PIEZO1 mechanically regulates the antitumour cytotoxicity of T lymphocytes
Published in Cancer, Cell & Molecular Biology, and Anatomy & Physiology
The landscape of oncoimmunology and cancer treatment has fundamentally reshaped by the discovery of immune checkpoint blockade (ICB). However, the key premise of successful ICB-targeted therapies is to secure effective desired immune activities throughout the treatment1, which still remains challenging. Joining the collective efforts in finding new strategies to boost T cell killing function, we sought an unconventional approach by tapping into the mechanical regulation of T cells since increasing evidence is suggesting that immune cells tailor their immune response to dynamic biomechanical environments2. Cytotoxic T cells adjust the killing function according to the mechanical properties of the target3-5. We wondered whether PIEZO1, a critical cell mechanical sensor and regulator6, 7, plays a central role in this process.
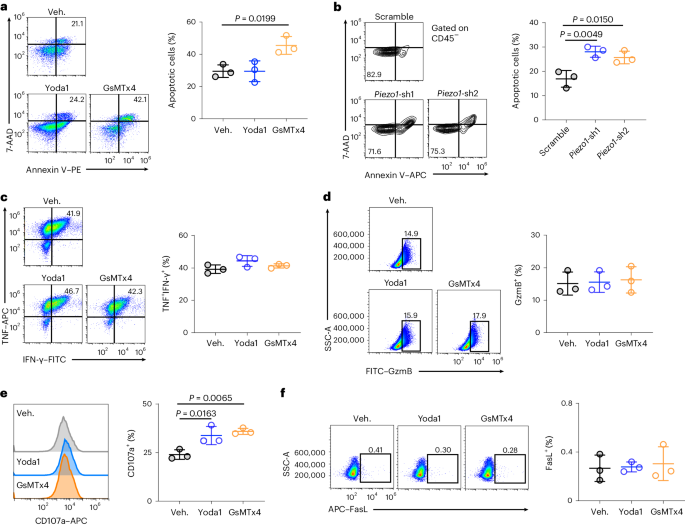
We firstly tracked the spatial and temporal dynamics of PIEZO1 in the CD8+ T cells upon activation. It turned out that PIEZO1 is upregulated promptly during activation, which continues when the cells become exhausted. Surprisingly, reversing this process by either chemically targeting or genetically knocking down PIEZO1, could effectively enhanced the cell toxicity when co-culturing T cells and tumour cells, without affecting the secretion of IFN-γ and TNF or the production of granzyme B and perforin. To pin down the drive force of this phenomenon, we checked the traction force exerted by T cells, which is critical for ramping up these immune attacks, by using traction force microscopy (TFM). Not only in the in vitro cell model but also the ex vivo tumour mice models, the traction force of T cells upon PIEZO1 blockade indeed significantly increased as revealed in fluorescence activated cell sorting (FACS) and TFM. Meanwhile, more T cells infiltrated into the tumour in response to PIEZO1 blockade. Accordingly, the tumour growth was hampered and mouse survival was considerably prolonged. This prompted us to consider the possibility of targeting PIEZO1 to facilitate ICB therapy. We were quite excited to see that greater therapeutic outcome was achieved when combining the PIEZO1 antagonist with anti-PD-1 antibody regiment, suggesting PIEZO1 as promising target for improving the ICB-based immunotherapies.
However, the question still remained as to how the dynamics in PIEZO1 activity orchestrated the traction fore and the cytotoxicity of T cells. We zoomed into the transcriptional shifts sensitive to PIEZO1 activity, from which a transcriptional factor GRHL3 stood out. Among the candidate genes subjective to GRHL3’s regulation listed from the chromatin immunoprecipitation (ChIP)-qPCR search, we narrowed down to RNF114, an E3 ubiquitin ligase which was downregulated upon PIEZO1 inhibition. Using the super resolution microscopy of direct stochastic optical reconstruction (dSTORM) or stimulated emission depletion (STED), we confirmed that RNF114 indeed attached to actin filament, and regulated the traction force by shaping the level as well as the arrangement of actin (Fig. 1). Moreover, compensating the loss of GRHL3 or RNF114 due to PIEZO1 disruption by overexpression effectively reversed the impact on actin filament and the traction force. These results helped establish PIEZO1-GRHL3-RNF114 as a critical function axis responsible for regulating the cytoskeleton architecture and traction force in cytotoxic T cells.
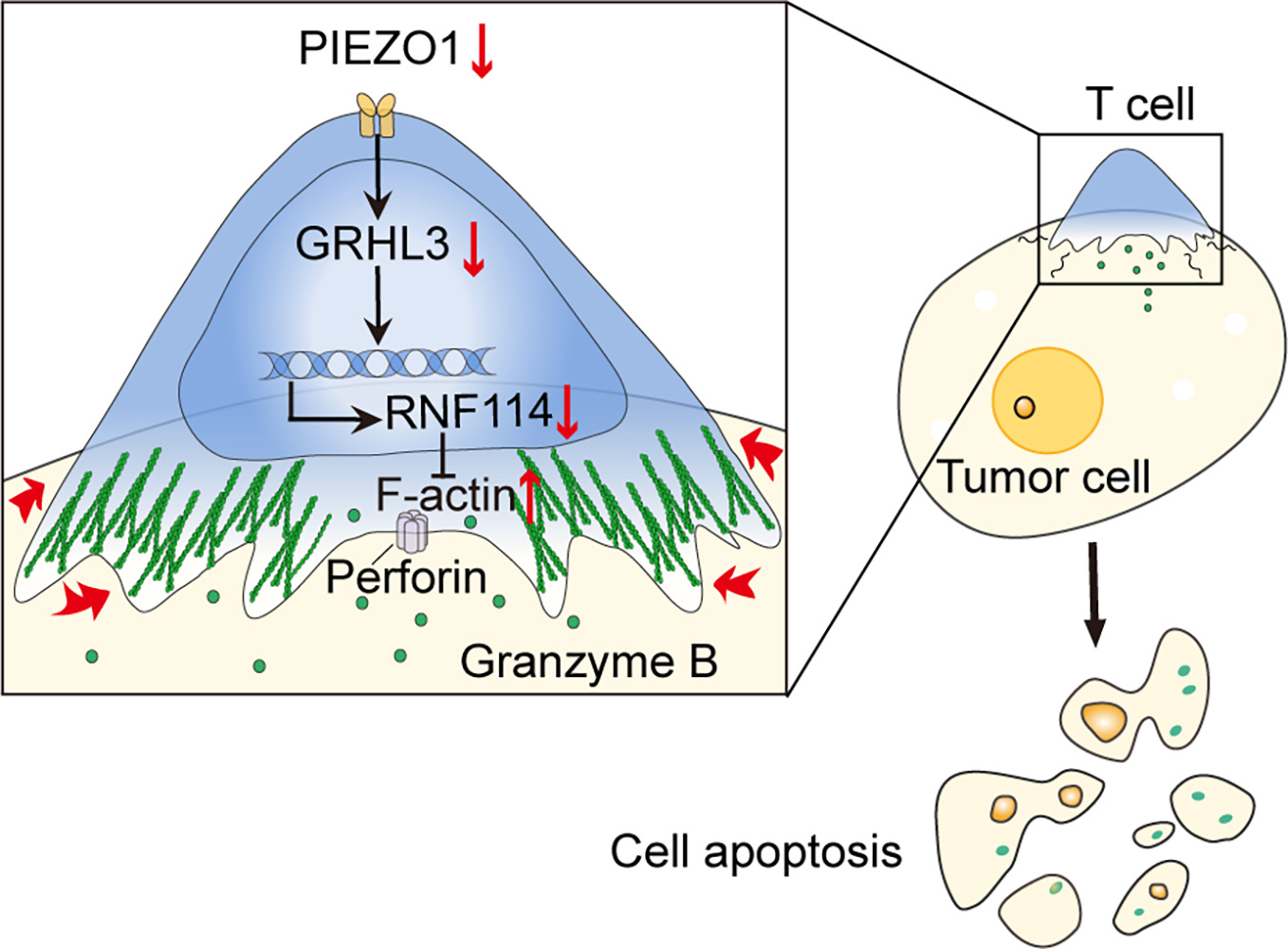
But we were not sure how these findings could be translated into the clinic settings. The first clue came from the observation that the expressions of PIEZO1, GRHL3, and/or RNF114 were notably increased in the CD8+ T cells collected from the tumours in patients with colorectal cancer, stomach adenocarcinoma or hepatocellular carcinoma, compared with the healthy counterparts. Blocking PIEZO1 suppressed both GRHL3 and RNF114, resulted in the increased actin filaments and enhanced traction force, consistent with the above results. More importantly, by using Kaplan-Meier plotter and TIMER2.0 dataset, we found that either GRHL3 or RNF114 level was negatively correlated to CD8+ T cells infiltration and the survival of patients.
In summary, our findings expand the regulatory role of PIEZO1 to the dynamic adaptation of T cell-mediated immune response to mechanical cues. This work also helps propose the cellular traction force can be served as a prognosis indicator of T cell immunotherapy.
In near future, we would like to investigate the role of PIEZO1 mediated mechanotransduction in autoimmune diseases, and understand further the mechanical characteristics of the formation and regulation of T cell memory.
Reference
- Topalian, S.L., Taube, J.M., Anders, R.A. & Pardoll, D.M. Mechanism-driven biomarkers to guide immune checkpoint blockade in cancer therapy. Nat Rev Cancer 16, 275-287 (2016).
- Huse, M. Mechanical forces in the immune system. Nat Rev Immunol 17, 679-690 (2017).
- Tello-Lafoz, M. et al. Cytotoxic lymphocytes target characteristic biophysical vulnerabilities in cancer. Immunity 54, 1037-1054.e1037 (2021).
- Liu, Y. et al. Cell Softness Prevents Cytolytic T-cell Killing of Tumor-Repopulating Cells. Cancer Res 81, 476-488 (2021).
- Lei, K. et al. Cancer-cell stiffening via cholesterol depletion enhances adoptive T-cell immunotherapy. Nat Biomed Eng 5, 1411-1425 (2021).
- Mukhopadhyay, A. et al. trans-Endothelial neutrophil migration activates bactericidal function via Piezo1 mechanosensing. Immunity 57, 52-67.e10 (2024).
- Solis, A.G. et al. Mechanosensation of cyclical force by PIEZO1 is essential for innate immunity. Nature 573, 69-74 (2019).
Follow the Topic
-
Nature Biomedical Engineering
This journal aspires to become the most prominent publishing venue in biomedical engineering by bringing together the most important advances in the discipline, enhancing their visibility, and providing overviews of the state of the art in each field.
Related Collections
With Collections, you can get published faster and increase your visibility.
Implantable wireless communication technologies
Publishing Model: Hybrid
Deadline: Nov 28, 2026
Medical Ultrasound: Emerging Techniques and Applications
Publishing Model: Hybrid
Deadline: Jan 29, 2027




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in