No Standard Path: How Research Results Are Reported Back—and Why It Matters
Published in Research Data, Sustainability, and Biomedical Research
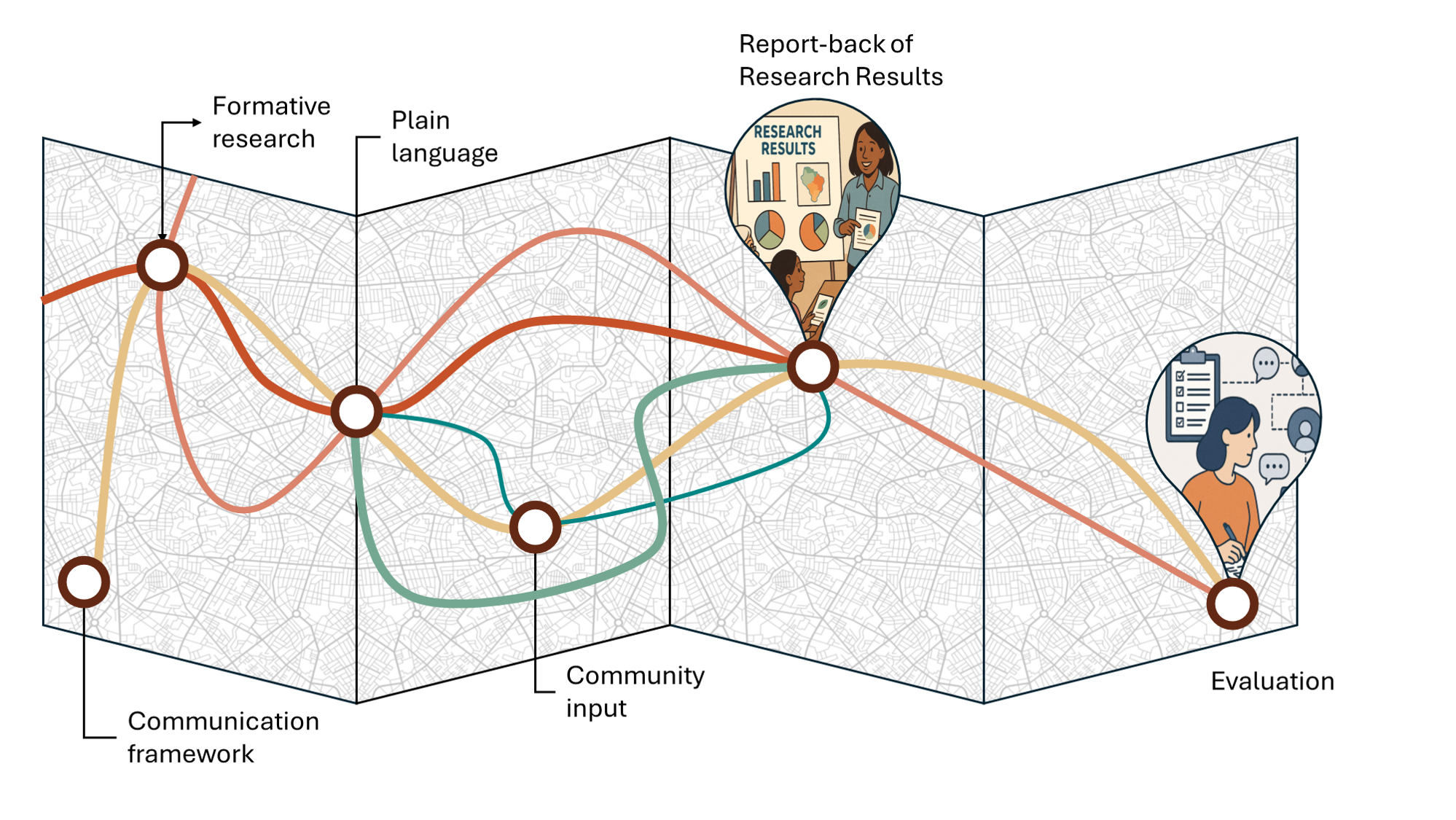
The return of research results to study participants—often referred to as reporting back research results (RBRR)—currently occupies an uneasy space in environmental health research. While ethical frameworks increasingly emphasize transparency, respect, and community engagement, practical guidance on how to appropriately report back research results (RBRR) has lagged behind. Our study, “Strategies to Facilitate the Report‑Back of Research Results: A Systematic Review of Methods and Evaluations,” was designed to directly address this gap. We hope it will be an important contribution to the field.
We were inspired by a 2009 paper, which evaluated the ethical foundation for RBRR.1 That paper focused on addressing the question of should RBRR be conducted. We wanted to investigate the ethical underpinnings to inform how RBRR should be done. To do that, we had two main questions: first, how were people developing RBRR materials? Second, were these RBRR strategies aligned with bioethical principles?
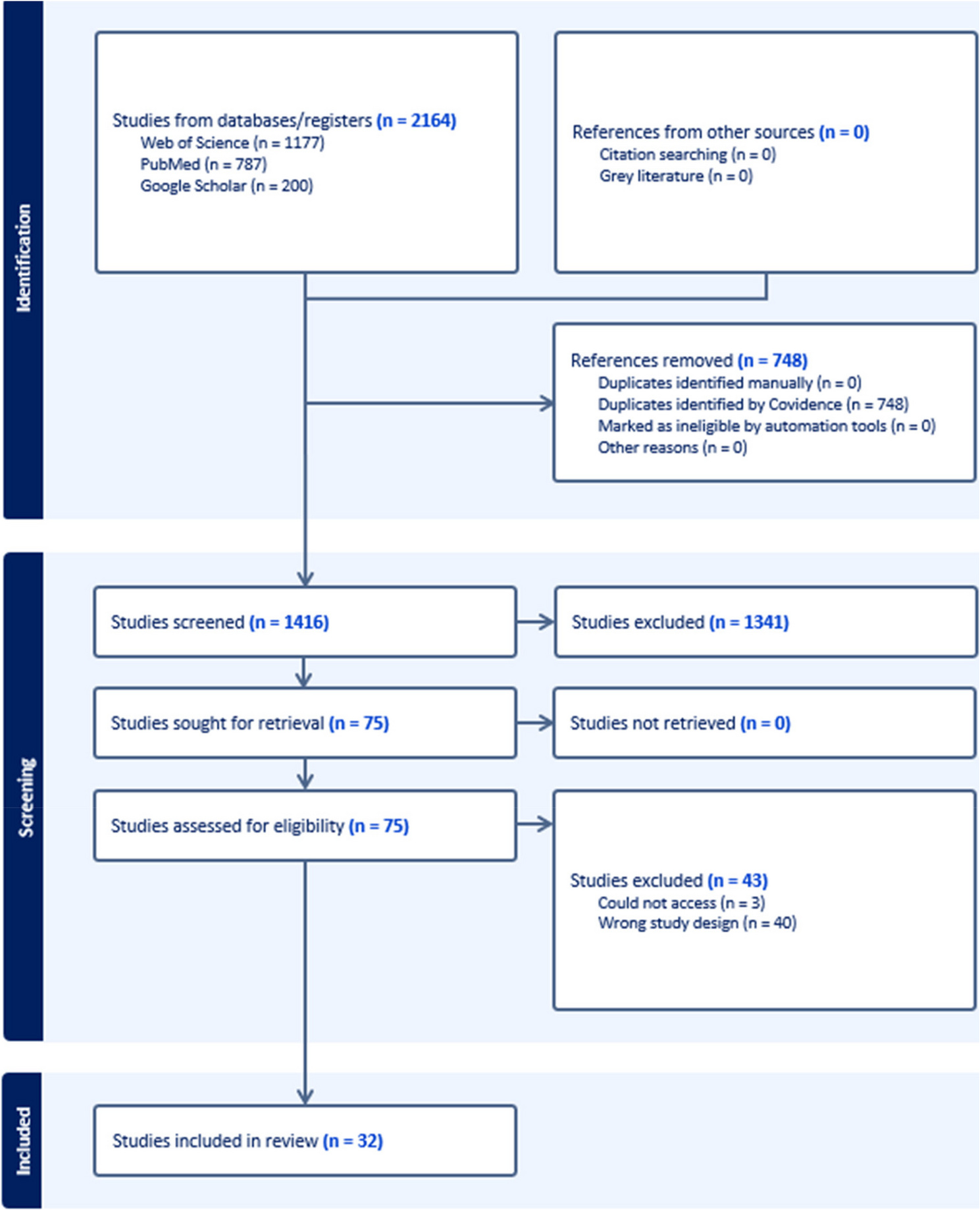
Drawing on a systematic review of 32 peer‑reviewed studies published between 2016 and 2024 our paper demonstrates that RBRR is increasingly viewed as necessary and important. Across environmental health, biomonitoring, and genomics research, RBRR is being framed as an ethical obligation—one tied to respect for participant autonomy, non-maleficence, beneficence, and justice, the four principles of bioethics. Participants are no longer seen merely as data sources, but as individuals with a legitimate claim to information generated through their contributions.
We focused our first analysis on the methods used to develop, and then evaluate, RBRR materials. Unfortunately, we found that RBRR strategies varied widely, with limited consistency in terms of cataloguing RBRR development, dissemination, and evaluation. While many studies emphasize the importance of plain language, multimodal dissemination, and tailoring materials to participant populations, only a minority clearly described how report‑back materials were developed. Even fewer applied formal communication, educational or ethical frameworks. Evaluation practices, though common, varied widely in timing, metrics, and rigor, making it difficult to assess whether RBRR efforts achieved their goals.
We then applied the bioethical framework to the RBRR practices, looking to see how bioethics may inform RBRR development and evaluation. Given the inconsistency in methodologies, this was difficult.
This inconsistency is precisely what makes the study so timely. As RBRR becomes an expected component of ethical and responsible research—particularly in community‑engaged and health disparities contexts—the absence of standardized guidelines poses practical and ethical risks. Without shared principles or benchmarks, well‑intentioned researchers may unintentionally undermine participant understanding, exacerbate uncertainty, or miss opportunities to support meaningful engagement. Our review suggests that flexibility and context‑specific tailoring are strengths of current practice but argues that these strengths should not come at the expense of transparency, rigor, or ethical accountability.
Our paper shifts the conversation from whether results should be returned to how results can be responsibly returned. It highlights reporting back not just as a dissemination task, but as a form of ethical practice that is shaped by design choices, communication strategies and evaluation priorities. By synthesizing what is currently known and, just as critically, what remains undocumented, the review provides a foundation for developing standardized yet adaptable guidelines for RBRR.
The paper’s findings are relevant to researchers, institutional review boards, funders and bioethicists grappling with how to operationalize ethical commitments in real-world research settings. As expectations around transparency and participant engagement continue to grow, this review offers both a clear assessment of the current landscape, recommendations for future work and a compelling call to action: to move from ad hoc approaches toward thoughtfully designed, ethically grounded, and consistently evaluated practices for reporting back research results.
References
- Morello-Frosch, R., Brody, J.G., Brown, P., Altman, R.G., Rudel, R.A. and Pérez, C., 2009. Toxic ignorance and right-to-know in biomonitoring results communication: a survey of scientists and study participants. Environmental Health, 8(1), p.6.
Follow the Topic
-
Environmental Health
This journal publishes manuscripts on important aspects of environmental and occupational medicine and related studies in toxicology and epidemiology that elucidate the human health implications of exposures to environmental hazards.




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in