Ozone pollution mitigation strategy informed by long-term trends of atmospheric oxidation capacity
Published in Chemistry
During past decades China experienced rapid industrialization, which brought economic advantages, but also caused significantly deteriorated air quality. According to WHO guidelines, ozone (O3) and fine particulate matter (PM2.5) are the chief hazards of China's air pollution. Over the past decade, China launched the toughest-ever clean air policy in the country. These efforts, by reducing emissions of primary pollutants such as volatile organic compounds (VOCs), nitrogen oxides (NOX) and others, improved PM2.5 air quality. However, O3 continued to increase. This contrasts with the generally decreasing O3 trend in Europe and North America due to continuous reductions of VOCs and NOX emissions since the 1990s. The causes of the contrasting trends and adverse O3 pollution trends in China are still not well understood, representing a grand challenge for O3 pollution control.
This study is based on August measurements in Beijing from 2006 to 2020, which is the most comprehensive, long-term observational data set of O3 and its precursors VOCs and NOX that is available in China. Using the dataset, we show that O3 concentrations increased before 2014, and then decreased in the following years (Figure 1B). By removing the impact of meteorological variability on O3, we obtain a normalized O3, which still shows a non-monotonic trend (Figure 1D). The OH reactivity of VOCs (VOCR) and NOX (NOXR) show consistently decreasing trends (Figure 1A), confirming the effects of emission controls. The trend of the ratio between VOCR and NOXR (VOCR/NOXR) is similar to that of normalized O3; both remained approximately constant before 2014, and then decreased rapidly after 2014 (Figure 1C).
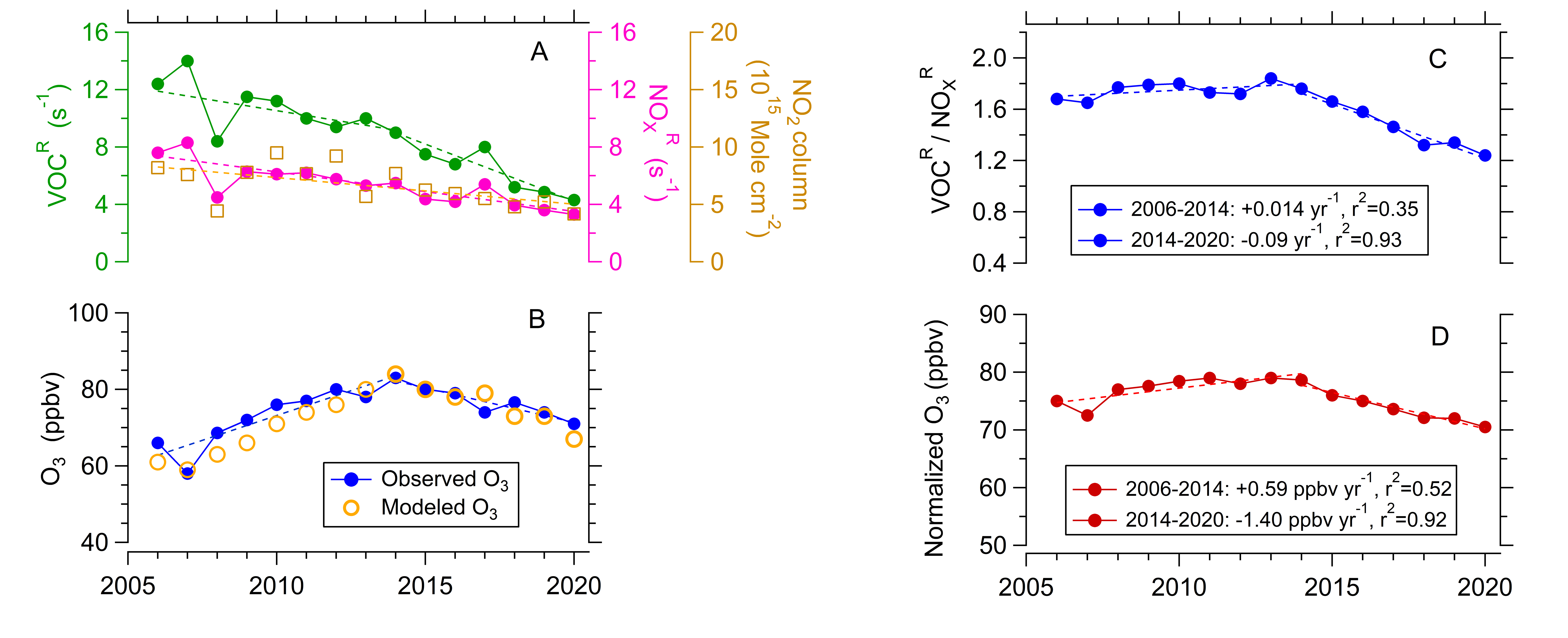
Figure 1. Variations in ozone and OH reactivity of precursors derived from observations in Beijing, August between 2006 and 2020. Variations in daytime (6:00-19:00) averages of VOCR (including primary hydrocarbons plus carbon monoxide), NOXR and column concentrations of NO2 (A), MDA8 O3 concentrations (B), the ratio of VOCR to NOXR (C), and normalized MDA8 O3 concentrations (D). Dashed lines show linear regression fits to observations in periods before and after 2014. Model-simulated MDA8 O3 concentrations are included in (B).
To gain a deeper insight, we investigated the detailed radical cycling and photochemistry by applying an advanced box model based on the updated Master Chemical Mechanism v3.3 chemistry scheme (MCM 3.3). The box model simulation shows that OH increased continuously during 2006-2020, giving an approximate doubling (Figure 2A). The main drivers of the hydroxyl radical (OH) change are the increase of photolysis frequencies, decrease in NOX emissions, decrease in VOC emissions, and reduced heterogeneous uptake (Figure 2B). The OH turnover rate induced by VOCs ([OH]×VOCR) shows non-monotonic rends, which is consistent with the trend of observed O3 concentrations (Figure 2C). This demonstrates that the change of ambient O3 over the entire 2006 to 2020 period is consistent with our understanding of O3 photochemistry and OH cycling. Further analysis suggests that VOCR/NOXR affects the O3 trend primarily through changing the primary production of atmospheric radicals, rather than changing the radicals’ chain length.
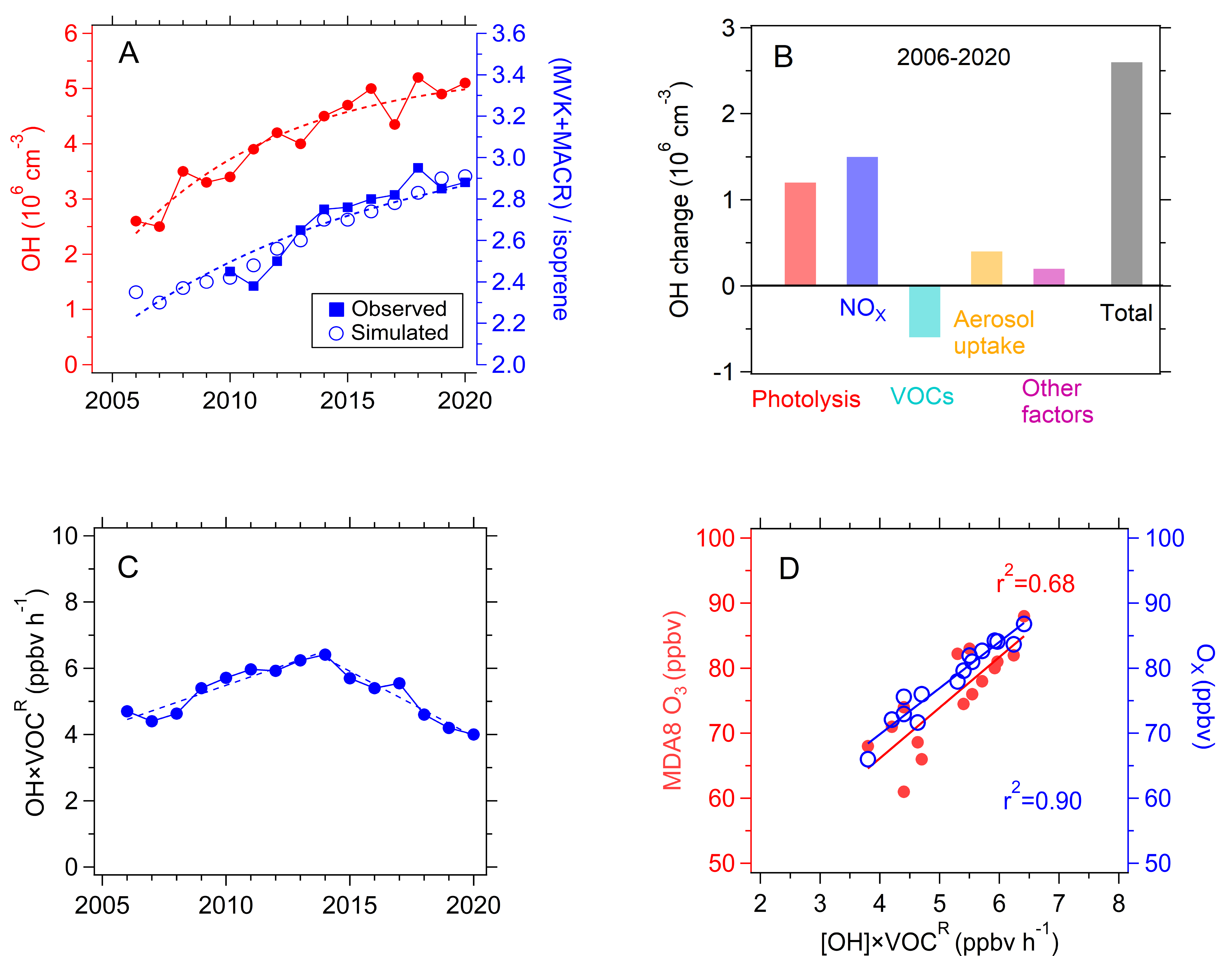
Figure 2. Variations in model-simulated OH concentrations and its driving forces in Beijing. (A) Variations in simulated daytime average OH concentrations (red dots) and measured and simulated (MVK+MACR)/isoprene ratios (blue dots and circles) in Beijing in August between 2006 and 2020. Dashed lines show exponential regression fits to trends. (B) Apportionment of the causes of the change in OH derived from box model sensitivity analysis. (C) Variations in OH turnover rate induced by VOCs (OH×VOCR). Dashed lines show linear regression fits to trends in periods before and after 2014. (D) Correlation of OH×VOCR with observed MDA8 O3 and daytime OX concentrations.
To propose a precise O3 mitigation strategy, we simulate the combined effect of VOCs and NOX on normalized O3 and total OH turnover rate induced by all reactive gases (including VOCs, NOX and SO2) (Figure 3). The Beijing environment during 2006-2020 was overall in the VOC-limited regime for both O3 and total OH turnover rate, with a trend of moving toward to the transition regime. Figure 3 clearly shows that the turning point of the O3 trend appears in 2014 due to a faster decrease in VOCR/NOXR after 2014 than before 2014. Future projection scenarios show that reactive VOCs abatement should receive near-future priority for O3 pollution control in China, followed by further NOX controls.
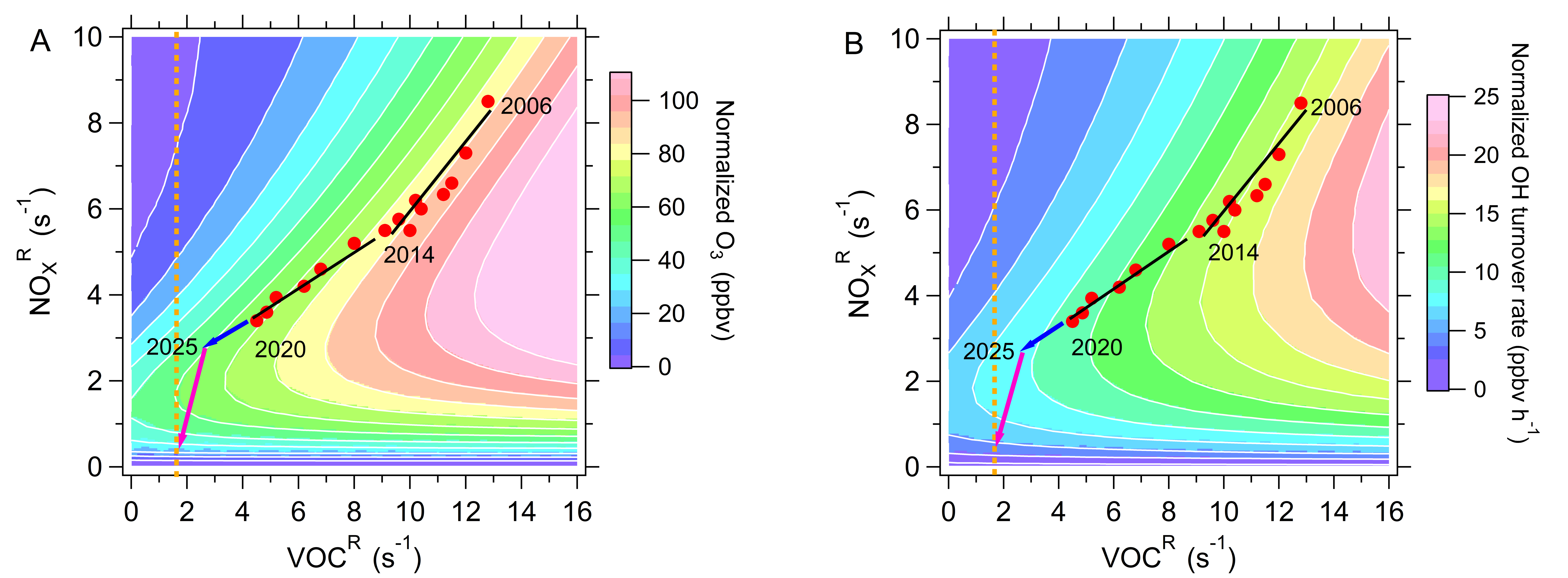
Figure 3. Isopleth plots for (A) ozone and (B) total OH turnover rate as a function of NOXR and VOCR. Red solid circles represent the average levels of MDA8 O3 and daytime OH turnover rate for each year during 2006-2020, and the solid lines indicate fits to the trends during 2006-2014 and 2014-2020. The yellow dotted lines indicate the VOCR level from biogenic emissions. The blue and magenta arrows indicate future routes for VOCs and NOX abatement that mitigate ozone effectively in the short term (2020-2025) and long term (after 2025), respectively.
In summary, we find that the different O3 trends can be well explained and predicted by a novel parameter, the reactivity ratio of VOCs to NOX. Our results show that reducing the reactivity ratio is the most effective way to control O3 pollution in China in the near future. With this approach, optimal emission reduction roadmaps can be derived directly from measurements of reactivity ratios, avoiding uncertainties of models and emission inventories.
For more detailed information, see our article “Ozone pollution mitigation strategy informed by long-term trends of atmospheric oxidation capacity” in Nature Geoscience.
Follow the Topic
-
Nature Geoscience
A monthly multi-disciplinary journal aimed at bringing together top-quality research across the entire spectrum of the Earth Sciences along with relevant work in related areas.
Related Collections
With Collections, you can get published faster and increase your visibility.
Urban fires around the globe
Publishing Model: Hybrid
Deadline: May 31, 2026
Atmospheric rivers
Publishing Model: Hybrid
Deadline: May 31, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in