Exploring Species-Specific Effector Proteins as Novel Fungicides: A Promising Approach for Crop Protection
Published in Chemistry, Plant Science, and Immunology
Structure-based designing is widely employed in the discovery of novel therapeutics in the treatment of human diseases. However, its application in developing new fungicides for crop protection is limited. The main challenge lies in identifying and obtaining small molecules with feasible suppressive activities against target proteins. Consequently, researchers are examining the interactions between pathogens and plant hosts to identify potential interfaces and agents that interfere with or disrupt such interactions. This approach could lead to the development of designer antifungal compounds with improved efficacy and biosafety.
Pathogenic microbes often secrete effector proteins that play a significant role in pathogenicity during their interactions with the hosts. In response, host plants deploy two defense mechanisms: Pathogen-Associated Molecular Pattern (PAMP)-Triggered Immunity (PTI) and effector-triggered immunity (ETI). Effector proteins, however, exhibit a pattern of rapid evolution characterized by instability and variability, which makes them less ideal as potential targets of antifungal agents. However, whether species-specific effector proteins are the exception or could be targeted is unknown.
The rice blast caused by Magnaporthe oryzae inflicts significant economic damages, resulting in 10-30% of the world's annual rice loss, equivalent to the food ration of approximately 60 million people. Current management strategies rely on various fungicides, including sterol demethylation inhibitors, mitochondrial respiration inhibitors, and melanin biosynthesis inhibitors. However, the emergence of fungicide resistance and environmental concerns necessitate the development of new fungicides with novel functional mechanisms. Previous studies suggested that plant papain-like cysteine proteases, such as RD19, XCP2, and RD21, localized in either vacuoles, cytoplasm, or multiple subcellular compartments of Arabidopsis thaliana, serve as central hubs in host immunity. Notably, A. thaliana AtSerpin1 and AtWSCP are different proteinase inhibitors exhibiting spatially differential activities against RD21.
To demonstrate that pathogens secrete effector proteins to circumvent host immunity and its potential application in crop protection, we examined the interaction between MoErs1, a species-specific effector protein secreted by M. oryzae, and OsRD21, a papain-like cysteine protease produced by rice (Oryza sativa) involved in rice immunity. We showed that MoErs1 exhibits structural similarity to the host water-soluble chlorophyll-binding protein (WSCP), indicating its potential as plant protease mimics that suppress OsRD21 activities.
While fungal effectors are crucial for pathogenicity, and our initial findings indicated the potential of targeting species-specific effectors for fungicidal effects, such functions or interactions are poorly understood. Most current investigations use existing chemical compounds to identify targets, which often yield limited progress. To circumvent such limitations, we proposed to design new chemicals based on the MoErs1-OsRD21 interaction. We reasoned that MoErs1 is an evolutionarily conserved effector, but it has no homologs in other species, which makes it an ideal candidate for evaluation as a fungicide.
We found that the interaction sites between MoErs1 and OsRD21 are mainly within a long and narrow surface region, and there are more hydrophobic amino acid residues on the interface, which suggests that flexible molecules may facilitate the binding. Due to their excellent metabolic stability and pharmaceutical properties, diaryl ethers were widely applied as agrochemical agents. In addition, the diaryl ether scaffold contains two aromatic ring systems and a flexible oxygen bridge, leading to sufficient molecular flexibility and excellent lipid solubility, which can significantly increase cell membrane permeability. Considering these advantages, we decided on the diphenyl ether ester as a core skeleton unit in the design. Based on a MoErs1-OsRD21 docking model, we designed the diphenyl ether ester compound FY21001 (Fig. 1). FY21001 inhibits MoErs1 functions and, significantly, it effectively controls rice blasts during field tests. Notably, MoErs1 is specific to M. oryzae. While this may limit FY21001 in managing other rice diseases, the high specificity may reduce the undesired consequences from broad-spectrum compounds.
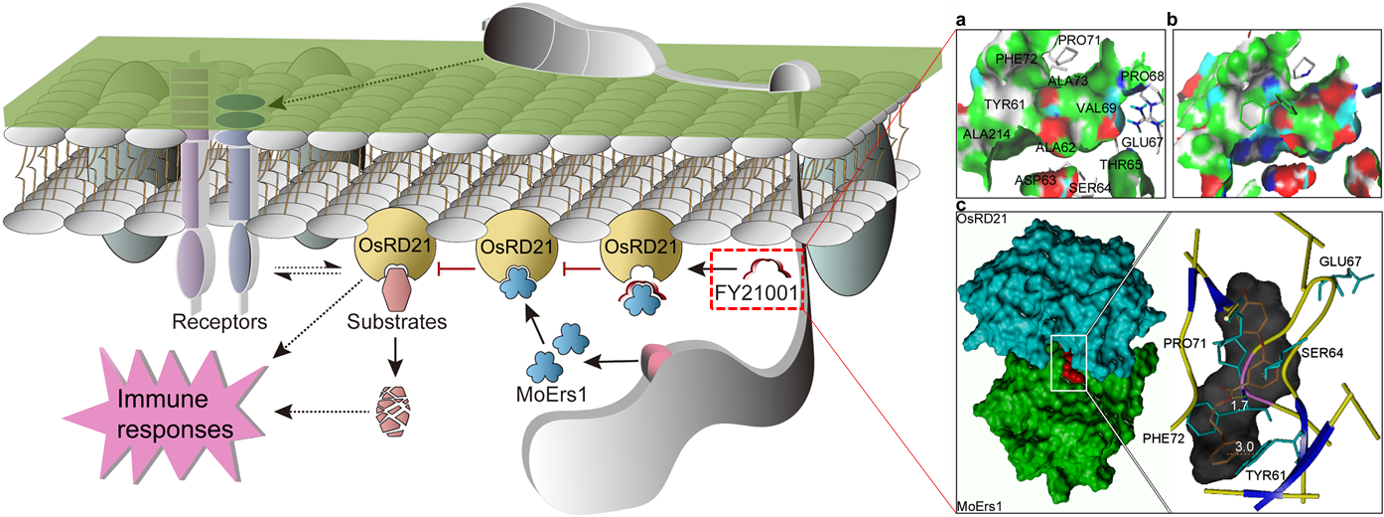
Fig. 1. Structure-based designing of the diphenyl ether ester compound. (a) The interaction region of MoErs1 and OsRD21. (b) The molecular docking model of diaryl ether-MoErs1. (c) The interaction model of MoErs1 (green)-OsRD21(blue), the red area represents the compound FY21001.
In summary, our studies revealed how synthetic compound FY21001 targeting M. oryzae-specific effector protein MoErs1 can be employed to control rice blasts (Fig. 2). Such an example opens up many possibilities for designing and developing novel fungicides that can effectively combat plant pathogens while minimizing environmental and animal health impacts.
Follow the Topic
-
Nature Plants
An online-only, monthly journal publishing the best research on plants — from their evolution, development, metabolism and environmental interactions to their societal significance.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in