Designing Carbon Catalysts for Green Hydrogen Peroxide Electrosynthesis
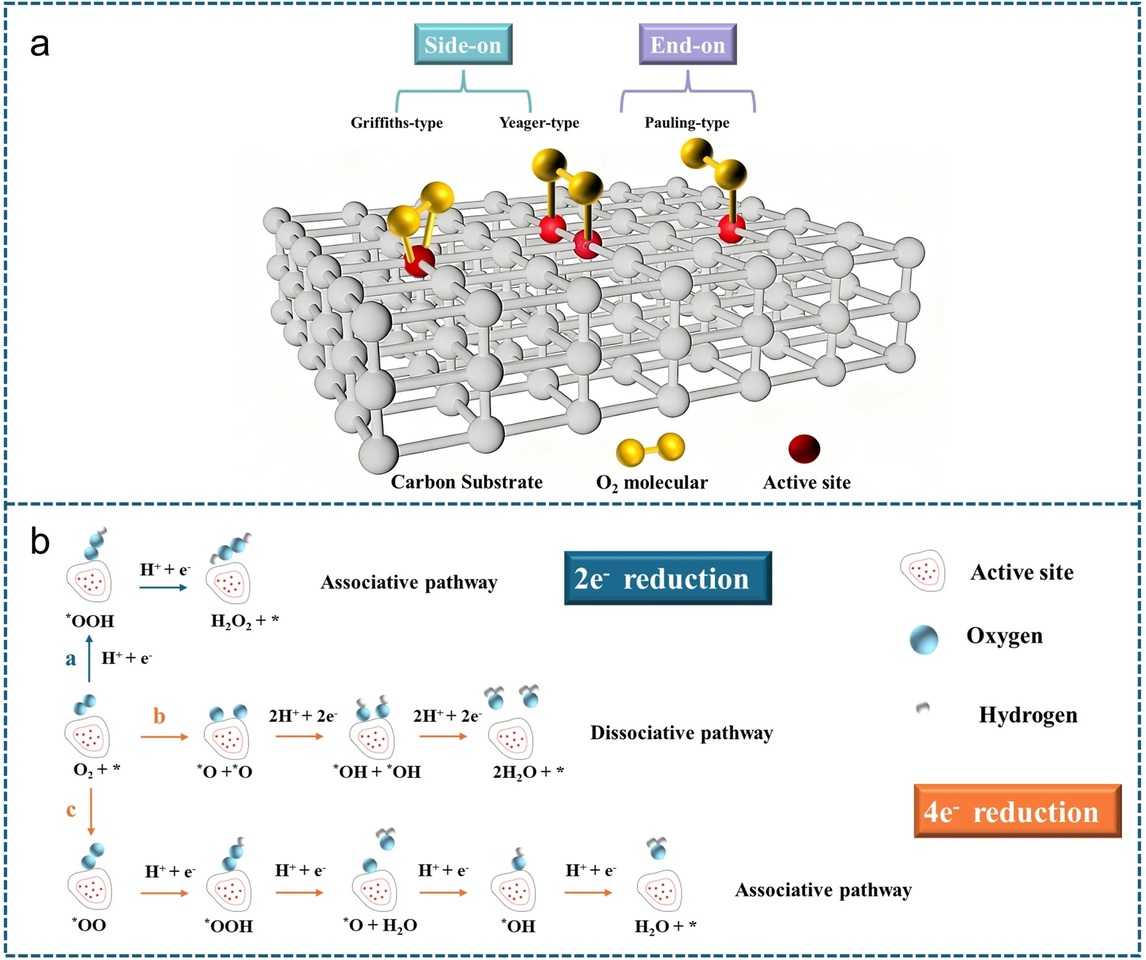
In this work, we provide a comprehensive review of carbon‑based electrocatalysts for the two‑electron oxygen reduction reaction (2e⁻ ORR) toward hydrogen peroxide (H₂O₂), a promising green alternative to the conventional anthraquinone process. We systematically summarize recent advances spanning metal‑free carbons, carbon‑supported noble and non‑noble metals, single‑ and dual‑atom catalysts, and MOF/COF‑derived materials, with catalyst design as the unifying theme. By integrating mechanistic insights—centered on regulation of the key *OOH intermediate—with materials engineering strategies, we establish clear structure–performance relationships to guide the rational development of highly selective, active, and durable catalysts for decentralized H₂O₂ electrosynthesis.
Key Insights
-
*OOH intermediate as the selectivity descriptor
The adsorption strength and stability of the *OOH intermediate determine the competition between the 2e⁻ pathway (H₂O₂ formation) and the 4e⁻ pathway (H₂O formation). Moderate *OOH binding is essential for achieving high selectivity and activity. -
Metal‑free carbon catalysts
Strategies including heteroatom doping (N, B, S, P, F), defect and edge engineering, carbon hybridization tuning (sp²/sp³/sp), and morphology regulation (porous, hollow, curved architectures) effectively tailor local electronic structures and reaction microenvironments, enabling efficient H₂O₂ production without metal leaching. -
Carbon‑supported metal catalysts
For both noble and non‑noble metals, particle‑size control, alloying, oxygen‑vacancy engineering, and strong metal–support interactions suppress O–O bond cleavage and steer ORR selectivity toward the two‑electron pathway. -
Single‑atom and dual‑atom catalysts
Precise regulation of first‑ and second‑shell coordination environments enables atomic‑level control over activity, selectivity, and stability, helping to overcome the long‑standing activity–selectivity trade‑off under industrially relevant current densities. -
MOF‑ and COF‑derived catalysts
Framework‑derived materials provide high active‑site density, tunable microenvironments, and enhanced mass transport, offering promising platforms for scalable and device‑integrated H₂O₂ electrosynthesis.
Significance
This review highlights carbon‑based electrocatalysts as a key materials platform for green, efficient, and decentralized hydrogen peroxide production. By unifying fundamental reaction mechanisms with catalyst design strategies, our work clarifies how atomic‑scale engineering translates into macroscopic performance. We further outline future directions, including AI‑assisted catalyst discovery, in situ/operando mechanistic studies, synergistic multi‑factor regulation, and reactor‑level integration, which are expected to accelerate the transition from laboratory research to practical, low‑carbon H₂O₂ manufacturing.
Author Information
Xuan Wei, Ying Cao, Yuxing Ma, and Rui Cao*
Key Laboratory of Applied Surface and Colloid Chemistry, Ministry of Education
School of Chemistry and Chemical Engineering, Shaanxi Normal University
Xi’an 710119, China
Corresponding authors:
📧 weixuan@snnu.edu.cn
📧 ruicao@snnu.edu.cn
How to Cite
Wei X., Cao Y., Ma Y., Cao R.
Carbon‑based electrocatalysts for selective two‑electron oxygen reduction.
Catal (2026) 2:5.
https://doi.org/10.1007/s44422‑026‑00019‑9
Follow the Topic
-
Catal

Catal is an open access journal covering full spectrum of catalysis critical advances. From biocatalysts to heterogeneous catalysts, it integrates fundamental and applied sciences. Catal offers a primary platform for researchers and practitioners in the field.





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in