Composition of the gut microbiome is associated with response and toxicity following anti-CD19 CAR T cell therapy
Published in Cancer
What is the relevant background?
The intestinal microbiome has emerged as an important endogenous factor that is associated with outcomes in cancer and cancer therapy. Studies have confirmed that the intestinal microbiome can modulate the anti-tumor immune response to chemotherapy, radiotherapy, immune checkpoint blockade, allogeneic hematopoietic cell transplantation, and adoptive cellular therapy [1,….12]. Cellular immunotherapy with CD19-targeted chimeric antigen receptor (CAR) T cells has provided new therapeutic options for patients with high-risk hematologic malignancies [13,14,15,16]. Yet, patients may experience disease relapse or CAR-mediated toxicity, cytokine release syndrome (CRS) or immune effector cell-associated neurotoxicity syndrome (ICANS) [17,18,19,20,21,22]. The contribution of the intestinal microbiome on the function of CAR T cell anti-tumor activity and toxicity is unknown.
What did we investigate?
In a multi-center study between Memorial Sloan Kettering Cancer Center and the University of Pennsylvania, we analyzed the association between clinical outcomes and (1) antibiotic exposure prior to CAR T cell infusion and (2) the composition and diversity of the fecal microbiome.
What did we report about antibiotic exposure?
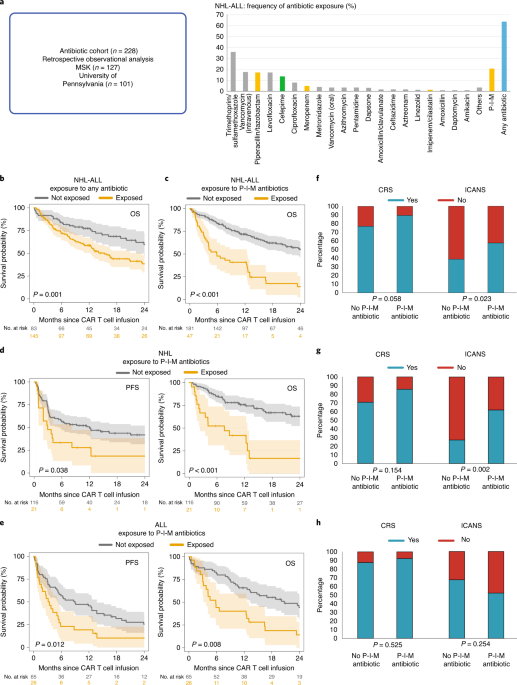
For evaluation of antibiotic exposure, we retrospectively collected clinical data and antibiotic exposures from patients with acute lymphoblastic leukemia (ALL, n=91) and non-Hodgkin lymphoma (NHL, n=137) treated with investigational or commercial CD19 CAR T cells at both centers. We focused our analysis on anaerobe-targeting antibiotics used in the setting of neutropenic fever: piperacillin-tazobactam, imipenem-cilastatin, and meropenem (here referred to as “P-I-M”). We found that overall survival (OS) was significantly decreased following CAR T cell infusion in patients exposed to P-I-M. We queried whether patients exposed to P-I-M had worse fitness or more aggressive disease. In in a multivariate model, we found that exposure to P-I-M, higher pre-lymphodepletion lactate dehydrogenase, and worse ECOG performance status were strong predictors of decreased OS. Exposure to P-I-M was also associated with increased ICANS but not CRS in patients in the combined NHL and ALL cohort as well as in patients with NHL.
What did we report about the fecal microbiome?
For assessment of the fecal microbiome, we prospectively collected baseline fecal samples prior to cell infusion from CD19 CAR T cells recipients (n=48). Samples were submitted for 16S ribosomal RNA sequencing of the V4-V5 region as well as metagenomic shotgun sequencing. In comparison to healthy controls (n=30), CD19 CAR T cell treated patients had an altered fecal microbiome before cell infusion as noted by lower alpha-diversity, increased frequency of bacterial dominance, and distinct bacterial composition. We evaluated the association of bacterial taxa and complete response and toxicity. Specifically, we found that species within the class Clostridia were associated with Day 100 complete response through both untargeted and targeted analysis of 16S sequencing. Metagenomic shotgun sequencing also identified that specific metabolic pathways were associated with Day 100 complete response as well as toxicity.
How is our study impactful?
In summary, our findings indicate that antibiotic exposure prior to CD19 CAR T cells is associated with poor outcomes, including decrease OS and increased ICANS. Our results also highlight that biomarkers of the intestinal microbiome in baseline fecal samples of CD19 CAR T cell recipients are associated with efficacy and toxicity therapy. These data provide insights as to how the intestinal microbiome can be harnessed to improve clinical outcomes of patients treated with cellular therapies.
https://www.nature.com/articles/s41591-022-01702-9
- Viaud, S. et al. The intestinal microbiota modulates the anticancer immune effects of cyclophosphamide. Science 342, 971–976 (2013).
- Daillère, R. et al. Enterococcus hirae and Barnesiella intestinihominis facilitate cyclophosphamide-induced therapeutic immunomodulatory effects. Immunity 45, 931–943 (2016).
- Uribe-Herranz, M. et al. Gut microbiota modulate dendritic cell antigen presentation and radiotherapy-induced antitumor immune response. J. Clin. Invest. 130, 466–479 (2020).
- Yang, K. et al. Suppression of local type I interferon by gut microbiota-derived butyrate impairs antitumor effects of ionizing radiation. J. Exp. Med. 218, e20201915 (2021).
- Sivan, A. et al. Commensal Bifidobacterium promotes antitumor immunity and facilitates anti-PD-L1 efficacy. Science 350, 1084–1089 (2015).
- Gopalakrishnan, V. et al. Gut microbiome modulates response to anti–PD-1 immunotherapy in melanoma patients. Science 359, 97–103 (2018).
- Matson, V. et al. The commensal microbiome is associated with anti-PD-1 efficacy in metastatic melanoma patients. Science 359, 104–108 (2018).
- Vétizou, M. et al. Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350, 1079–1084 (2015).
- Routy, B. et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science 359, 91–97 (2018).
- Andrews, M. C. et al. Gut microbiota signatures are associated with toxicity to combined CTLA-4 and PD-1 blockade. Nat. Med. 27, 1432–1441 (2021).
- Peled, J. U. et al. Microbiota as predictor of mortality in allogeneic hematopoietic-cell transplantation. N. Engl. J. Med. 382, 822–834 (2020).
- Uribe-Herranz, M. et al. Gut microbiota modulates adoptive cell therapy via CD8α dendritic cells and IL-12. JCI Insight 3, e94952 (2018).
- Park, J. H. et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N. Engl. J. Med. 378, 449–459 (2018).
- Lee, D. W. et al. T cells expressing CD19 chimeric antigen receptors for acute lymphoblastic leukaemia in children and young adults: a phase 1 dose-escalation trial. Lancet 385, 517–528 (2015).
- Davila, M. L. et al. Efficacy and toxicity management of 19-28z CAR T cell therapy in B cell acute lymphoblastic leukemia. Sci. Transl. Med. 6, 224ra225 (2014).
- Maude, S. L. et al. Tisagenlecleucel in children and young adults with B-cell lymphoblastic leukemia. N. Engl. J. Med. 378, 439–448 (2018).
- Chong, E. A., Ruella, M. & Schuster, S. J. Five-year outcomes for refractory B-cell lymphomas with CAR T-cell therapy. N. Engl. J. Med. 384, 673–674 (2021).
- Abramson, J. S. et al. Lisocabtagene maraleucel for patients with relapsed or refractory large B-cell lymphomas (TRANSCEND NHL 001): a multicentre seamless design study. Lancet 396, 839–852 (2020).
- Sotillo, E. et al. Convergence of acquired mutations and alternative splicing of CD19 enables resistance to CART-19 immunotherapy. Cancer Discov. 5, 1282–1295 (2015).
- Orlando, E. J. et al. Genetic mechanisms of target antigen loss in CAR19 therapy of acute lymphoblastic leukemia. Nat. Med. 24, 1504–1506 (2018).
- Ruella, M. et al. Induction of resistance to chimeric antigen receptor T cell therapy by transduction of a single leukemic B cell. Nat. Med. 24, 1499–1503 (2018).
- Spiegel, J. Y. et al. Outcomes of patients with large B-cell lymphoma progressing after axicabtagene ciloleucel therapy. Blood 137, 1832–1835 (2021).
Follow the Topic
-
Nature Medicine
This journal encompasses original research ranging from new concepts in human biology and disease pathogenesis to new therapeutic modalities and drug development, to all phases of clinical work, as well as innovative technologies aimed at improving human health.
Related Collections
With Collections, you can get published faster and increase your visibility.
Microbiome and energy metabolism
Publishing Model: Hybrid
Deadline: Dec 06, 2026
The expanding therapeutic landscape of GLP 1 receptor agonists
Publishing Model: Hybrid
Deadline: Jan 23, 2027




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in