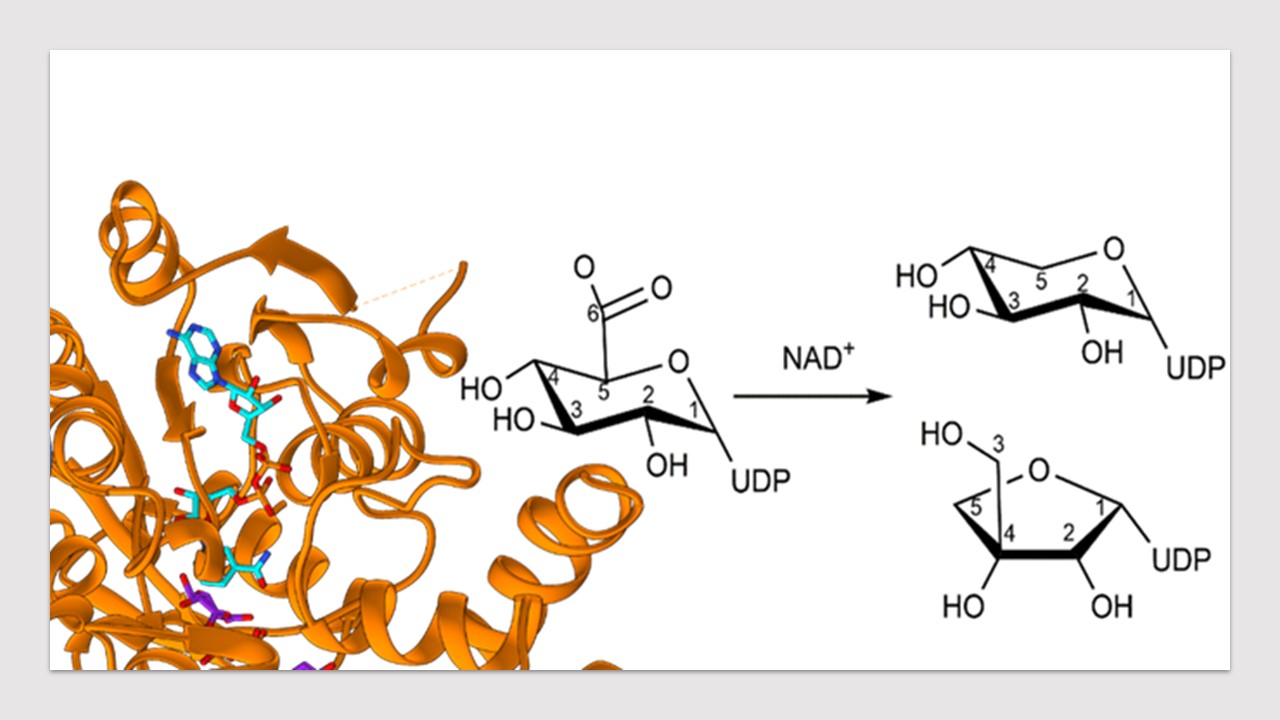
Apiose is a fascinating branched-chain sugar molecule that can form bonds with a tetracoordinated boron atom in the Rhamnogalacturonan-II complex present in the plant cell wall. This structure contributes to the overall mechanical resistance that characterises plant cells. The oxidoreductase responsible for producing the activated form of this sugar, UDP-apiose, also makes UDP-xylose at the same time. This enzyme, known as UAXS, is present in plants, algae and bacteria, and makes both products from UDP-glucuronic acid and using the redox potential of NAD+. While the ability of making UDP-xylose is shared with few more enzymes, UDP-apiose is known to be exclusively produced by UAXS. This is made possible by a tailored ring contraction reaction that is the signature of this enzyme.
In our study, the first step has been the determination of the three-dimensional structure of UAXS. Since the enzyme is extremely soluble (perfectly stable at 100 mg/ml), we performed screening of crystallisation conditions at high concentration. Puzzled by the lack of positive hits after thousands of single conditions tested, we performed small-angle X-ray scattering experiments to assess UAXS state at different concentrations. To our surprise, the enzyme showed a clear pattern, with the more concentrated samples displaying a lower apparent mass then the less concentrated ones (Figure 1).
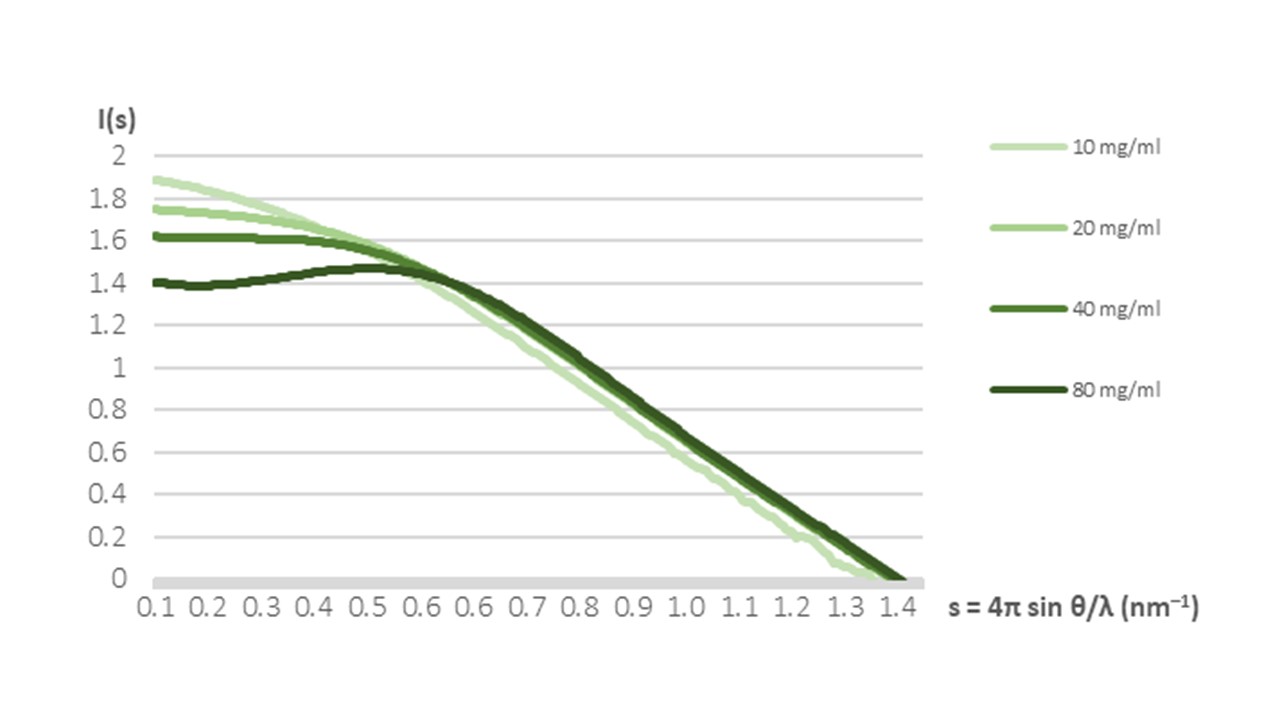
This quite unusual concentration-dependent intermolecular repulsion leads, from a crystallographic perspective, to a less favourable state for an ordered aggregation (crystallisation). Only by taking this into consideration we could tune our strategy and obtain protein crystals.
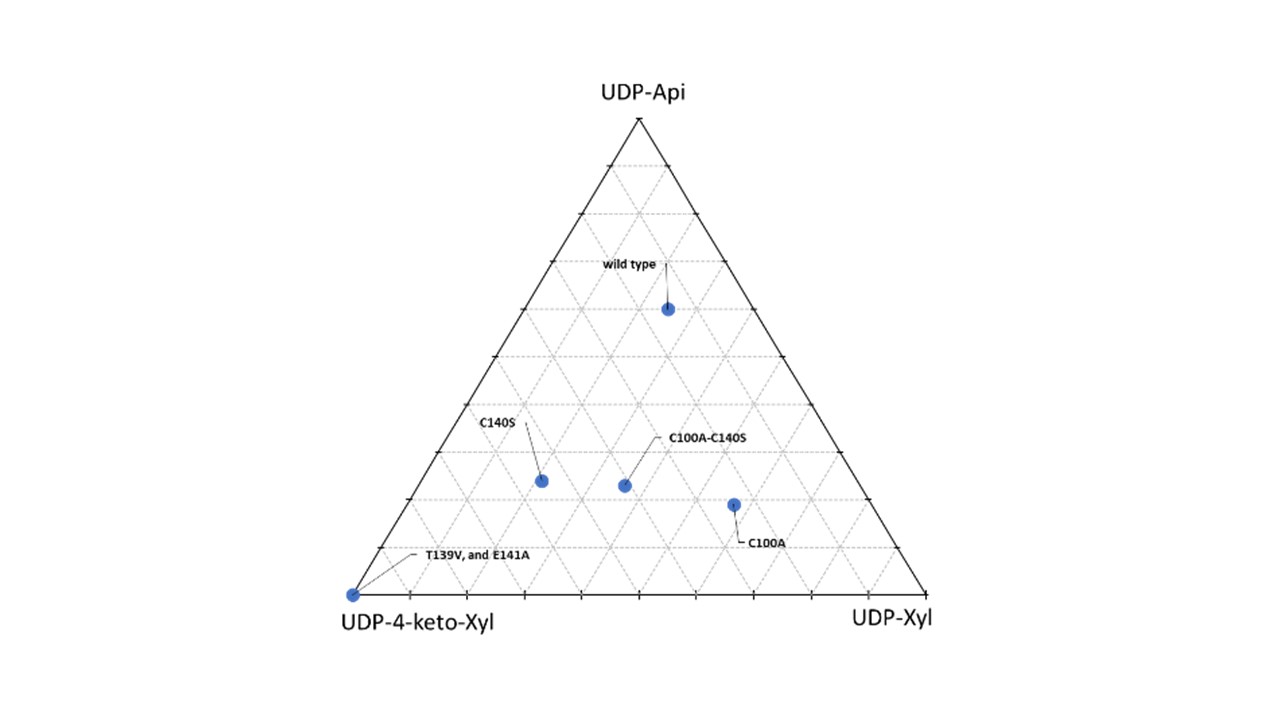
During our research we understood why and how UAXS is intrinsically incapable of producing UDP-apiose only. This is due to its mechanism, which inevitably leads to the formation of the by-product UDP-xylose. When we pushed the enzyme towards performing the ring contraction reaction only, we obtained mutants releasing a reaction intermediate or we reduced the amount of UDP-apiose produced, in favour of other products (Figure 2). While this can be disappointing from an applicative point of view, it is certainly an interesting evolutionary observation. The UDP-apiose synthase activity totally hijacked a pre-existing mechanism, deputed to UDP-xylose production, and pushed it as far as possible. In doing so, UAXS expands the mechanism landscape of the well-known class of short chain dehydrogenase/reductases.
In our work, we combined different approaches, from structural and biochemical assays to computational simulations, to build a final model for a reaction that puzzled researchers in the last 50 years.
Follow the Topic
-
Nature Catalysis
This journal brings together researchers from across all chemistry and related fields, publishing work on homogeneous catalysis, heterogeneous catalysis, and biocatalysts, incorporating both fundamental and applied studies.



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in