Proteomic analysis of COVID-19-associated Acute Kidney Injury is significantly driven by markers of hemodynamic instability, tubular injury and myocardial damage
Published in Healthcare & Nursing
COVID-19 has a variety of multi systemic adverse outcomes
We know that COVID-19, the clinical syndrome resulting from SARS-CoV-2 infection is broad, ranging from asymptomatic infection to severe disease with extra-pulmonary manifestations3, including acute kidney injury4, acute myocardial injury5,6 and thrombotic complications7,8,9,10,11.
Kidney function decline in hospitalized COVID-19 patients
As increased COVID-19 severity surged through the hospitals, clinicians began to observe increasing incidences of kidney injury in many of the hospitalized patients. AKI was then recognized as a common complication of COVID-19 in the hospitalized setting and increased the risk of in-hospital morbidity and mortality19. While the rates of AKI have decreased with the progression of more non-fatal, milder COVID-19 "waves" since the initial surge in 2020, the incidence of AKI still remains high17 among the hospitalized. In addition to morbidity and mortality in the acute setting, COVID-19 is also associated with long term manifestations i.e., the post-acute sequelae of SARS-CoV2 (PASC)25. Kidney function decline is a major component of PASC and a study of >1 million individuals found that survivors of COVID-19 had an elevated risk of post-acute eGFR decline26, suggesting long term kidney dysfunction may occur following the acute infection.
Understanding of the pathophysiology of COVID-19-associated AKI
There is yet, a limited understanding of the molecular mechanisms behind the incidence and progression of AKI in COVID-19. A slew of recent research works have attempted to understand this pathophysiology via multi-'omic' approaches using postmortem kidney samples or histopathological reports. While some reports identified common inflammatory pathways and regulatory responses between COVID-19 AKI and sepsis-AKI, others indicated acute tubular injury21 as a hallmark mechanism in patients that died with COVID-19. In addition to these mechanisms, systemic effects of critical illness (hypovolemia, mechanical ventilation) and cardiac dysfunction have also been shown to contribute to COVID-19 AKI.
Using proteomic profiling to characterize the progression of AKI in COVID-19
Prior research using minimally invasive proteomics assays has shown the use of peripheral serum as a readily accessible source of proteins that accurately reflects the human disease state30,31,32,33. Proteomic biomarkers have previously shown success in predicting COVID-19 outcomes27,28,29. Therefore, to understand the yet, unclear underlying mechanism that drives AKI in hospitalized, COVID-19 patients who were still alive, we sought to characterize the proteomic changes associated with COVID associated AKI.
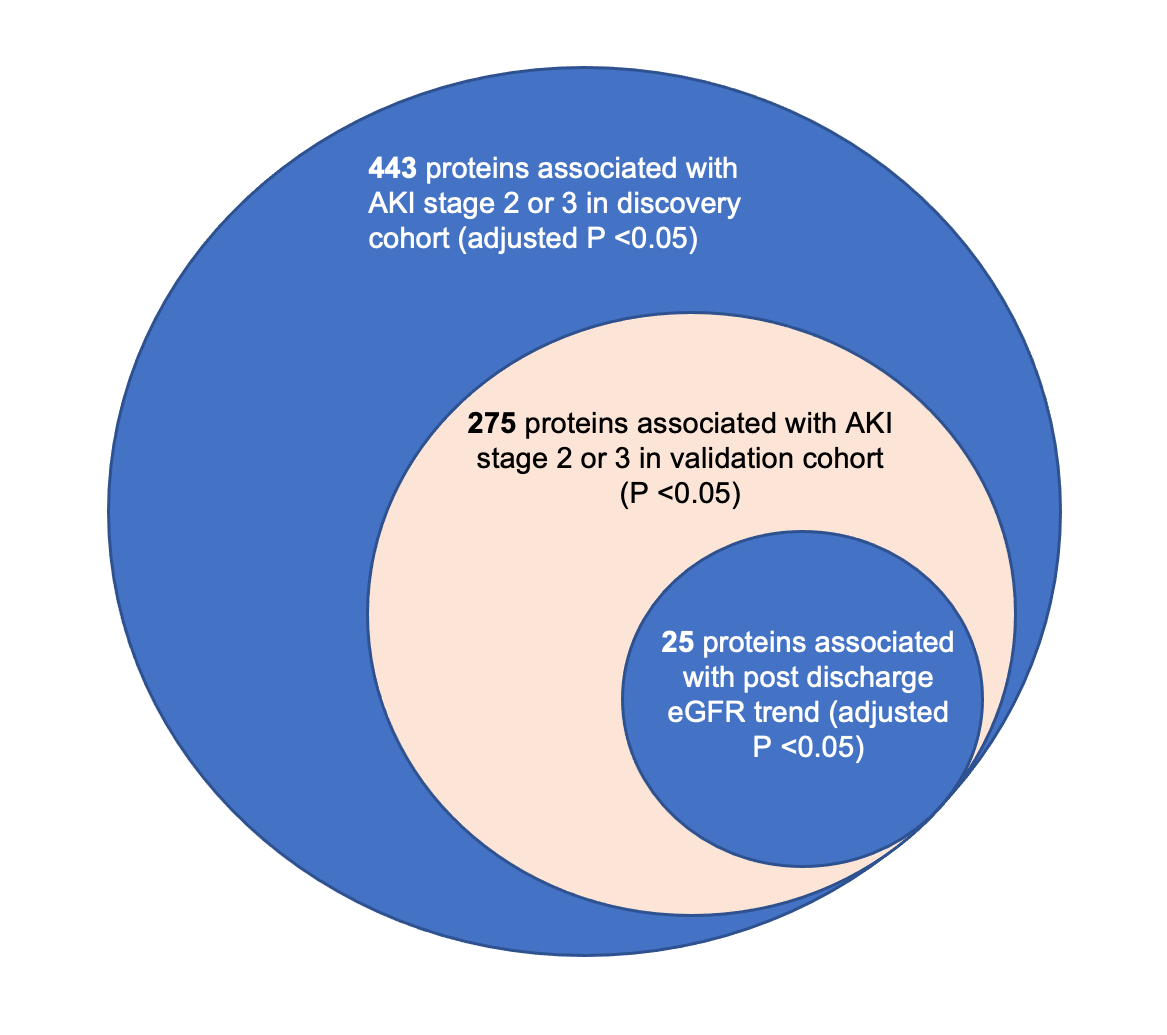
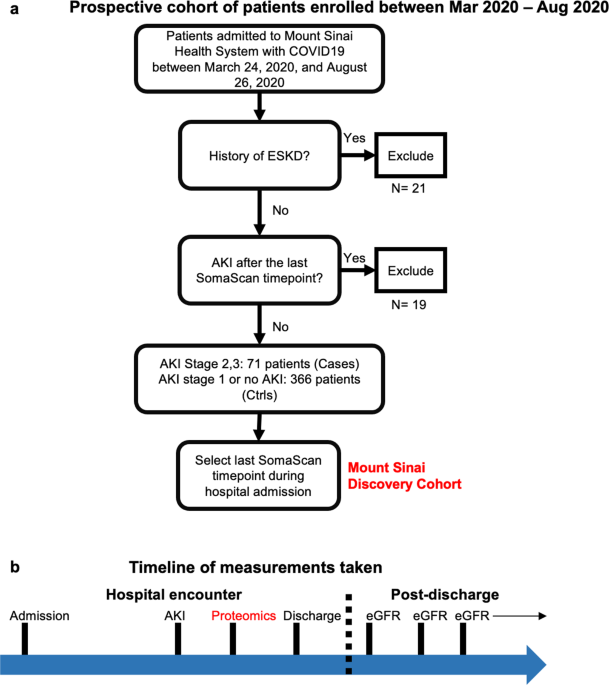
We measured protein expression of >4000 proteins from serum samples collected in a diverse large cohort of living, hospitalized patients with COVID-19 and validated significant results in an independent cohort.
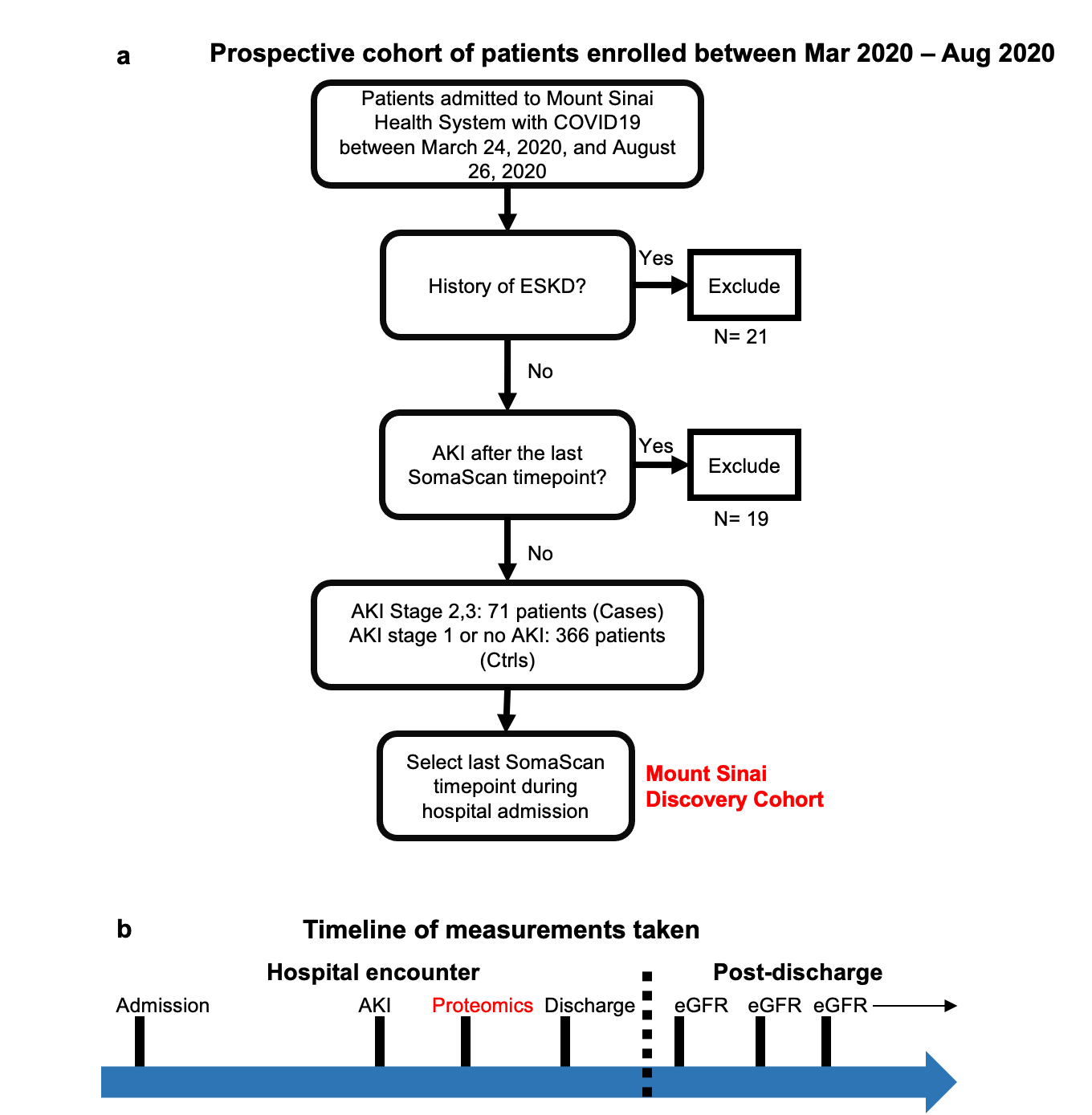
We identified proteins that were significantly different between patients with and without AKI. To determine whether these proteomic perturbations also characterize post-discharge kidney function decline we also correlated these markers with estimated glomerular filtration rate (eGFR) of returning patients post-discharge.
Our findings
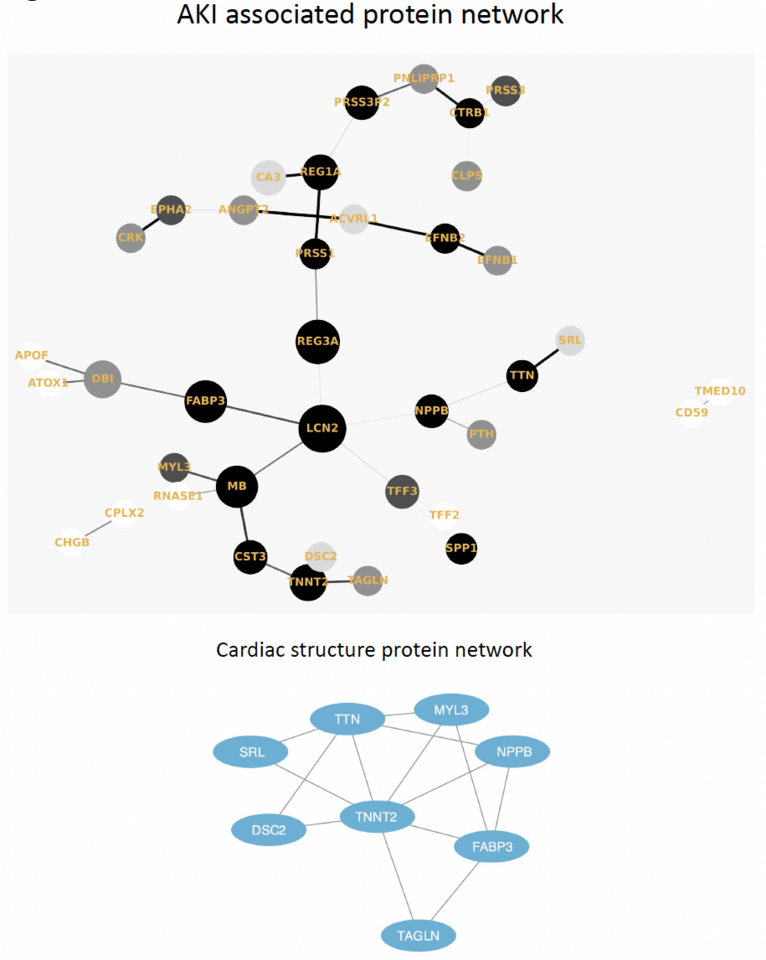
We found that our results pointed to the involvement of several mechanisms in responsible in the driving of AKI in COVID-19: tubular injury, neutrophil activation, and hemodynamic perturbation. Surprisingly, we also found that although almost half of the AKI-associated proteins were also significantly associated with post-discharge long-term eGFR decline signaling chronic kidney dysfunction.
Conclusions
Our study was not devoid of limitations and logistical challenges (due to being in the peak of the first, most severe surge of the COVID-19 pandemic) and necessitates follow-up studies in future to confirm our findings. However, our results shed significant light on the evidence of tubular damage in the peripheral blood as having a role to play in the decline of kidney function in the acute and long-term setting.
We hope that you read our paper to find more details on our findings and implications!
Follow the Topic
-
Communications Medicine

A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Reproductive Health
Publishing Model: Hybrid
Deadline: Mar 30, 2026
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in