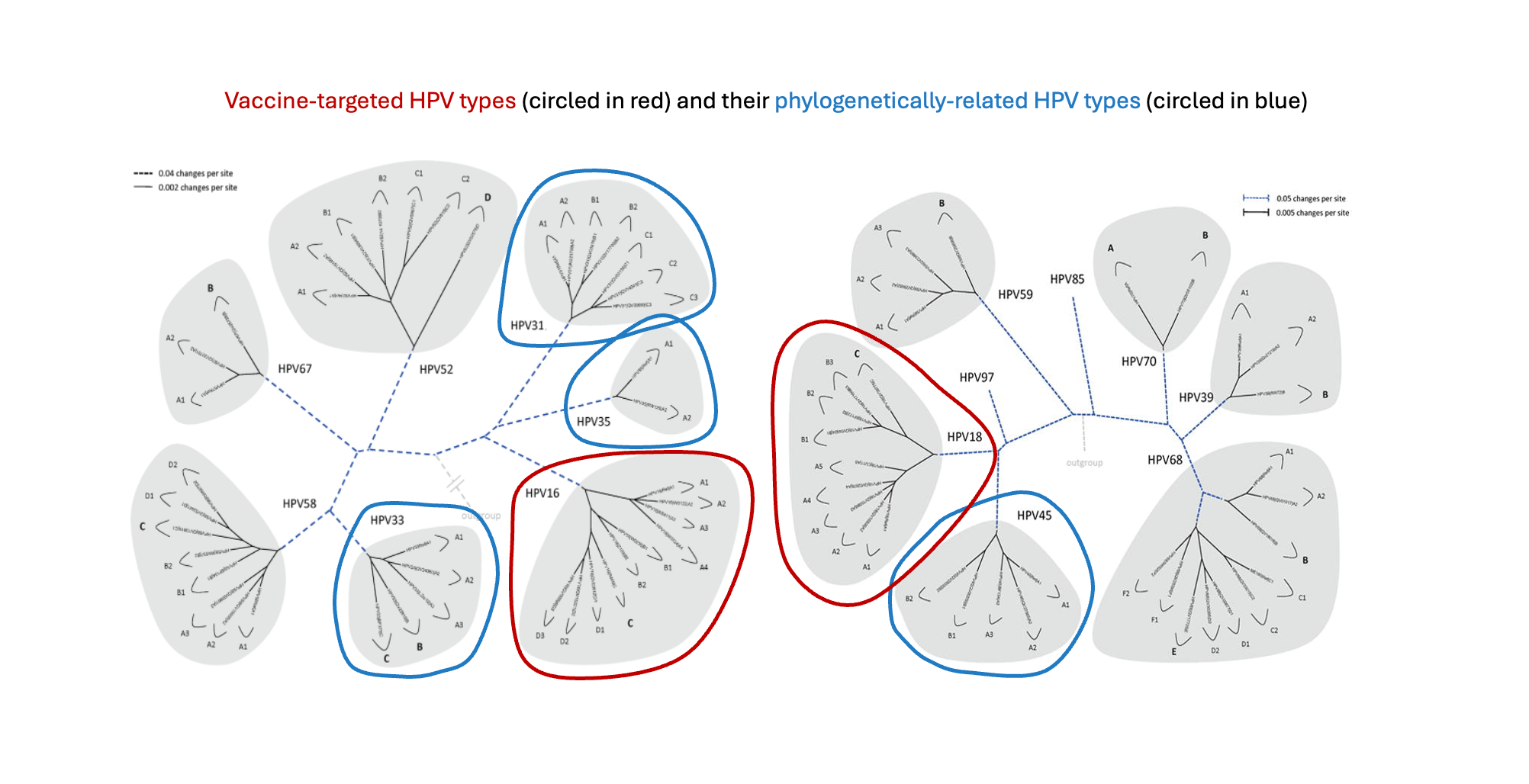
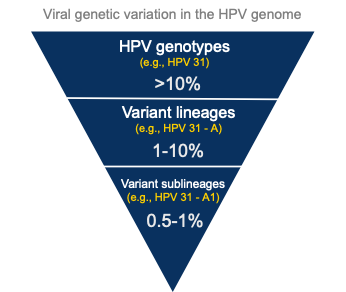
In our recent npj Vaccines publication, we report significantly different long-term protection following vaccination with a bivalent human papillomavirus (HPV) vaccine against variants of some HPV genotypes that are closely related to the vaccine-targeted HPV genotypes. Within a genotype, there is considerable viral genetic variation, which are divided into lineages, sublineages, and single nucleotide polymorphisms (SNPs), referred to as “variants” (Figure 1).
Results from our study could have large global health implications. HPV is the most common sexually transmitted infection in the world. Persistent HPV infection with a carcinogenic HPV type is the necessary cause of cervical precancer and cancer. HPV burden varies widely by world region. In recent years, researchers discovered that different HPV types and their variants are more prevalent in specific populations. Some are these variants are more likely to lead to cancer than others. For example, HPV type 35 (HPV35) sublineage A2 infections are much more frequent among women with African ancestry and are associated with an increased risk of developing high-grade cervical precancerous lesions.
Of note, HPV35 (regardless of variant) is not covered by the currently available prophylactic HPV vaccines. However, in the Costa Rica HPV Vaccine Trial, we observed that the bivalent HPV vaccine may provide some protection of specific HPV35 variants over 11 years of follow-up. We identified one SNP in the L1 gene region (position 5939) for HPV35 that provided significant vaccine protection against one of the nucleotides; L1 codes the capsid formation and is the basis for the HPV sub-unit vaccines. The bivalent HPV vaccine was 65% efficacious against HPV35 infections with nucleotide G at position 5939. However, for the more common nucleotide A at position 5939, there was no evidence of vaccine protection. If this observation is real and not due to chance, it may have substantial impacts on the vaccine’s cancer prevention potential in populations with high HPV burden. For example, prior studies show that nucleotide A at position 5939 is present in all women with an HPV35 sublineage A2 infection. As mentioned above, this sublineage is much more prevalent and poses greater cancer risk for women with African ancestry.
We also observed differences in HPV vaccine efficacy across lineages of HPV31. Over 11 years of follow-up, women who received the bivalent HPV vaccine had a 94% lower risk of incident HPV31 lineage A infections compared with unvaccinated women. For incident HPV31 lineage B infections, the vaccine was only 61% efficacious. Both HPV31 lineages A and B are associated with elevated risks of cervical precancer and cancer. The much lower protection against HPV31 lineage B could have clinical implications.
At the SNP-level, we identified one nonsynonymous substitution on the FG loop (position 6372) with significantly different vaccine protection by nucleotide. The FG loop is an essential domain for neutralization. We found that nucleotide C at position 6372 (associated with HPV31 lineage A) had a much higher vaccine efficacy (94%) compared with nucleotide A at position 6372 (associated with HPV31 lineage B or C) (75%). This suggests that even the relatively small change at this position could result in differences in epitope recognition, which may impact the ability of the HPV vaccine to protect against certain infections.
The signals we observed in our study warrant further investigation and validation in other study populations. For instance, in the predominately White Costa Rican population, some HPV variants were rare, such as HPV35 sublineage A2, which limited our ability to examine vaccine efficacy differences robustly across HPV35 sublineages. Thus, it is imperative to replicate our analysis in other populations with greater prevalence of these variants to better ascertain the vaccine’s protection against them.
Poster image adapted from two sources:
- Chen, Z. et al. Evolution and Taxonomic Classification of Human Papillomavirus 16 (HPV16)-Related Variant Genomes: HPV31, HPV33, HPV35, HPV52, HPV58 and HPV67. PLOS ONE 6, e20183 (2011).
- Burk, R. D., Harari, A. & Chen, Z. Human papillomavirus genome variants. Virology 445, 232–243 (2013).
Follow the Topic
-
npj Vaccines
A multidisciplinary journal that is dedicated to publishing the finest and high-quality research and development on human and veterinary vaccines.
Related Collections
With Collections, you can get published faster and increase your visibility.
Therapeutic HPV vaccines
Publishing Model: Open Access
Deadline: Jun 30, 2026
Lipid nanoparticle (LNP)-adjuvanted vaccines
Publishing Model: Open Access
Deadline: May 19, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in