Cation‑Disordered Rock‑Salt Lithium Titanium Oxyfluoride Anode Enabling High‑Rate Li‑Ion Storage Through a 3D Percolation Network
As the demand for fast-charging energy storage systems continues to grow, conventional lithium-ion batteries face limitations in power density and charging speed. Now, researchers from Shandong University, led by Professor Xiaohang Lin, Professor Yuanwei Sun, Professor Longwei Yin, and Professor Rutao Wang, have presented a breakthrough pseudocapacitive anode material that bridges the gap between batteries and supercapacitors.
Why This Anode Matters
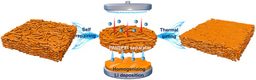
Traditional pseudocapacitive materials typically suffer from high operating potentials (>1.0V vs. Li+/Li), which reduces overall cell voltage and energy density. The novel cation-disordered rock-salt lithium titanium oxyfluoride (DRX-LixTiOF2) overcomes this limitation by enabling pseudocapacitive Li+ storage at remarkably low potentials (down to 0.1V), combining battery-level capacity with supercapacitor-like rate performance.
Innovative Design and Mechanism
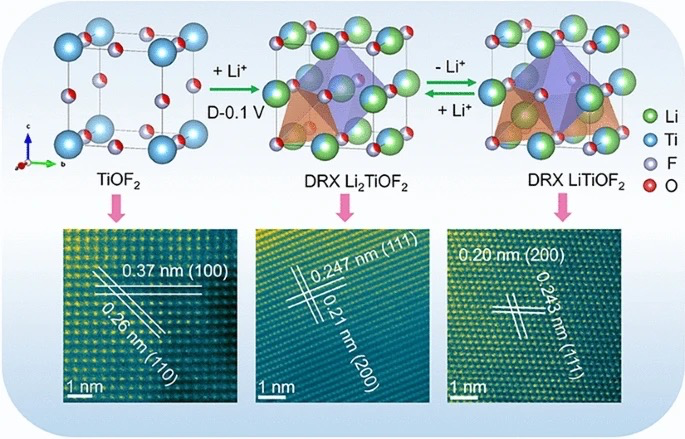
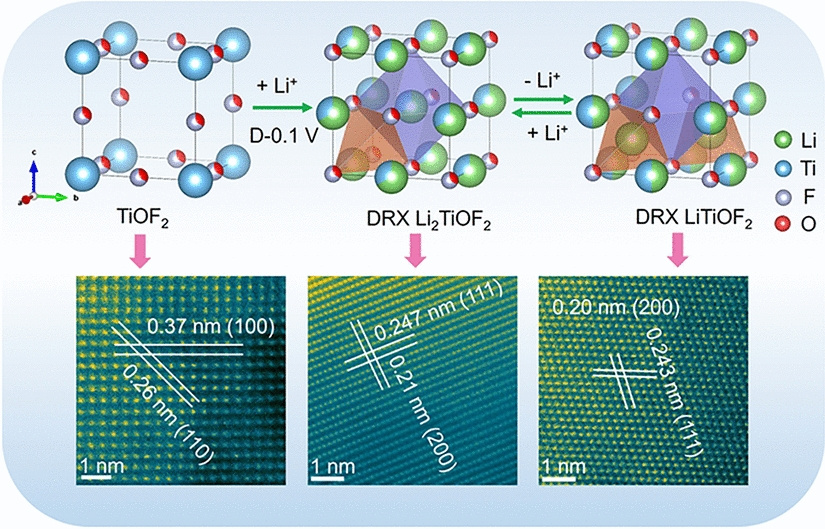
The material is synthesized through an electrochemically induced transformation from cubic TiOF2 to a disordered rock-salt structure. Monte Carlo simulations reveal that its exceptional performance originates from a unique three-dimensional percolation network of 0-TM (zero transition metal) channels—tetrahedral sites lacking transition metals that facilitate fast Li+ migration with low energy barriers. The cation/anion-disordered structure, arising from mixed Li/Ti and O/F occupancy, creates interconnected diffusion pathways enabling "percolating pseudocapacitance" throughout the bulk material.
Outstanding Performance
DRX-LixTiOF2 delivers a high reversible capacity of ~310 mAh g-1 and maintains 93 mAh g-1 at an ultrahigh rate of 20 A g-1 (64.4C). The material exhibits characteristic pseudocapacitive signatures: quasi-rectangular CV curves, sloping charge/discharge profiles, and 88.8% capacitive contribution at 1 mV s-1. Notably, it achieves 88.6% capacity retention over 1,000 cycles.
Applications and Future Outlook
When paired with an activated carbon cathode in a lithium-ion capacitor, the device achieves exceptional metrics: 4.0V operating voltage, 197.9 Wh kg-1 energy density, and an ultrahigh power density of 50,000 W kg-1 with 73.7% retention after 20,000 cycles. This work establishes a new family of intercalation pseudocapacitive materials, opening promising avenues for next-generation energy storage systems combining high safety, fast charging, and high energy density.
Stay tuned for more groundbreaking research from this collaborative team at Shandong University and Shandong First Medical University!
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in