Contrasting single-molecule magnet behavior in dysprosium and terbium bis(stannolediide) complexes
The phenomenon in which an individual molecule exhibits a slow relaxation of magnetization due to an energy barrier separating spin-up and spin-down states, gave rise to the concept of single-molecule magnets (SMMs). These are molecular entities that are capable of retaining magnetic memory below a characteristic blocking temperature, even in the absence of an external magnetic field. Because of this property, SMMs have attracted attention for their potential applications in ultra-high-density data storage, quantum computing and spintronic devices.
The discovery of the first single-molecule magnet, a Mn12 acetate complex, in the early 1990s, demonstrated for the first time that magnetic memory can be stored at molecular level. Over the past three decades, close collaboration between synthetic chemists, physicists and theoreticians, have steadily pushed the performance of SMMs to higher temperatures, in some cases even beyond the boiling point of liquid nitrogen. A major breakthrough came in 2017, when highly axial dysprosium bis(cyclopentadienide) complexes have shown exceptional energy barriers to magnetic reversal (Ueff) and high hysteresis temperatures (TH). Further improvements were achieved in 2025 through the use of bulky amide ligands. More recently, dianionic heavy group 14 cyclopentadienides analogues containing one Si, Ge, Sn or Pb element, have become promising ligands due to their higher charge density. Therefore, making them as attracting alternatives for the design of highly axial dysprosium SMMs.
We entered this field in 2022 with the introduction of the plumbole dianion into f-element chemistry. For the first time, we incorporated a dianionic group 14 metallole into the coordination sphere of a lanthanide sandwich complex. In that sandwich system, the co-ligand was an eight-membered bulky silyl-functionalized cyclooctatetraendiide (COT) dianion. Because of its equatorial coordination mode, the magnetic properties of the erbium congener were dominated by the COT ligand rather than the plumbole ligand. Attempts to generate a homoleptic [L₂Ln]⁻ type complex with the plumbole dianion were unsuccessful at that time. We next explored the lighter silole and germole analogues, hoping they would deliver the homoleptic, solely axially coordinated [L₂Ln]⁻ type framework we were aiming for. Instead, the stronger nucleophilicity of the Si and Ge-based ligands led to the formation of dimeric species in both cases. These dimeric motifs introduced equatorial interactions at the lanthanide center, the type of coordination environment we were trying to avoid.
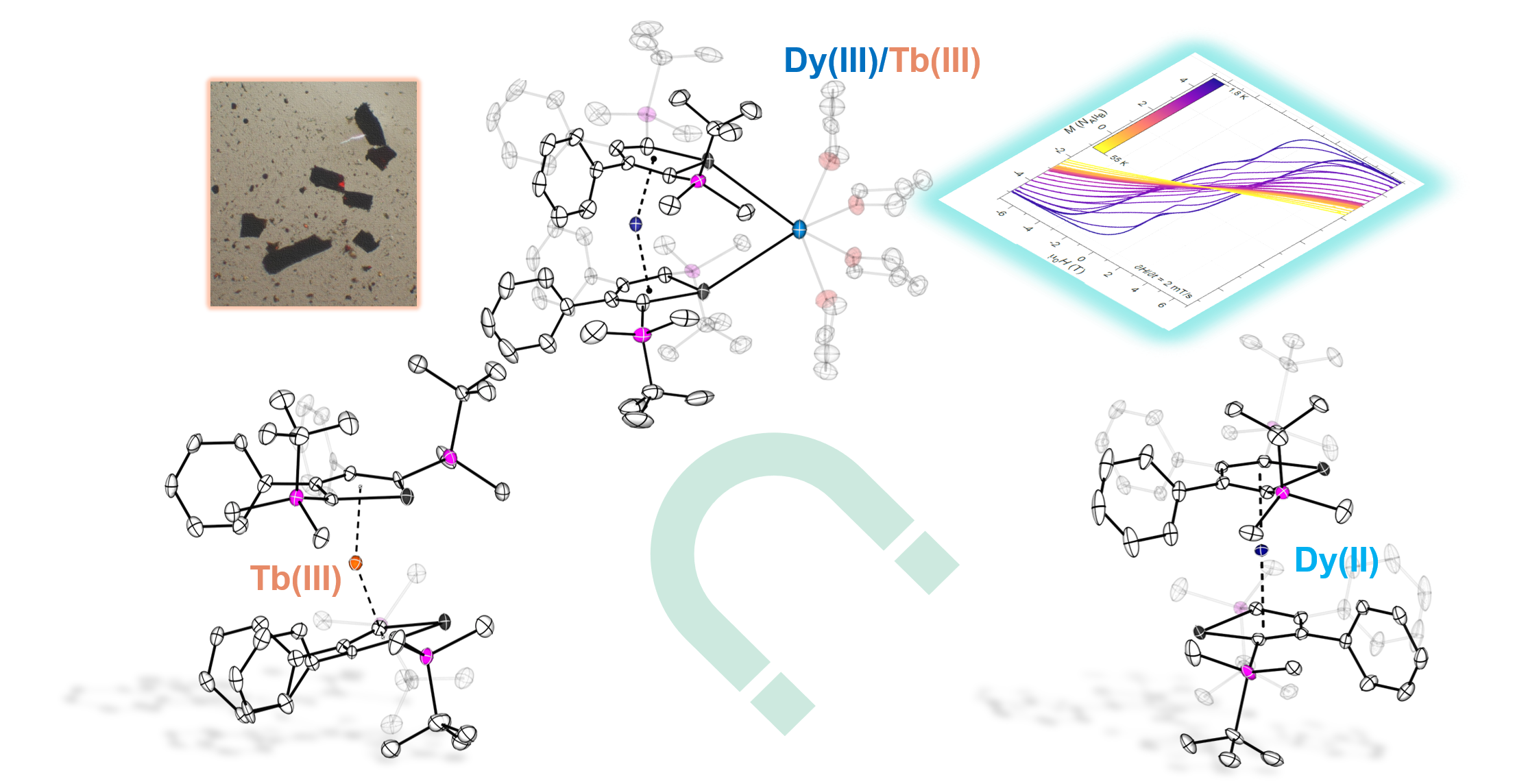
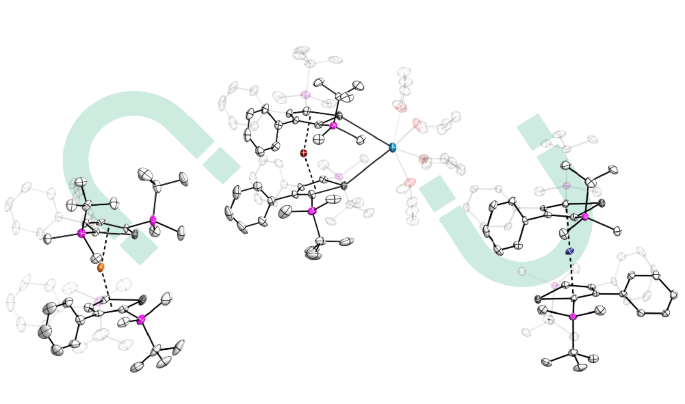
At this stage, the only group 14 metallole dianion which we have not explored, was the stannole dianion. While the stannole dianions have already been successfully used in d-block metal chemistry, including the generation of neutral and anionic sandwich complexes, it had never been used in lanthanide chemistry. We therefore focused on developing the synthesis of the hitherto unknown dipotassium salt of the stannole, which was essential for the synthesis of our aimed lanthanide complexes. Compared to the lighter silole and germole ligands, stannoles are generally less nucleophilic. Together with the steric shielding from the bulky silyl groups which we introduced at 2,5-positions, we anticipate that the dimer formation which we observed previously, should be largely suppressed. Luckily, the metathesis reaction with the lanthanide iodide starting materials proceeded cleanly and the homoleptic and monomeric bis(stannoldiide) complexes were isolated for trivalent dysprosium and terbium. Dy(III) was the obvious starting point of our investigation. its oblate shape of electron density and Kramers ion nature with 4f9 configuration, makes it the first choice for obtaining high-performance single-molecule magnets. Tb(III), its neighbour in the periodic table, has similar oblate shape but is a non-Kramers ion with only one electron less compared to Dy(III). This made it interesting in the structure and magnetic property comparison of both species. Not surprisingly, the Dy(III) and Tb(III) compounds are isostructural, with only minimal differences in their bonding metrics. For charge balance, one K+ cation remains in the product compounds, which is coordinating between two Sn(II) atoms, giving bending angles of 154.3° (for Dy) and 153.5° (for Tb). Synthesizing both complexes allowed us not only study how the heavy Sn-containing ligand influence the magnetic behaviour but also to perform a rare comparison of between the magnetic behaviour of the Dy(III) and Tb(III) systems with similar coordination environment. While the Dy(III) complex showed strong magnetic anisotropy with an energy barrier of 1502(4) K and open magnetic hysteresis up to 55 K. In contrast, The Tb(III) analogue showed a lower energy barrier of 553 K and open hysteresis up to only 9 K. Taking into account that the coordinated K+ cation might influence the bending angle of the system and thus the magnetic behaviour, we used 18-crown-6, a well-known K+ surrogate, for the generation of “naked” bis(stannolediide) lanthanidocenate anions. For Tb, the reaction proceeded straightforwardly and the isolated anionic sandwich complex is slightly more linear (156.3°) and displays slightly elongated Tb-Ct distances. This enabled us to directly compare the magnetic dynamics of the two Tb(III) complexes with similar ligand environment but potassium coordination/decoordination. Magnetic investigations showed that the “naked” anion exhibits faster magnetic relaxation with a lower barrier of 455 K and lower hysteresis temperature of 7 K. Most likely, subtle differences in spin–phonon coupling and relaxation pathways lead to different magnetic dynamics in the two terbium complexes. Unexpectedly, when the same strategy was applied to the dysprosium system, the reaction with 18-crown-6 did not simply generate the anticipated separated ion pair. Instead, it led to the formation of a rare divalent dysprosium complex, with significantly diminished anisotropy, attributed to its linear geometry and the electronic interplay between Dy(II) and Sn centers.
Overall, our work reported the synthesis of a series of homoleptic bis(stannolediide) complexes with Tb(III), Dy(III) and Dy(II) and a detailed comparison of their magnetic behaviour. The results showed that the stannole dianion can serve as an alternative ligand for the synthesis of high-performance Dy(III) single molecule magnets. The unexpected isolation of a Dy(II) complex highlights the strong reducing character of the stannole dianion, enabling the stabilization of unusual low oxidation states of 4f elements. This study opens new opportunities for using heavy group-14 metallole ligands in molecular magnetism and in accessing unusual lanthanide oxidation states. It also shows that unexpected reactions can sometimes lead to the most interesting discoveries in chemistry.
Follow the Topic
-
Nature Chemistry

A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in