Rapid and reagent-less bacterial identification by combining RNA-cleaving DNAzymes with electrochemistry
Published in Chemistry
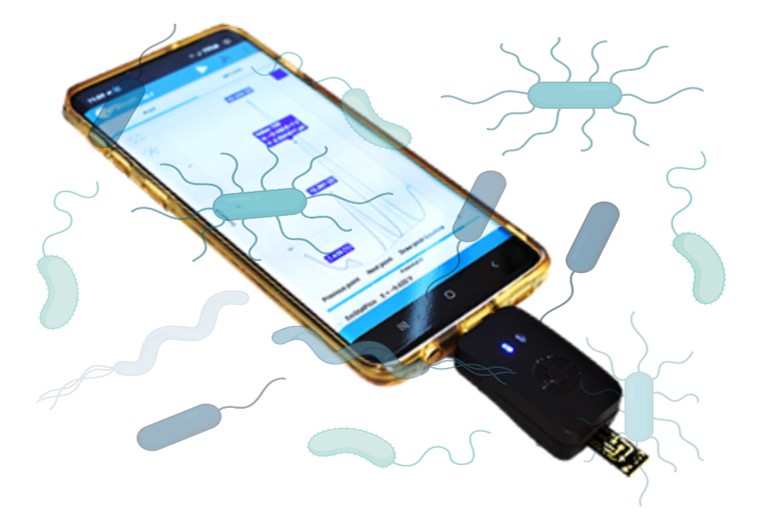
We developed a rapid test for the detection and identification of bacterial pathogens, which is urgently needed for the diagnosis and precision treatment of bacterial infections. The current gold standard for bacterial testing relies on lengthy growth cultures that lead to sample-to-result times in the order of days (>18 hours).
With the rise in the emergence of difficult-to-treat antibiotic resistant pathogens, rapid and precision diagnostics are a priority for clinicians. Such technologies can help clinicians prescribe effective and precise therapies when needed and avoid antibiotics when clinical symptoms are linked to other causative agents.
The test developed by our multidisciplinary team, with researchers from engineering, biochemistry, and clinical microbiology, combines advances in synthetic biology, functional hierarchical materials, and electrochemistry to deliver a unique solution for rapid sensing.
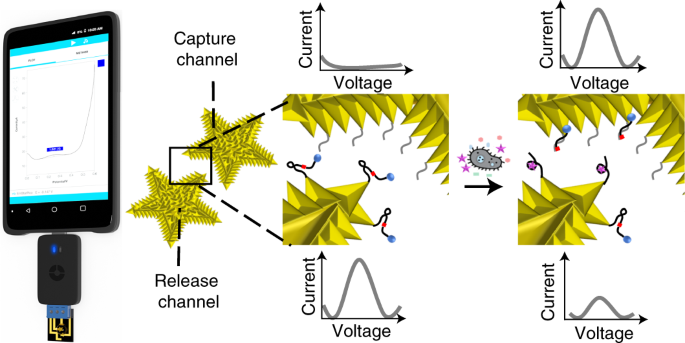
Central to this solution are RNA-cleaving DNAzymes (RCDs), a class of functional nucleic acids, that have been selected for precisely identifying specific bacterial species. Herein, we developed a new class of RCDs that could be integrated with electrical readout to take advantage of the ultra-sensitivity of the electrical sensing, thus eliminating the need for target enrichment through bacterial culture or nucleic acid amplification that represent key barriers to making rapid point-of-care pathogen testing a clinical reality.
The assay leverages on the programmability of RCDs to recognize specific bacterial targets and in response release an electroactive DNA barcode. This barcode diffuses to an electrochemical transducer, at which point it is measured by the generation of an electrochemical signal. The use of programmable molecules and inherent molecular processes enable the assay to operate without the addition of reagents or user intervention.
Together with a strong clinical team, we applied this assay to the rapid diagnosis of urinary tract infections (UTI), one of the top five healthcare-acquired infections with a yearly healthcare cost of $ 1 billion. This rapid UTI test, evaluated with more than 40 clinical samples, diagnoses UTIs caused by Escherichia coli with a sensitivity of 100% and specificity of 78% in less than one hour.
The assay developed herein allows us to eliminate all of the manual steps related to the addition of labeling and amplification reagents by getting the molecules, rather than the users, to do the work. Given the versatility of DNAzymes in targeting a wide range of analytes (small molecules, proteins, pathogens), we expect the new developments achieved here to reach beyond bacterial sensing and into universal molecular and cellular sensing.
Follow the Topic
-
Nature Chemistry
A monthly journal dedicated to publishing high-quality papers that describe the most significant and cutting-edge research in all areas of chemistry, reflecting the traditional core subjects of analytical, inorganic, organic and physical chemistry.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in