Decoding Hydrogen‑Bond Network of Electrolyte for Cryogenic Durable Aqueous Zinc‑Ion Batteries
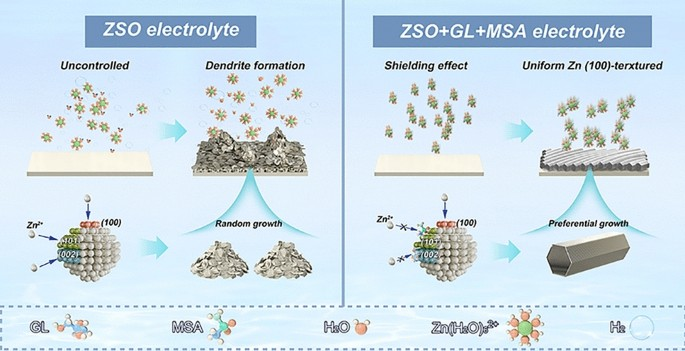
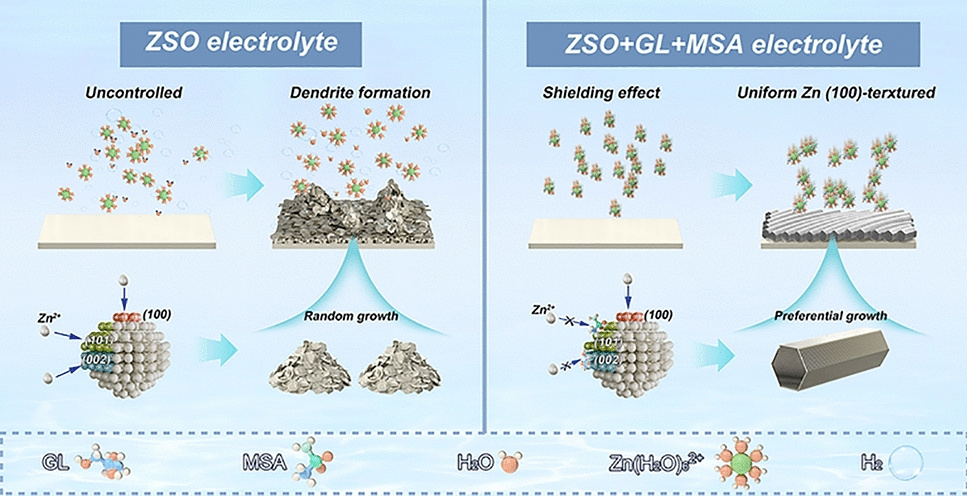
As renewable energy storage expands into polar and high-altitude regions, conventional aqueous zinc-ion batteries (AZIBs) face a triple threat: dendrite growth, hydrogen-evolution corrosion, and electrolyte freezing below –20 °C. Now researchers from Southern University of Science and Technology, Soochow University and Guilin University of Technology—led by Prof. Lin Zeng, Prof. Yongbiao Mu and Prof. Jingyu Sun—report a dual-additive electrolyte that re-wires the hydrogen-bond network, guides (100)-oriented zinc deposition, and delivers record lifespan from room temperature down to –40 °C. The work provides a universal recipe for cryogenic-tolerant, high-rate AZIBs deployable in extreme climates.
Why Hydrogen-Bond Reconstruction Matters
- Anti-Freeze & Anti-Dendrite: Glycerol (GL) and methylsulfonamide (MSA) raise configurational entropy, suppress ice nucleation and lower freezing point to –45 °C while blocking water access to the Zn surface.
- (100)-Textured Anode: Cooperative adsorption of GL on Zn(101) and MSA on Zn(002) channels growth along the highly active Zn(100) plane, cutting activation energy for stripping by 50 %.
- Entropy-Driven Kinetics: Elevated disorder offsets viscosity penalty, enabling 40 mA cm-2 reversible plating and 5 A g-1 cathode rates without concentration polarization.
Innovative Design & Features
- Dual-Additive Synergy: 50 vol % GL disrupts the H-bond lattice; 1 M MSA tunes solvation shell from 5.7 to 4.2 coordinated H2O per Zn2+, suppressing HER and forming a 7–10 nm SEI rich in ZnCO3 and ZnS.
- In-Situ Spectroscopy & MD: ATR-FTIR, Raman and NMR capture blue-shifted O–H modes; MD simulations reveal Cl- enrichment near GL and Li+ clustering along PEI chains, validating lane-separated ion transport.
- Cryo-Electrochemistry: Symmetric cells run 5400 h at –20 °C (0.5 mA cm-2, 0.5 mAh cm-2) and 600 h at 40 mA cm-2(30 °C), while full cells retain 85 % capacity after 2 000 cycles at –20 °C.
Applications & Future Outlook
- Grid Storage in Cold Climates: pouch cells (300 μL electrolyte) deliver 78 % capacity retention after 600 cycles at 1 A g-1, proving scalability for alpine micro-grids and polar base stations.
- Fast-Charging Devices: critical current density rises to 60 mA cm-2 at 30 °C and 25 mA cm-2 at –20 °C, unlocking 10-min charge capability for portable electronics and e-bikes.
- Electrolyte Platform: the entropy-boosted, H-bond-engineered concept is compatible with existing ZnSO4supply chains; next steps include antifouling polymers for natural brine and fire-retardant variants for aerospace.
This comprehensive study demonstrates that entropy-mediated hydrogen-bond reconstruction can simultaneously defeat dendrites, hydrogen evolution and electrolyte freezing, pushing aqueous zinc batteries into the cryogenic frontier. Stay tuned for more sub-zero breakthroughs from Prof. Lin Zeng, Prof. Yongbiao Mu and Prof. Jingyu Sun and their teams!
Follow the Topic
-
Nano-Micro Letters
Nano-Micro Letters is a peer-reviewed, international, interdisciplinary and open-access journal that focus on science, experiments, engineering, technologies and applications of nano- or microscale structure and system in physics, chemistry, biology, material science, and pharmacy.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in