Neuronal organoids to study patient heterogeneity and personalized in vitro drug testing in Alzheimer's Disease
Published in Neuroscience, Cell & Molecular Biology, and General & Internal Medicine
Neuronal organoids to study patient heterogeneity and personalized in vitro drug testing in Alzheimer's Disease
Cristina Zivko, PhD1,2, Vasiliki Mahairaki, PhD1,2
1Department of Genetic Medicine, Johns Hopkins School of Medicine, 21205 Baltimore, MD, USA
2The Richman Family Precision Medicine Center of Excellence in Alzheimer’s Disease, Johns Hopkins School of Medicine, 21205 Baltimore, MD, USA
Alzheimer's Disease (AD) is a devastating neurodegenerative disorder. It affects more than 55 million people around the globe, with the number of patients projected to more than double in the next 25 years[1]. We don’t really understand how exactly someone develops AD, with its onset, progression and specific clinical manifestations varying substantially across people.
There is no cure for AD: at best we can only modestly slow its inevitable progression. Moreover, patients respond differently to available or candidate treatments, with some not benefitting at all. Research into novel therapies for AD has had an astonishing 98% failure rate in the past 20 years[2]. Treatments that seem to work well enough to justify clinical trials, often do not translate into positive outcomes for patients.
But there’s hope: what if pre-clinical models could account for patient variability? What if we could evaluate drug efficacy by testing on a platform developed from the patients themselves?
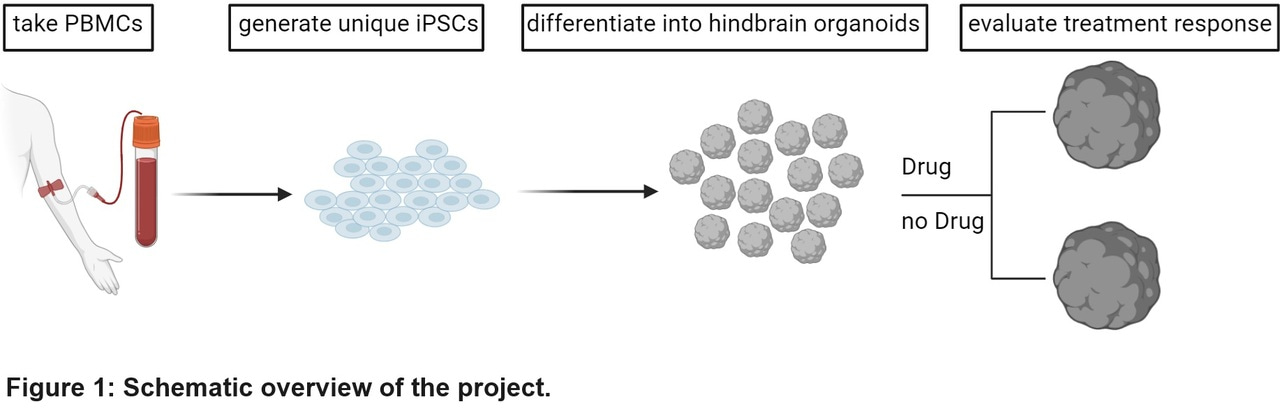
Our precision medicine approach specifically targets that goal. Healthy individuals and different AD patients were recruited to our study. We reprogrammed their peripheral blood monocytes and generated their human induced pluripotent stem cells (iPSCs). We then checked for the presence of pluripotency markers (TRA-1-60, OCT4, NANOG) and performed spontaneous differentiation assays to confirm the iPSCs can differentiate into all three germ lineages (endoderm, ectoderm, mesoderm).
Then we used the iPSC lines of several individuals to generate their 3D hindbrain organoids containing serotonin-producing neurons (5-HT-organoids). We were interested in the functioning of the serotonergic system as predictor of the development of neuropsychiatric symptoms (NPS) for some patients but not others. We were also looking for a way to evaluate in vitro the effects of the FDA approved antidepressant escitalopram, a selective serotonin reuptake inhibitor.
We derived 5-HT-organoids from the iPSCs of 3 healthy individuals and 3 AD patients, two of who were additionally diagnosed with NPS (Fig. 1). All experiments were performed three separate times to ensure reproducibility and robustness. Growing from iPSCs into 5-HT-organoids, cells lost pluripotency markers associated with stem cells (e.g., TRA-1-60, Oct4), while initially gaining markers associated with neuronal precursor cells (NKX2.2, Nestin) then markers associated with neurons (LMX1b, TUJ1), finally with serotonin-producing neurons specifically (TPH2, 5-HT). These we evaluated with quantitative real time PCR, flow cytometry, and immunocytochemistry imaged by confocal fluorescent microscopy. We then developed a python code in-house to measure the growth and circularity of hundreds of organoids over the course of 42 days [4], all consistent with each other across biologically independent triplicates, for the 6 different individuals. All these organoids released serotonin into the culture supernatant at levels that we could detect by enzyme-linked immunosorbent assay, and within a similar quantitative range.
Having confidently established the reliability of our platform, we moved to drug testing. Escitalopram specifically inhibits the reuptake of serotonin by the pre-synaptic neurons that release it, leaving more of it available to post-synaptic neurons[5].
One hour of exposure to between 10 and 100 µM of the drug on average led to a dose dependent increase in serotonin concentration in the supernatant of these iPSC-derived organoids. This is comforting news: the drug worked as hypothesized (and previously reported) on the target cells.
Importantly, measurement of escitalopram effects at the individual level paints a more complicated, albeit more interesting picture. The organoids originating from one of the healthy individuals’ blood and those originating from one of the AD+NPS patients did not show an increase of serotonin in the supernatant regardless of drug dose used in vitro. We consider these to be non-responders to escitalopram.
This really goes to the heart of precision medicine, and why it is so important. From our work: “The different responses to escitalopram we measured in hindbrain organoids originating from different individuals suggest that this in vitro method might identify subgroups of AD patients who are more likely to respond to therapy with escitalopram in vivo. We propose that patient-specific hindbrain organoids can be effectively used for drug screening purposes to predict pharmacological benefits. At a minimum these methods can help us understand the heterogeneity of response to treatment.”
References:
[1] GBD 2019 Dementia Forecasting Collaborators. Estimation of the global prevalence of dementia in 2019 and forecasted prevalence in 2050: an analysis for the Global Burden of Disease Study 2019. Lancet Public Health. 2022 Feb;7(2):e105-e125. doi: 10.1016/S2468-2667(21)00249-8.
[2] Kim CK, Lee YR, Ong L, Gold M, Kalali A, Sarkar J. Alzheimer's Disease: Key Insights from Two Decades of Clinical Trial Failures. J Alzheimers Dis. 2022;87(1):83-100. doi: 10.3233/JAD-215699.
[3] Sagar R, Zivko C, Xydia A, Weisman DC, Lyketsos CG, Mahairaki V. Generation and Characterization of a Human-Derived and Induced Pluripotent Stem Cell (iPSC) Line from an Alzheimer's Disease Patient with Neuropsychiatric Symptoms. Biomedicines. 2023 Dec 15;11(12):3313. doi: 10.3390/biomedicines11123313.
[4] Lopez Montes A. github.com/alopezmontes/organoid_segmentation. 2024.
[5] Bræstrup C, Sanchez C. Escitalopram: a unique mechanism of action. Int J Psychiatry Clin Pract. 2004;8 Suppl 1:11-3. doi: 10.1080/13651500410005496.
Follow the Topic
-
Molecular Psychiatry
This journal publishes work aimed at elucidating biological mechanisms underlying psychiatric disorders and their treatment, with emphasis on studies at the interface of pre-clinical and clinical research.






Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in