Fragile Organoids, Handle with CUBE
Published in Bioengineering & Biotechnology

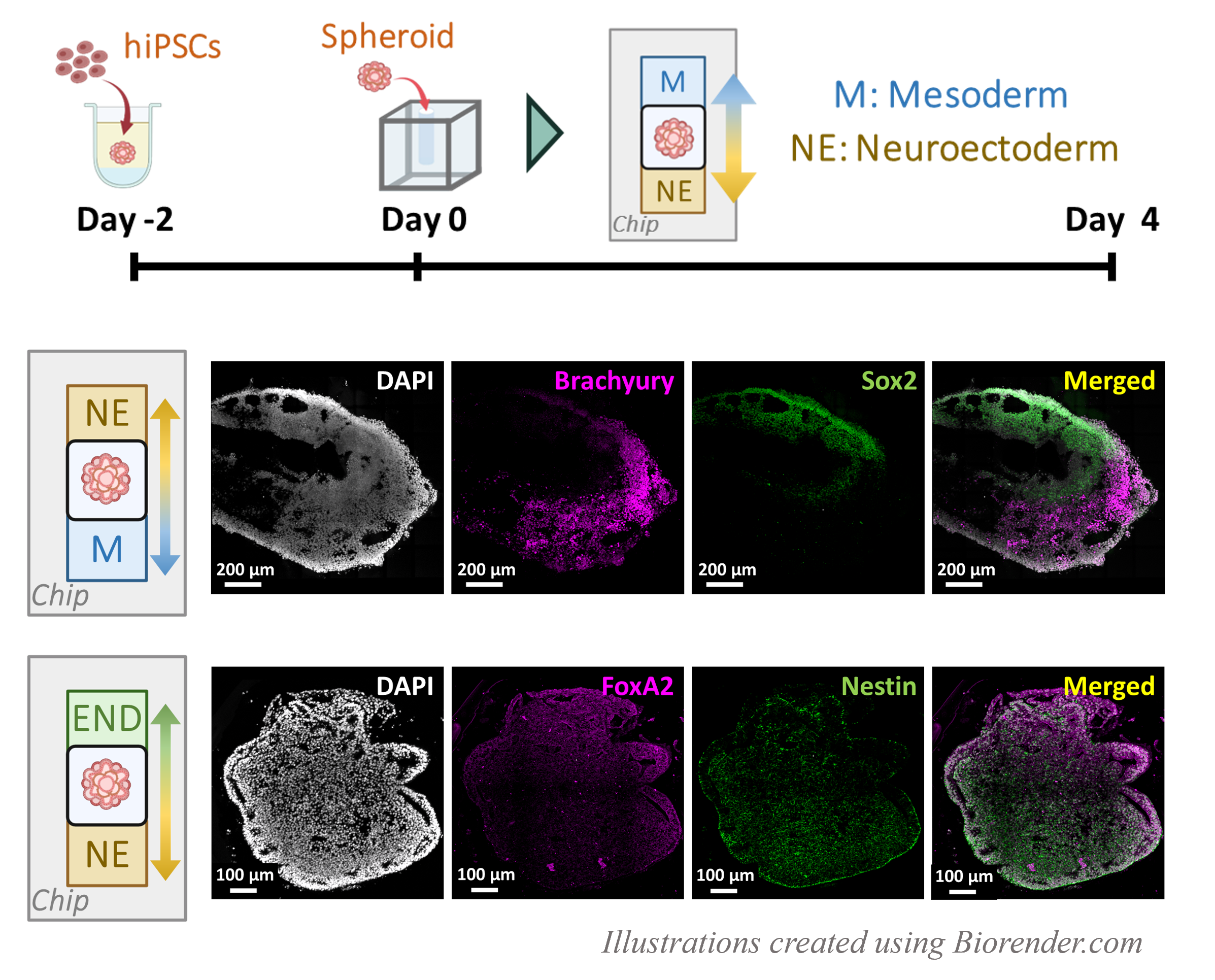
In the last decades, there has been a boom in stem cell research that has led to the generation of organoids (or more commonly referred to in laymen’s term as “mini organs”) that contain many of the cell types found in the native organ and exhibit some of their functions. These organoids are typically formed from a spherical cluster of cells suspended in a well-plate or encapsulated in an extracellular matrix (ECM) hydrogel and supplied with a cocktail of growth factors that prompt cells to differentiate to certain cell types and self-organise into something resembling an organ. However, this conventional culture method limits the potential for further developmental progress in organoids in the sense that all cells receive the same differentiation cues from the same growth medium in which they are suspended, whereas cells in the developing embryo may receive different signals from neighbouring cells depending on their spatial position within the embryo. For example, cells closer to the signal-secreting source will be stimulated to a different degree than cells further along the concentration gradient, which are likely closer to a different signal-secreting source. We therefore set about developing a platform to culture organoids with two different media to form opposing gradients of differentiation factors across the cluster of cells (Fig. 1).

Culturing cells with opposing gradients is relatively easy to achieve by devising a device with separate compartments for the cells and the different media. However, organoids are notoriously fragile and can easily get damaged when transferring to and from the culture container. Furthermore, it is near impossible to maintain or identify the gradient orientation of the organoid once taken out of the gradient culture device for post-differentiation analyses. In our article, we killed these two birds with one CUBE. The CUBE is a culture vessel comprising on the outside a hard-material frame and clear walls, with cells or organoids suspended in a hydrogel scaffold on the inside. As the size and shape of the CUBE is fixed, we can position the organoid in the CUBE in a precise and consistent manner by creating a seeding pocket in the CUBE. With the organoid contained within the CUBE, it can now be safely handled with a pair of tweezers to transfer the CUBE to a gradient culture device without causing damage to the sample. At the end of the differentiation, the organoid can be retrieved from the gradient device and sectioned together with the CUBE, using the CUBE frame as a reference for the orientation of the sample (Fig. 2).

As a demonstration of the application of this platform to organoid culture, we cultured iPSC spheroids in CUBEs with Neuroectoderm-Mesoderm (NE-M) and Neuroectoderm-Endoderm (NE-E) combinations of differentiation media using the gradient culture device, then sectioned the spheroids and immuno-stained for the differentiation markers Sox2-Brachyury for (NE-M) and Nestin-FoxA2 for (NE-E). The localized expressions of the differentiation markers to the side of the spheroid in which the respective differentiation medium was added confirmed that we were able to direct the differentiation of a single spheroid into two separate regions (Fig. 3).
We look forward to the utilization of this gradient culture platform to culture increasingly complex organoids such as those with dorsal-ventral and anterior-posterior body axes patterning, bringing us closer to better replicating the structure and functions of native organs to allow the study of human development, disease mechanisms, and drug treatment in vitro.
Behind the scenes: The struggles with fluidic devices
As most engineers may know, and most biologists may be blissfully unaware of, preventing or minimizing the leakage of fluids is a hard struggle, especially when only biocompatible materials and methods that do not cause harm to cells can be used. Where different types of materials are used (i.e. hard material of the CUBE and relatively softer material of the gradient device made of PDMS), the differences in surface roughness leads to tiny gaps in between the two material types that allows fluid to leak through. This is bad news if we are trying to form a gradient in the CUBE, as we do not want media leaking around between the outside of the CUBE and the culture device – this would lead to the media in separate chambers mixing together outside of the CUBE. To overcome this issue, we employed an ingenious invention commonly found in everyday life: the humble O-ring that seals your water-tight bottles, air-tight containers, washing machine pipes. O-rings are typically made from very soft and flexible materials that enable it to mould to and cover the tiny gaps between surface when pressure is applied. After much optimisation with regards to the material choice and design of the CUBE, gradient device, O-ring, and sealing method, we were able to finalize a protocol that we believe is easy to use and does not require many additional material, equipment, or specialist skills outside the comfort zone of a non-engineer.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in