Journal Club | All-organic liquid crystals with photomagnetic response
Published in Chemistry
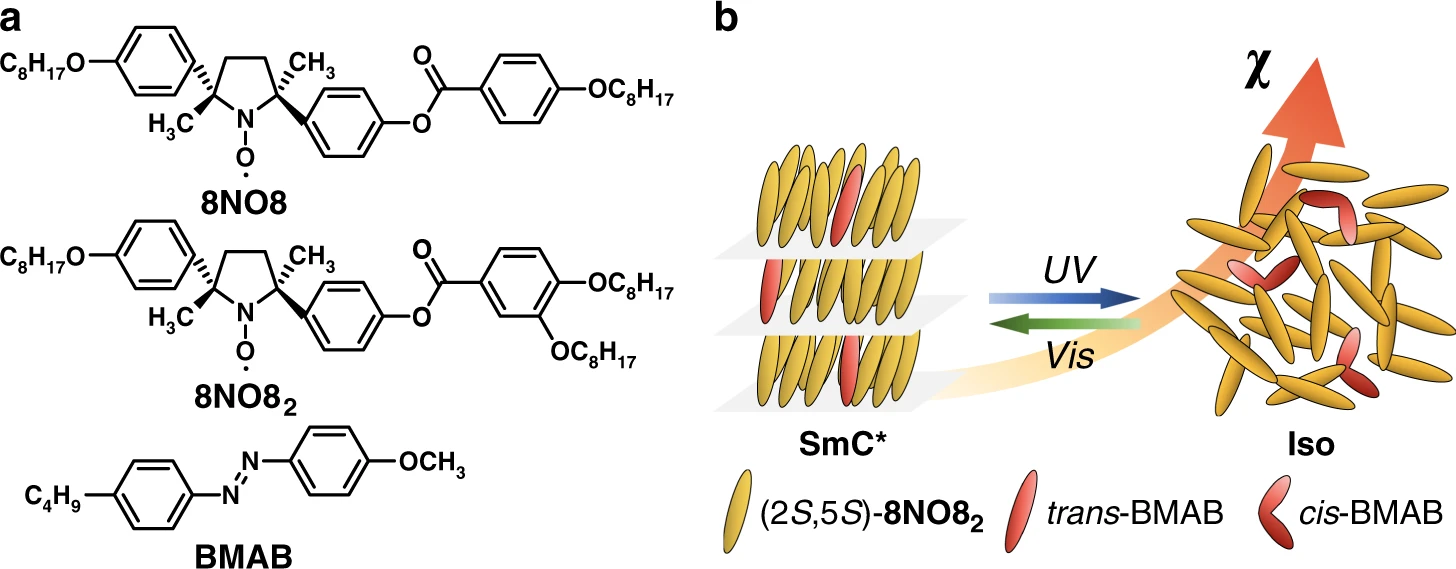
The orientation of molecular magnets in LCs can be controlled by magnetic and electric fields, through the magnetic and dielectric anisotropies of the LC-forming molecules. Due to the high viscosity of metal-containing LCs, the necessary fields to position the molecular magnets are quite strong (> 1 T). Metal-free LCs are easier to control, however, their magnetic properties can be hard to measure, as their inherent field strength is extremely low.
In a recent Communications Chemistry publication, Yoshiaki Uchida and colleagues from Osaka University study a new, all-organic LC material built up from comparably small, stable, chiral, and radical nitroxide molecules they call 8NO8_2 and an azobenzene derivative. These components show ferromagnetic spin-spin dipole interactions in magnetic fields – referred to as the "positive magneto-LC effect". The change in the overall magnetic properties at the LC melting point is large enough to be readily detectable by SQUID magnetometry in a magnetic field of only 0.5 T.
The inclusion of an azobenzene photoswitch allows the magnetic properties of the LC to be controlled by light. Azobenzenes have been widely used to enable photoswitching of LCs. Ultraviolet and visible light irradiation respectively induce trans-to-cis and cis-to-trans photoisomerization of the azobenzene. Here, when the azobenzene is in its trans conformation, the LC material in its ordered state is paramagnetic with a normal magnetic susceptibility. When irradiated with UV light, the azobenzene photoisomerizes to its cis conformation, which disturbs the LC structure. This induces a phase transition to the disordered liquid state, which shows high magnetic susceptibility. Additionally, the material naturally changes in its magnetic properties when melting and solidifying.
As a result of this elegant design, unlike previous magnetically active LCs, this system is not only influenced by temperature and the presence of even weak magnetic fields. The easily detectable magnetic properties of this metal-free LC material can be turned up or down simply by flipping the light switch, as it loses and regains its LC structure through photoisomerization.
Follow the Topic
-
Communications Chemistry
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the chemical sciences.
Related Collections
With Collections, you can get published faster and increase your visibility.
Chemical modification of proteins
Publishing Model: Open Access
Deadline: Jun 30, 2026
Experimental and computational methodology in structural biology
Publishing Model: Open Access
Deadline: Jul 31, 2026







Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in
I'm one of the authors of this paper. Thank you for your simple commentary on this recondite theme.
It is generally difficult to understand the concept "magneto-LC effect" because it is different from the other magnetic effects. The LC material in its ordered states like crystals is magnetic off-state and that in its disordered state like LCs and liquids is magnetic on-state. Therefore, irradiated with UV light, the azobenzene photoisomerizes to its cis conformation and the LC material in its disordered state is magnetically active.
We will theoretically explain the mechanism of this peculiar magnetic properties of the materials in the next paper; this magnetic properties are enhanced by molecular motion and structural disorder.
P.S. The new molecule reported in the paper is not 8NO8 but 8NO8_2
Thank you for your comment! I now corrected the error in the post, thanks for pointing that out.
Thank you for your correction. But, the switching behavior you wrote remains the opposite.
Liquid state = on (disorder but it has high magnetic susceptibility)
LC state = off (ordered but it has normal magnetic susceptibility)
And all states are paramagnetic.
Thank you so much for clarifying, I clearly misunderstood! Please accept my apologies, I corrected the post.
Thank you for your quick reply. No problem. This phenomenon is counterintuitive.