Breaking the Unbreakable: How Magnetic Spinel Ferrites are Destroying PFAS and Microplastics
Published in Chemistry and Earth & Environment
Explore the Research
 sciencedirect.com
sciencedirect.com
Just a moment...
Please confirm you are a human by completing the captcha challenge below.
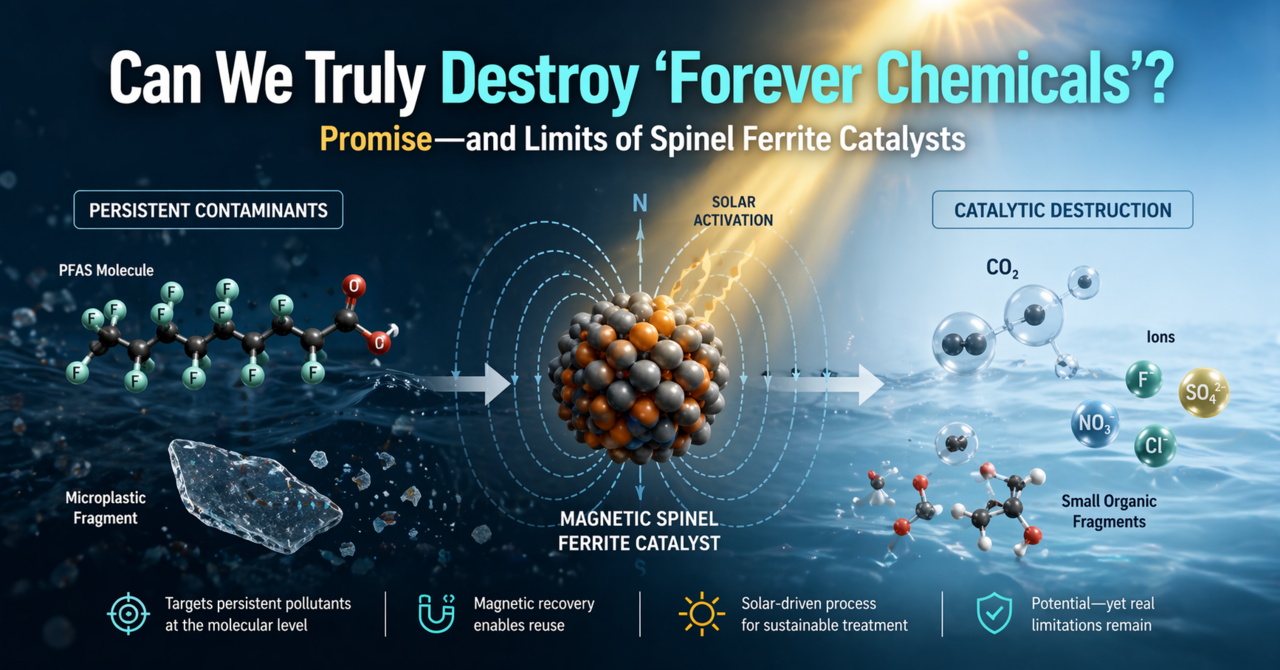
The Age of Indestructible Contaminants
We have engineered molecules to resist degradation—and they perform exactly as designed. Per- and polyfluoroalkyl substances (PFAS) are stabilized by some of the strongest bonds in organic chemistry, while microplastics consist of robust polymeric networks designed for durability. These materials bypass conventional treatment systems and persist across environmental compartments.
Physical removal strategies often shift the burden rather than eliminate it. Concentrating contaminants into sludge or secondary waste streams does not constitute remediation—it delays it. True solutions require chemical transformation at the molecular level via Advanced Oxidation Processes (AOPs).
The Magnetic Catalyst: Why Spinel Ferrites?
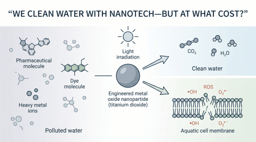
Traditional photocatalysts, such as titanium dioxide (TiO2), have demonstrated strong oxidative capabilities, but their real-world deployment remains constrained by two major limitations: their dependence on UV irradiation and the severe difficulty of post-treatment recovery.
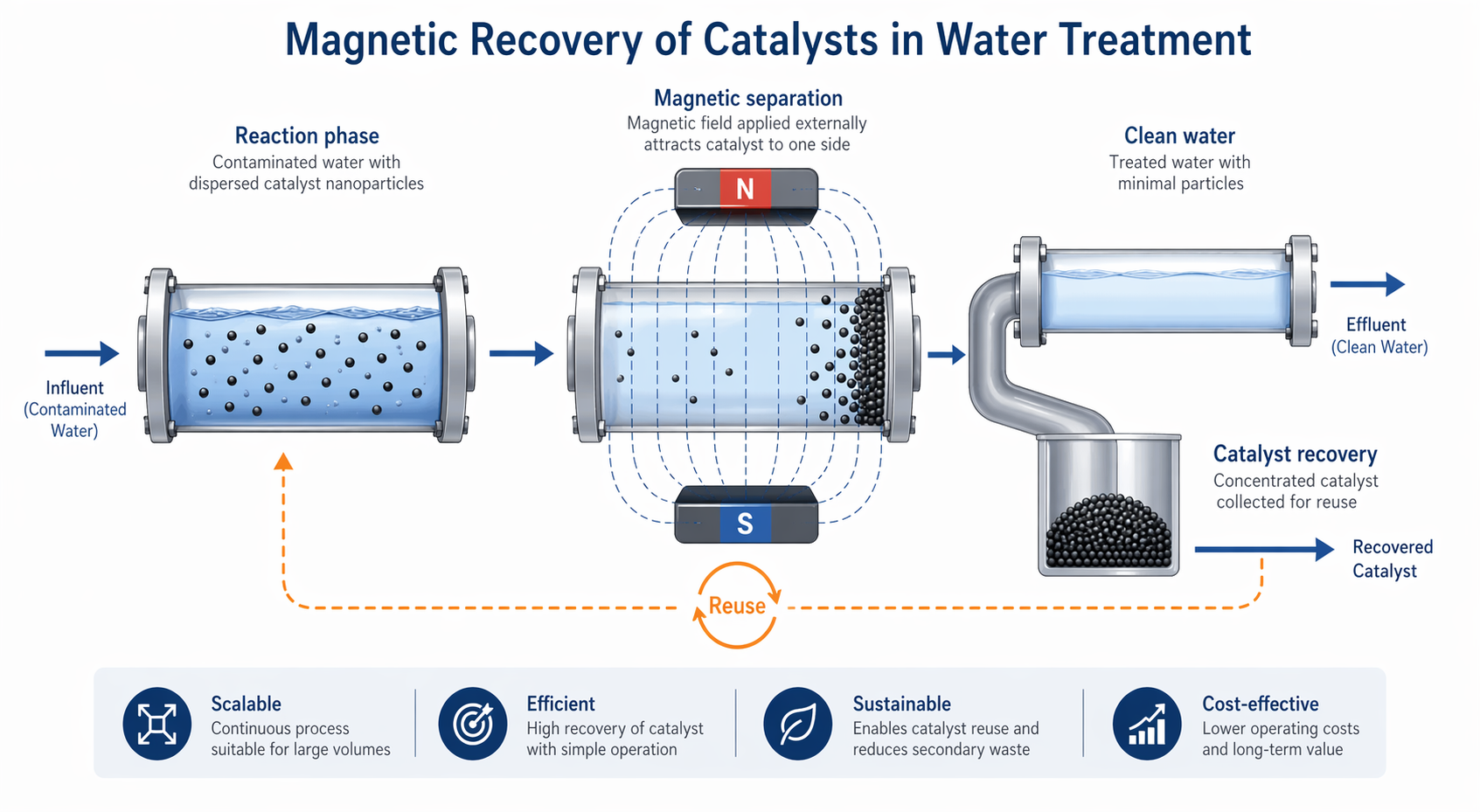
Spinel ferrites offer a fundamentally different design space. At the nanoscale, they exhibit intrinsic superparamagnetic properties, enabling rapid separation via external magnetic fields without complex filtration systems. More importantly, their tunable crystal structure—featuring mixed-valence cations and defect-rich lattices—enables visible-light activation, expanding their utility under solar irradiation. This combination of magnetic recoverability and solar activity makes them uniquely suited for scalable applications.
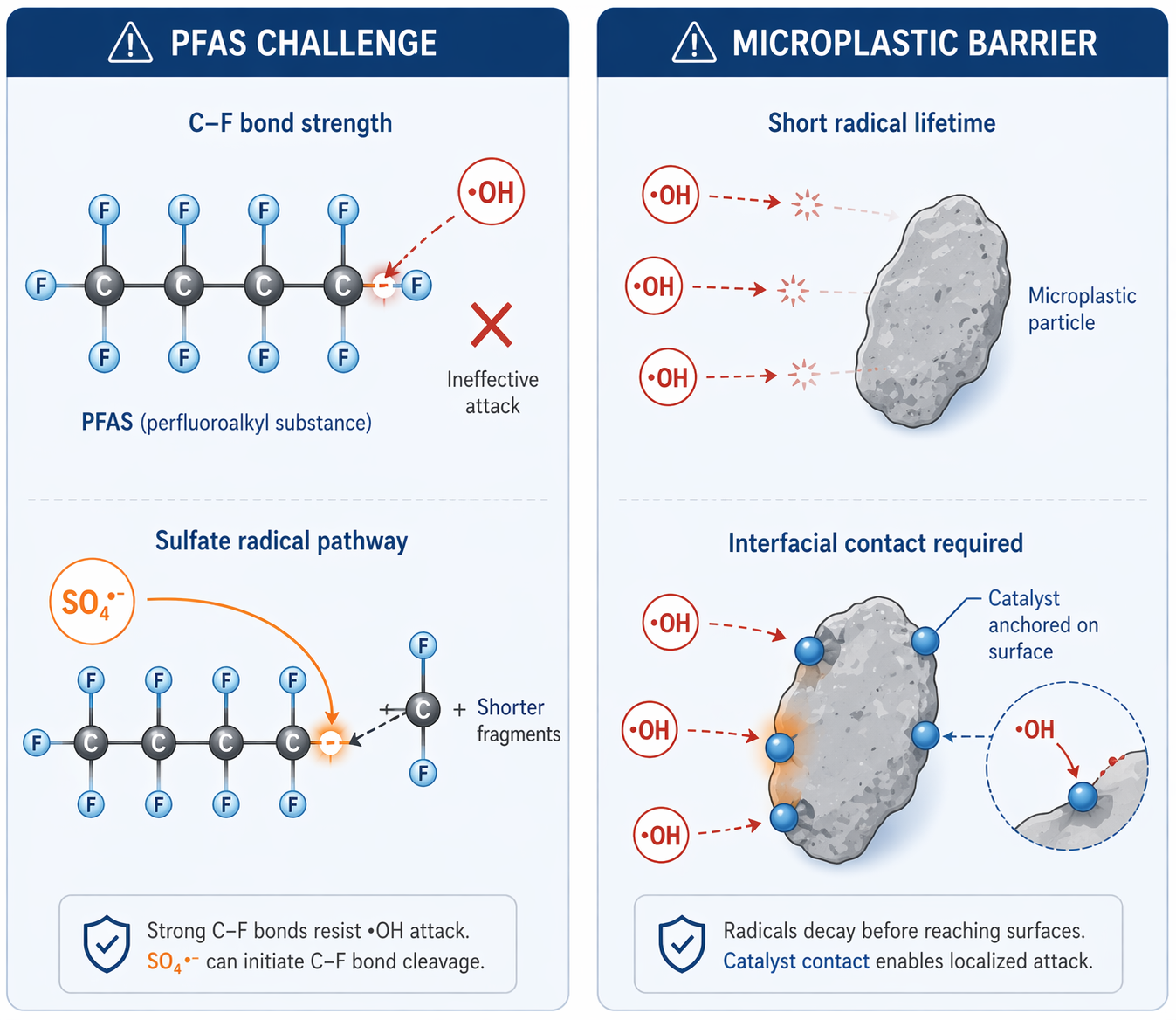
The Thermodynamic Hurdle: Destroying PFAS
Generating reactive oxygen species is necessary—but not sufficient. The extreme stability of PFAS arises from the strength of the C-F bond (∼485 kJ/mol), which limits the effectiveness of conventional oxidative pathways. Hydroxyl radicals (OH•), while highly reactive, often exhibit limited efficiency toward perfluorinated chains.
To overcome this barrier, alternative pathways are required. In my latest critical review, I explore how spinel ferrites can be engineered to:
-
Activate peroxymonosulfate (PMS) to generate highly potent sulfate radicals (SO4•-).
-
Facilitate localized electron transfer processes that promote reductive defluorination.
These pathways enable stepwise degradation mechanisms, including sequential decarboxylation and the progressive shortening of the perfluoroalkyl chain.
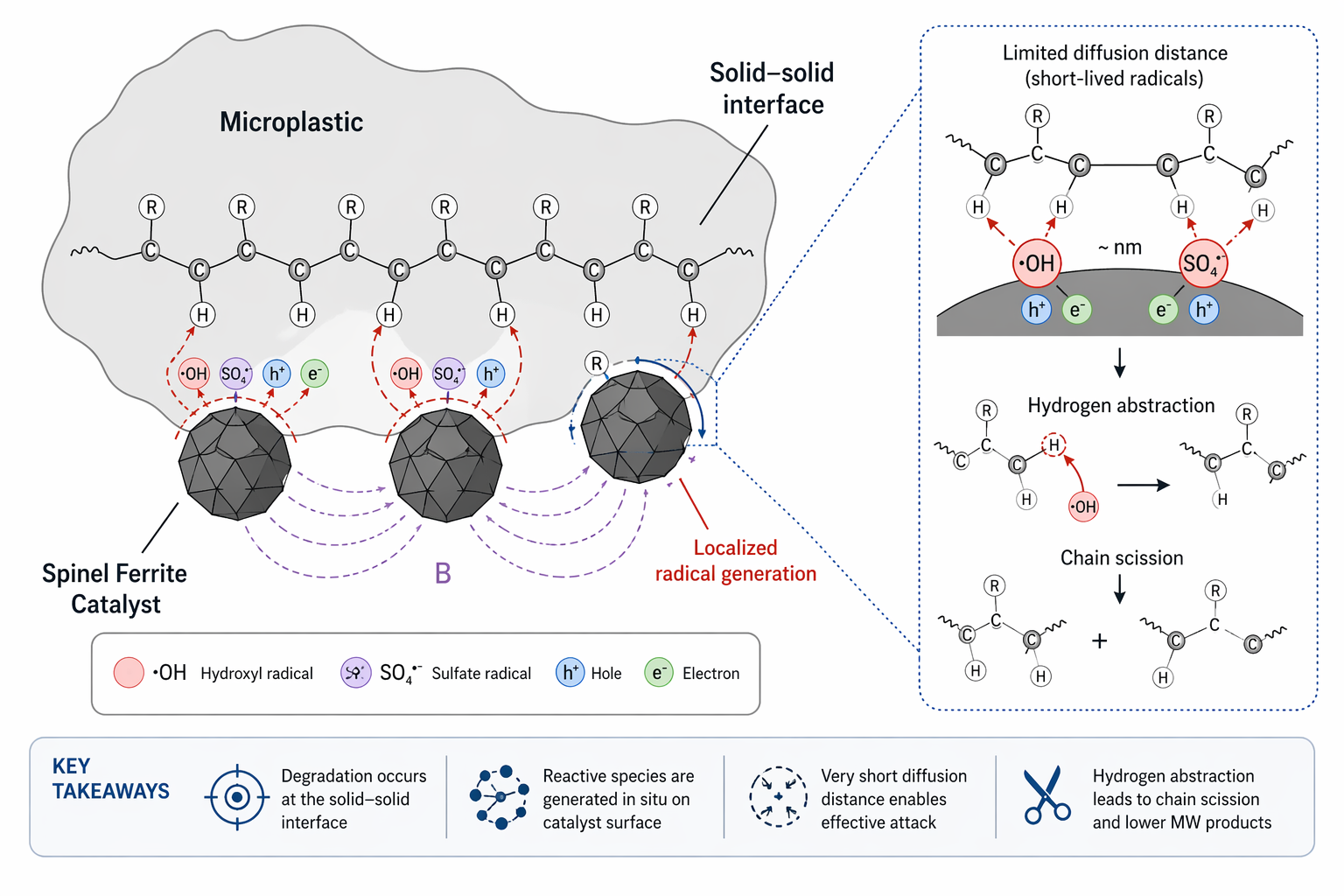
The Solid-Solid Barrier: Attacking Microplastics
Microplastics introduce a fundamentally different constraint: phase separation. Unlike dissolved contaminants, microplastics exist as massive solid particles. Reactive radicals have extremely short lifetimes (nanosecond to microsecond range) and limited diffusion distances. As a result, they often decay in the bulk solution before reaching the polymer surface, creating a severe mass-transfer bottleneck.
To address this, catalyst design must enforce direct interfacial contact. Surface-engineered spinel ferrites—for example, hydrophobically modified CoFe2O4—can spontaneously hetero-aggregate with polymer surfaces. This anchoring localizes reactive species generation directly at the solid-solid interface, resulting in:
-
Enhanced hydrogen abstraction on the polymer backbone.
-
The immediate initiation of chain scission.
-
Progressive oxidation and fragmentation into leachable monomers.
The Achilles' Heel: Metal Leaching and Stability
Despite their immense promise, spinel ferrites are not without limitations. Under the highly oxidative and acidic conditions typical of AOP systems, metal leaching (Fe, Co, Ni) can occur. This leads to catastrophic catalyst deactivation, loss of structural integrity, and potential secondary heavy metal contamination. Addressing this requires robust structural design (e.g., carbon encapsulation) and the stabilization of active sites. Long-term stability—not just activity—will determine real-world viability.
Conclusion
The challenge of destroying "forever" contaminants is not merely chemical—it is interfacial and thermodynamic. Spinel ferrites demonstrate that intelligent materials design can bridge this gap, linking defect chemistry, charge transport, and reactive species generation.
But the future of water treatment will not be defined by how many radicals we generate—it will be defined by how effectively we deliver them to the contaminant itself.
For a detailed analysis of structure-property relationships, specific ROS pathways, and the techno-economic considerations of scale-up, read my full study published in the Journal of Environmental Chemical Engineering here.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in