Harnessing Solar Energy: Electrolyte-Assisted Charge Polarisation in Seawater Splitting at High Temperature
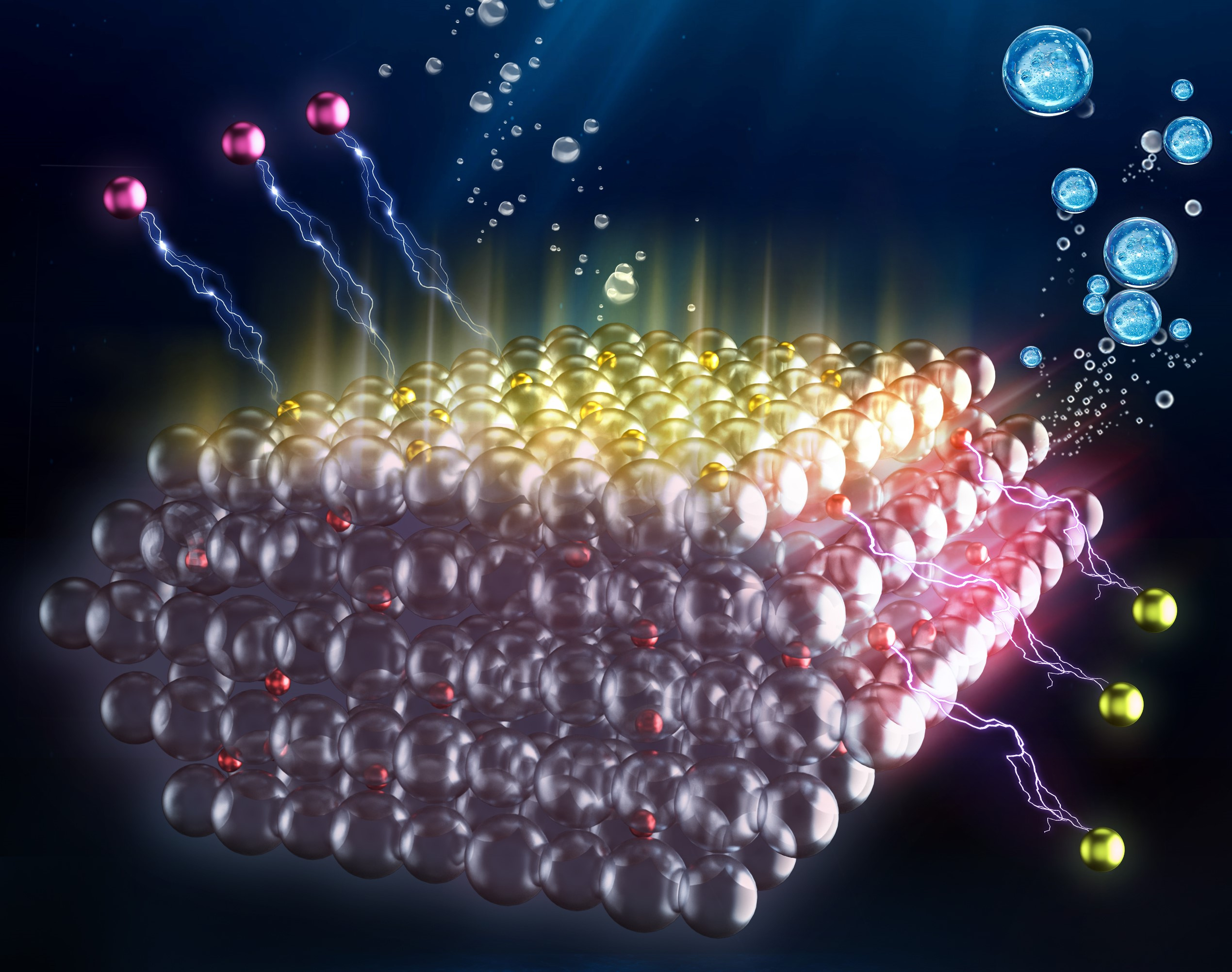
The pursuit of sustainable and efficient clean energy has led scientists to explore innovative approaches for decades. The generation of hydrogen – a light and highly reactive alternative to traditional fossil fuels – is considered as a promising strategy. One of the most clean and renewable pathways involves utilising abundant solar energy to split water (H2O) into hydrogen (H2) and oxygen (O2) using particulate photocatalysts. It is noteworthy that the use of vast amounts of pure water for H2 production may aggravate the shortage of freshwater resources, whereas treating impure water to a level of water purity, including desalination and deionisation, will increase the operation and maintenance cost of this technology. With over 95% of Earth's water resources residing in seas and oceans, solar-driven seawater splitting has emerged as a promising avenue for green H2 production. However, the hurdles associated with this process, such as compromised efficiency and stability, have necessitated novel solutions.
Overcoming Challenges in Seawater Splitting:
Traditional photocatalytic overall water splitting (POWS) reactions encounter limitations due to rapid recombination of the photo-generated charge carriers, resulting in low solar-to-H2 conversion efficiencies (ηSTH). Our previous studies have explored the use of local electric fields or magnetic fields to enhance charge separation (see Refs. 1 – 4). Considering these findings, we hypothesised that the large number of electrolytes in seawater might induce local electric fields, potentially improving the POWS reaction activity. However, as previously mentioned, seawater splitting presents its own challenges, including compromised energy conversion efficiency and poor photocatalyst stability in saline environments.
In this work, we have developed an electrolyte-assisted charge polarisation technique over an N-doped TiO2 (N-TiO2) photocatalyst, showcasing its effectiveness in achieving stoichiometric evolution of H2 and O2 from solar-driven seawater splitting. Remarkably, seawater, without any pre-treatment, significantly enhanced the photocatalyst's performance at 270°C, producing H2 and O2 steadily without by-products. To further improve performance, we tested five artificial seawater samples simulating the seawater in different regions of the world. Impressively, the Dead Sea water, which had the highest salt content, showed the best photocatalytic activity of 34.04 mmol g-1 h-1, surpassing reported values in similar systems.
Photocatalytic Performance Evaluation
Quantum efficiency (QE) measurements, indicating the ratio of produced H2 to incident photons, revealed that the Pt (1 wt%) supported N-TiO2 photocatalyst's QE was significantly enhanced by the electrolyte-assisted polarisation effect. When the artificial Dead Sea water was used, the QE exceeded 90% at short wavelengths and maintained over 50% even at 850 nm, bearing in mind that most literature in this field report QE of less than 40%.
Although QE provides valuable information for understanding the fundamental aspects of this system, we also need other evaluations to assess how well this system may perform in real world. One of such properties that are more related to practical applications is , which indicates the ratio of the produced H2 energy to the input of solar energy. In this work, an of 20.2±0.5% is achieved from this particulate POWS system in the artificial Dead Sea water at 270 oC, which is well over the goal of 10% for practical applications of POWS systems imposed by the United States Department of Energy.
Since the POWS system in this work is operated at 270 oC, we must consider the energy input for heating up the reaction system and maintaining the reaction temperature. Therefore, the overall energy conversion efficiency () has been evaluated subsequently. A very promising of 15.9±0.4% is demonstrated over 8 hours (i.e., typical hours of daylight). Furthermore, we attempted to demonstrate the feasibility of solar heating: a lab-scale light concentrated furnace was used to mimic a solar furnace, which shows that the stable and efficient POWS activity at 270 oC can be maintained solely by the concentrated light without any other light sources or heating devices. Thus, in a future scenario, we anticipate that the energy for heating could be provided by concentrated solar light, which will minimise or even exclude the energy consumption for heating.
Understanding the Mechanism
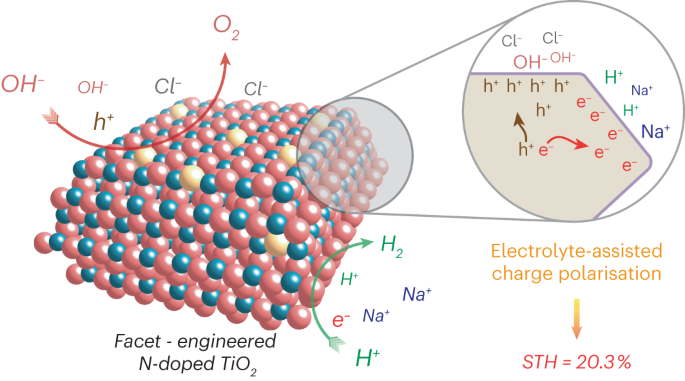
Beyond photocatalytic performance, it is also crucial to understand the mechanism of this electrolyte-assisted polarisation effect. Early attempts in literature revealed conflicting views regarding the effect of seawater on photocatalytic performance. In our study, it has been unambiguously shown that charge carrier lifetimes are prolonged in the presence of electrolytes, confirmed by the time-resolved photoluminescence spectroscopy. Scanning transmission electron microscopy was also employed, with Pt and CoOx nanoparticles photo-deposited as indicators for surface charges of the facet-controlled N-TiO2. It is shown that the photogenerated electrons and holes preferentially migrate to the (101) and (001) facets of N-TiO2, respectively. This facet-specific charge migration was further supported by near ambient pressure X-ray photoelectron spectroscopy studies. As charges can go to different facets selectively, it means the electrons and holes will separate spatially upon solar irradiation, resulting in prolonged charge carrier lifetimes and enhanced photocatalytic activities. In addition, the charged facets could attract ions of the opposite charge, creating local electric fields. These local electric fields, in turn, will further facilitate the facet-specific charge migration. This mechanism was also supported by the density functional theory calculations.
Future Prospects and Applications
The successful implementation of electrolyte-assisted charge polarisation over particulate photocatalysts opens up exciting possibilities for advancing seawater splitting as a viable method for solar H2 production. The implications of this research may extend to other photocatalytic processes and contribute to the development of advanced materials for energy conversion. By significantly improving the efficiency and stability of the photocatalytic process in saline environments, this research marks a critical milestone towards sustainable and scalable H2 production without exacerbating global freshwater scarcity. In conclusion, the fusion of solar power and seawater splitting opens doors to a greener future, where H2 production is not only efficient but also environmentally conscious. In conclusion, the combination of solar power and seawater splitting opens doors to a greener future, where H2 production is not only efficient but also environmentally conscious.
References
- Li, Y. et al. Local magnetic spin mismatch promoting photocatalytic overall water splitting with exceptional solar-to-hydrogen efficiency. Energy Environ. Sci. 15, 265–277 (2022).
- Li, Y. et al. Photocatalytic water splitting by N-TiO2 on MgO (111) with exceptional quantum efficiencies at elevated temperatures. Commun. 10, 4421 (2019).
- Li, Y. et al. 2D photocatalysts with tuneable supports for enhanced photocatalytic water splitting. Today 41, 34–43 (2020).
- Foo, C. et al. Characterisation of oxygen defects and nitrogen impurities in TiO2 photocatalysts using variable-temperature X-ray powder diffraction. Commun. 12, 661 (2021).
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in