Illuminating diamondoid soft porous crystals reveals the facets of their structural flexibility
Published in Chemistry
Covalent organic frameworks, or COFs for short, are nanoporous framework materials that are completely built up from strong covalent bonds between organic building units. They are light, metal-free, tunable, and stable materials, and have incredible internal surface areas. These properties make them perfect for a large variety of applications, such as catalysis, drug delivery, and more. While (stimulus-driven) flexibility would endow the materials with even more functionality, discovering flexible COFs is usually a serendipitous occasion because of their strong bonds. As such, design strategies to systematically discover new ‘soft’ COFs are highly sought after.
While investigating which COFs show structural flexibility, we found that practically all exhibited an interpenetrated diamondoid topology. Interpenetration regularly occurs in porous framework materials, as the void space within one framework can be occupied by one or more independent frameworks. Although they are not connected to one another, the interpenetrated frameworks cannot be separated without breaking any bonds, creating a unique collective framework. While reducing the available void space, the increased interactions between the individual frameworks typically prevent large frameworks from collapsing and increase the accessible surface area within a given volume. However, the impact of interpenetration on the possible flexibility of a material has yet to be explored. As such, we were intrigued by how–and to which extent–this diamondoid framework topology could give rise to ‘softness’, despite the strong covalent bonds, and what role the interpenetration plays in its structural flexibility.
Shedding light on the diamondoid framework
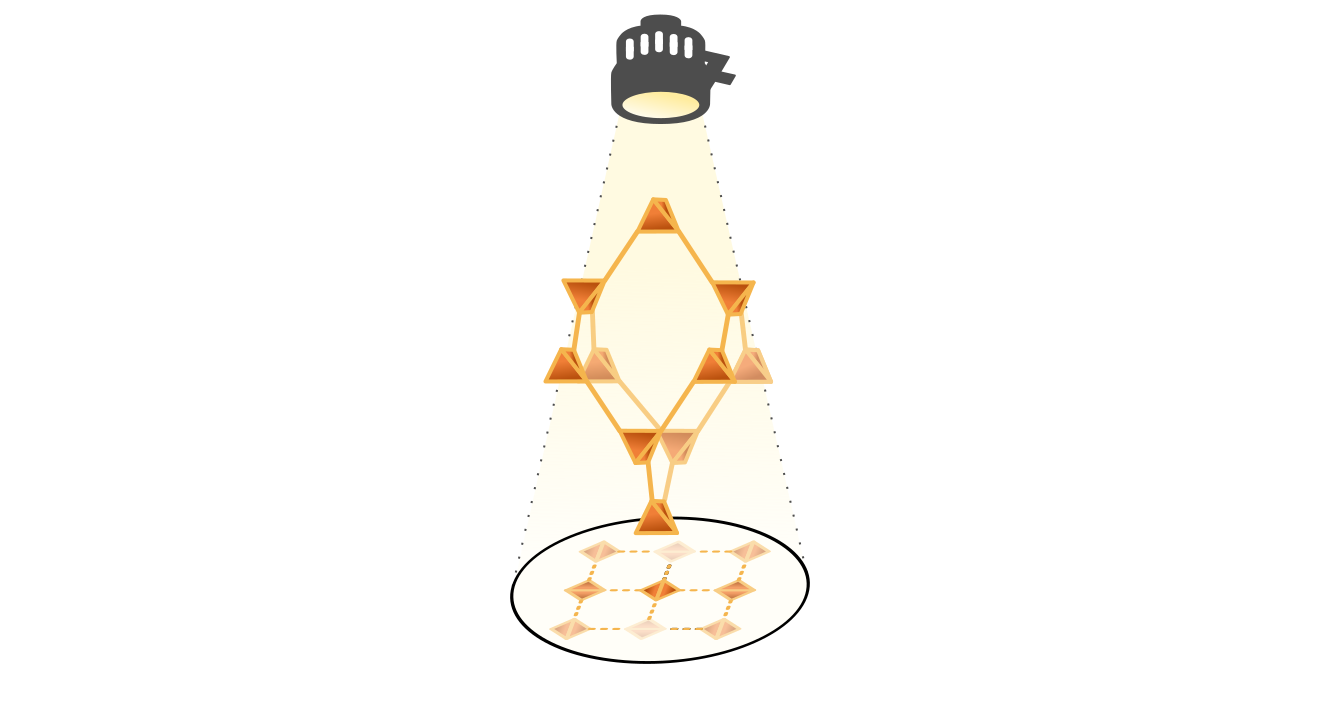
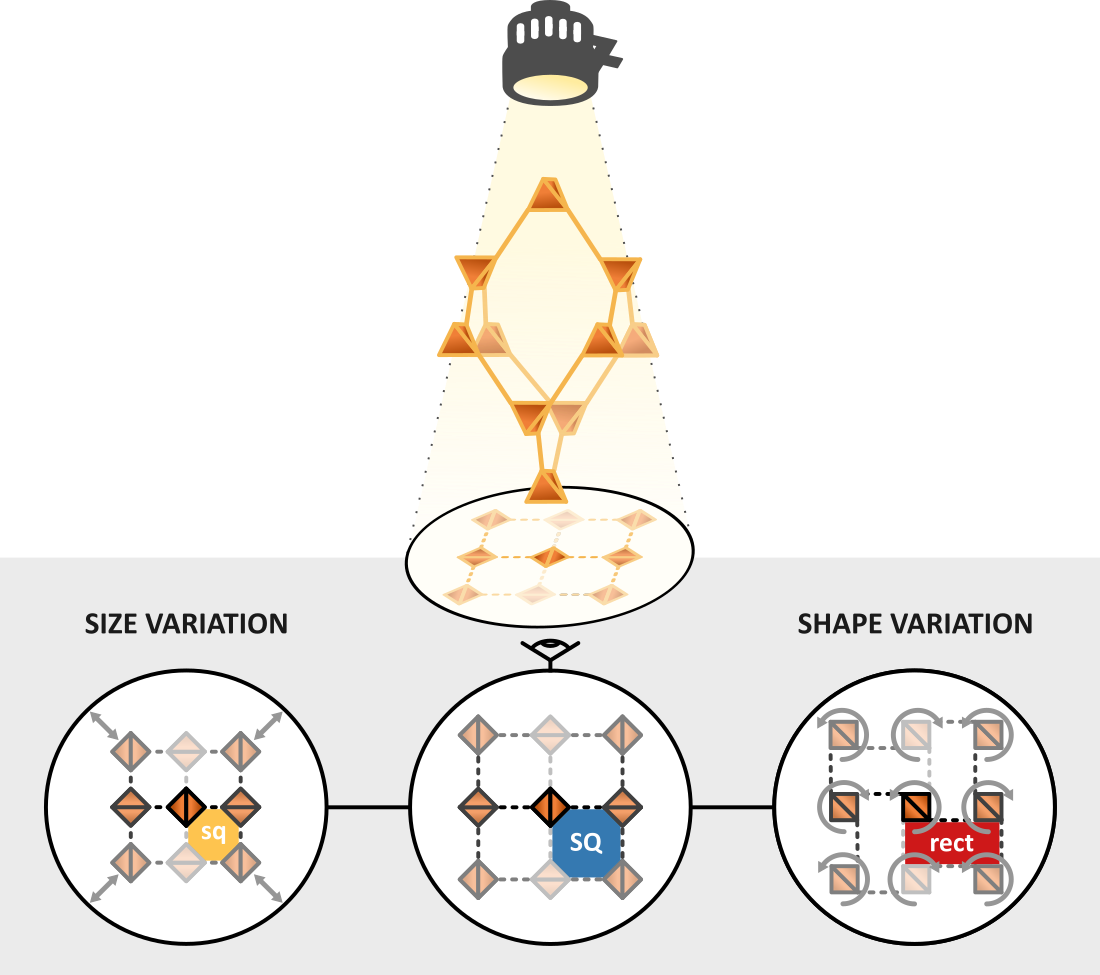
When looking at the diamondoid framework from a specific angle, the square-shaped channels of Fig. 1 appear. As interpenetration typically happens along this channel direction, these channels appear regardless of the number of interpenetrating frameworks. More interestingly, in experiments, the shape and size of these channels vary widely among several diamondoid materials, driven by external stimuli such as the presence of guest molecules. As such, to understand where this potential for flexibility comes from and to unearth the necessary conditions for it to come to expression, we needed to uncover the underlying transition mechanism. Through our simulations, we discovered that the channel size and shape variations are driven by a collective motion in which neighbouring vertices rotate and move. This collective motion is accompanied by an elongation or compression along the channel direction of the framework, similar to how the width of a rubber band compresses when pulling along its length. However, since this motion is only feasible when the connections between the vertices are pliable, our simulations reveal design principles a diamondoid material should satisfy in order to be flexible.
Promoting or suppressing softness
After identifying the internal mechanism behind the channel variations, we explored different approaches to impact flexibility. To this end, we systematically varied the degree of interpenetration for a series of materials, chosen based on the pliability of the connections between the vertices. Afterward, we also considered the influence of external triggers, such as temperature and the presence of guest molecules, to replicate experimental observations. Our results reveal that the potential for flexibility can be entirely quenched by choosing rigid connections, as they suppress the internal transition mechanism. Interestingly, the number of interpenetrating frameworks can also fundamentally shift the equilibrium between stabilization and destabilization effects, affecting the structural flexibility. This originates in the variation of the attractive and repulsive interactions between the interpenetrating frameworks. Finally, while we discovered that temperature could not straightforwardly be used as a driving force to (de)stabilize certain channel phases, introducing water molecules can induce size variations in line with experimental results. This illustrates that our current methodology can reproduce experimentally observed structural flexibility and paves the way for the further exploration of soft porous crystals.
This work has been performed at the Center for Molecular Modeling (CMM), an interdisciplinary research group at Ghent University.
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Sustainable waste management through polymer upcycling
Publishing Model: Open Access
Deadline: May 31, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in