Reversal of the diabetic bone signature with anabolic therapies in mice
Published in Healthcare & Nursing
Explore the Research

Reversal of the diabetic bone signature with anabolic therapies in mice - Bone Research
Bone Research - Reversal of the diabetic bone signature with anabolic therapies in mice
https://www.nature.com/articles/s41413-023-00261-0
The problem
The prevalence of diabetes mellitus (DM) around the world is high with more than 500 million adults living with the disease (1 in 10, ages 20-79), and it is estimated to increase to 783 million by 2045.1 The American Diabetes Association declared DM a nationwide epidemic, being the 7th leading cause of death in the US. In 2019, ~11% of Americans had DM and ~50% of Americans 65 years and older had prediabetes. The disease impacts all tissues and organs, accounting for substantial morbidity and mortality. In 2021, over 400 billion dollars were spent on DM-related health-care costs in the US. DM is characterized by high glucose levels due to insufficient production or inefficient utilization of insulin in Type 1 (T1-DM) or T2-DM, respectively.2-4 Regardless of the cause, a major complication of DM is the bone disease and increased fragility with a 32% increased risk of bone fractures in diabetic patients compared to non-diabetics.5-7
The mechanisms underlying DM-induced bone disease are complex and uncertain. T1-DM patients can exhibit low bone mass, measured as bone mineral density (BMD), whereas those with T2-DM can exhibit normal or even increased BMD. Yet, bone fragility is increased potentially due to accumulation of advanced glycation end products (AGEs) in collagen and other bone matrix proteins, which decreases bone toughness and resistance to fracture.8 Cortical bone micro-architectural deterioration secondary to reduced thickness and increased porosity is another feature of diabetic bone.7,9,10 Thus, bone fragility in DM is associated with deteriorated intrinsic as well as extrinsic properties of bone accompanied or not by changes in BMD.
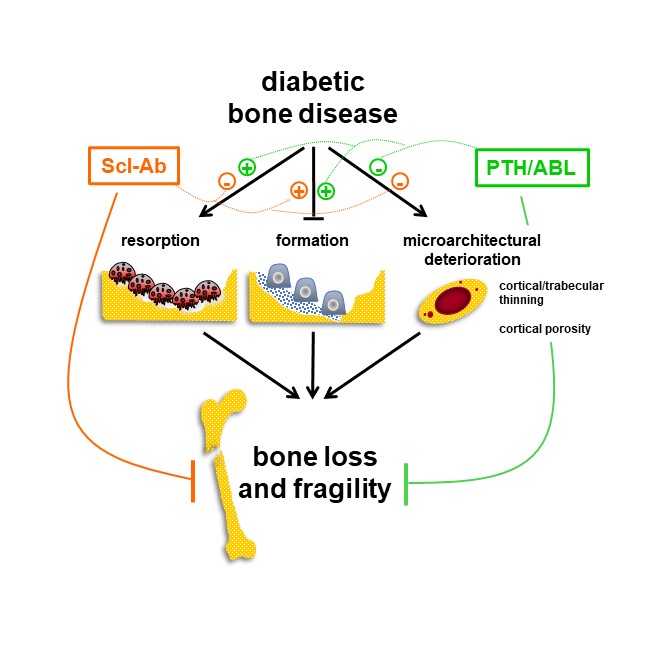
A common feature of DM-associated bone disease is reduced osteoblast number and function and low bone formation.6 However, the standard of care are anti-resorptive agents, bisphosphonates or denosumab, which stop bone loss but do not increase bone formation, thus failing to repair the deteriorated bone architecture.
Our contribution
We investigated the effectiveness of the three agents with bone anabolic properties approved by the FDA for the treatment of osteoporosis: teriparatide/parathyroid hormone 1-34 (PTH), abaloparatide/PTH-related peptide 1-34 (ABL), and romosozumab/anti-sclerostin antibody (Scl-Ab)], in restoring the weakened bone structure using a preclinical murine model of established T2-DM.
The current study reveals the signature of the diabetic bone at the tissue, cell, and transcriptome levels and demonstrates that all anabolic agents reverse it, rebuilding the bone lost with DM, increasing bone formation, correcting the elevated cortical porosity, and restoring bone strength. However, whereas PTH/ABL increased resorption, the Scl-Ab decreased it leading to further bone gain. In addition, all agents restored bone area, increased cortical thickness and corrected the weakened structural properties of diabetic bone. Furthermore, ABL and Scl-Ab increased toughness, a measure of the energy absorbed by bone before breaking, associated with fracture risk.
The impact
Our findings demonstrate the efficacy of agents that increase bone formation (regardless on their effects on resorption) to restore the damaging effects of diabetes on bone mass and structure, and to increase bone strength.
The bone protective effects of the anabolic therapies were independent of the diabetic status, as the loss of mineral, the damaged architecture, and the low activity of the bone forming cells were corrected despite the presence of overt hyperglycemia.
This pre-clinical mechanistic evidence suggests the need for revisiting the current treatment recommendations for bone fragility in diabetes and highlights the potential applicability of bone anabolic regimens to restore skeletal strength regardless of the presence or not of active diabetic disease.
REFERENCES
1 Sun, H. et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract 183, 109119, doi:10.1016/j.diabres.2021.109119 (2022).
2 DiMeglio, L. A., Evans-Molina, C. & Oram, R. A. Type 1 diabetes. Lancet 391, 2449-2462, doi:10.1016/S0140-6736(18)31320-5 (2018).
3 Chatterjee, S., Khunti, K. & Davies, M. J. Type 2 diabetes. Lancet 389, 2239-2251, doi:10.1016/S0140-6736(17)30058-2 (2017).
4 Association, A. D. 2. Classification and Diagnosis of Diabetes: Standards of Medical Care in Diabetes—2021. Diabetes Care 44, S15-S33, doi:10.2337/dc21-S002 (2020).
5 Wang, H., Ba, Y., Xing, Q. & Du, J. L. Diabetes mellitus and the risk of fractures at specific sites: a meta-analysis. BMJ Open 9, e024067, doi:10.1136/bmjopen-2018-024067 (2019).
6 Hofbauer, L. C. et al. Bone fragility in diabetes: novel concepts and clinical implications. Lancet Diabetes Endocrinol 10, 207-220, doi:10.1016/S2213-8587(21)00347-8 (2022).
7 Napoli, N. et al. Mechanisms of diabetes mellitus-induced bone fragility. Nat Rev Endocrinol 13, 208-219, doi:10.1038/nrendo.2016.153 (2017).
8 Shah, V. N. & DiMeglio, L. A. in Basic and Applied Bone Biology (Second Edition) (eds David B. Burr & Matthew R. Allen) 425-441 (Academic Press, 2019).
9 Samakkarnthai, P. et al. Determinants of Bone Material Strength and Cortical Porosity in Patients with Type 2 Diabetes Mellitus. J Clin Endocrinol Metab 105, doi:10.1210/clinem/dgaa388 (2020).
10 Sewing, L. et al. Bone Microarchitecture and Strength in Long-Standing Type 1 Diabetes. J Bone Miner Res 37, 837-847, doi:10.1002/jbmr.4517 (2022).
Follow the Topic
-
Bone Research
This journal highlights the breakthrough discoveries in basic and clinical aspects of bone biology, pathophysiology and regeneration, as well as other significant findings related to bone.


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in