Chronic kidney disease, bone and mineral affection and the search for the perfect biomarker
Published in Biomedical Research

CKD is a growing global factor negatively impacting morbidity and mortality, affecting more than 10% of the general population, which is equivalent to more than 800 million people (Bikbov et al 2021, Kovesdy 2022). Mineral and bone disorder (MBD) is a common consequence of advanced CKD, resulting in major cardiovascular and non-cardiovascular complications, such as myocardial infarction, congestive heart failure, bone fractures, peripheral artery disease or stroke, limiting both the quality of life and longevity of patients with CKD. Current treatment strategies for CKD-MBD focus on optimization of vitamin D supplementation, managing intact parathormone (iPTH) levels, and addressing hyperphosphatemia and hypocalcemia (KDIGO guideline on the treatment of CKD-MBD 2017). However, more knowledge is needed when it comes to early detection of those patients at risk for complications from CKD-MBD.
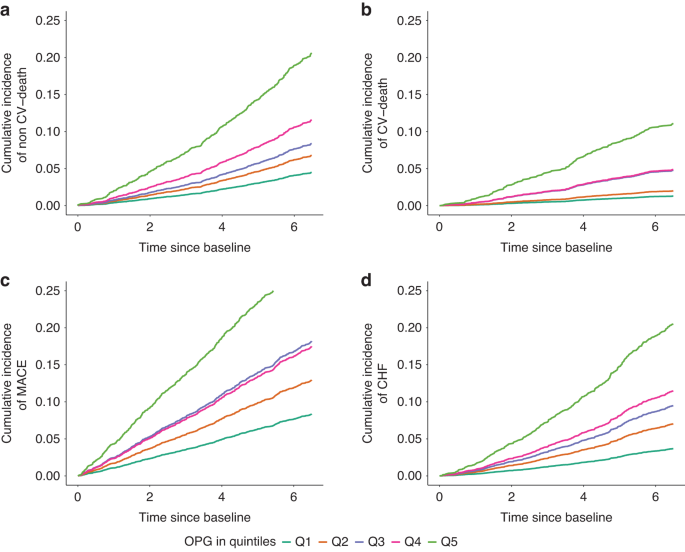
Osteoprotegerin correlates strongest with all four investigated outcomes
In our study (Reimer et al. 2023), we compared the prognostic value of nine MBD biomarkers to determine those associated best with adverse cardiovascular outcomes and mortality. Of 5217 patients enrolled in the German CKD (GCKD) study, serum osteoprotegerin , C-terminal fibroblast growth factor-23 (FGF23), intact parathyroid hormone (iPTH), bone alkaline phosphatase (BAP), cross-linked C-telopeptide of type 1 collagen (CTX1), procollagen 1 intact N-terminal propeptide (P1NP), phosphate, calcium, and 25-OH vitamin D 4246 data sets were complete at baseline and included in the further analysis. During a median follow-up of 6.5 years, non-cardiovascular deaths, cardiovascular deaths, nonfatal major adverse cardiovascular events (MACE) and hospitalizations for congestive heart failure (CHF) were observed. We correlated all serum biomarker levels at baseline to the occurrence of these events and found osteoprotegerin to be associated strongest with all four outcomes.
Osteoprotegerin is involved in regulating bone metabolism - we need to know more
Seeing these results, osteoprotegerin naturally became the focus of our attention. What is the role of osteoprotegerin in the human body and in the context of CKD-MBD in specific? To discover this, it is essential to dive into the basics of bone metabolism, which is a tightly regulated process characterized by constant bone formation and resorption. Osteoprotegerin is an important mediator and regulator in bone metabolism. It is also referred to as the osteoclastogenesis inhibitory factor (OCIF) and functions as a cytokine receptor of the tumor necrosis factor (TNF) family. As a soluble factor, it is mainly expressed in osteoblast lineage cells of the bone, but also by a manifold of other cell types including dendritic and B cells of the immune system, vascular endothelial cells, and various epithelial cells (Simonet et al. 1997). Osteoprotegerin is part of the RANKL/RANK/OPG pathway regulating and balancing osteoclast and osteoblast activity as a decoy receptor to RANKL (Zaidi et al. 2023). Our findings are in alignment with the research conducted in the Korean CKD cohort KNOW-CKD. They found osteoprotegerin levels to be independently associated with CKD progression (Oh et al. 2022). The exact mechanism of a) how osteoprotegerin is activated during CKD and b) its exact role in the fostering of cardiovascular complications remain largely unclear to date. Stimulators of osteoprotegerin secretion other than CKD are widely debated and include, amongst others, proinflammatory signals (Marques et al. 2021). In our data, hsCRP was included in the COX regression models to rule out an effect of nonspecific inflammation on the correlation between serum osteoprotegerin levels and investigated outcomes. However, osteoprotegerin is known to be directly secreted by immune cells, including B cells, thus, in inflammatory conditions, it is likely to play a role in modulating the bone together with other proinflammatory cytokines and influence the course of CKD-MBD (Mazzaferro et al. 2021). To further understand the kinetics involved, it would be intriguing to generate a time course profile of osteoprotegerin expression in health and disease in order to more tightly define osteoprotegerin kinetics in CKD patients. Furthermore, it will be of value to measure more parameters involved in bone metabolism, such as RANKL, RANK, TRAIL, and sclerostin to gain a more complete picture of what is happening during the progression of CKD. This will likely contribute to unveiling the pathophysiology of CKD-MBD and subsequently leading towards precise targets for intervention and modification.
Cardiovascular complications pose a major threat to patients with CKD-MBD. In our 4246 CKD patients, out of nine CKD-MBD biomarkers osteoprotegerin correlates best with four endpoints MACE, hospitalization for CHF, cardiovascular and overall mortality, illustrating this relationship. Osteoprotegerin holds great potential as a biomarker in the context of CKD-MBD and might help to stratify those patients at risk for complicative events during their course of disease, paving the way for early onset treatment and intervention.
We thank all patients, GCKD investigators and sponsors for their continued contribution. For more information please visit the GCKD website .
Follow the Topic
-
Bone Research
This journal highlights the breakthrough discoveries in basic and clinical aspects of bone biology, pathophysiology and regeneration, as well as other significant findings related to bone.

Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in