Sensing the world: What determines the information update rate in echolocating bats
Published in Neuroscience

The world around us is changing from one moment to the next. In order to safely navigate our surroundings, we have to constantly perceive these changes and adapt accordingly. We often do this without even being aware, by looking around or sniffing the world. Different senses can be used to gather information and different animals use various sensory systems for this task (Fig. 1). By far, the most studied sensory modality is vision. By scanning our environment, we collect new information and update the image in front of us. The decision of how often to push the ‘refresh button’ is ultimately determined by each animals’ behavioral characteristics. The speed and frequency of visual update is determined by the speed of temporal processing and the required reaction time of each animal. The maximal speed at which the visual system can perceive flickering light as continuous defines the animal’s processing abilities and is referred to as the critical flicker fusion frequency (CFF)1. The higher the frequency the faster the performance. Snapping shrimps, for instance, have high CFFs of around 160 Hz. They live in turbid waters and require high update rates in order to avoid fast approaching objects or predators as well as to detect small prey2. Birds have been studied intensively in this field since flying requires rapid visual updating to avoid collisions. In air, birds are only outperformed by flying insects some of which exhibit CFFs of 200-300 Hz3 (e.g., Black fire beetles4 and Common evening brown butterfly5). But different birds perform differently according to their behavior. Agile birds like pied flycatchers exhibit very high visual update rates (138 Hz)6. They fly rapidly and need to update the information around them much more often than, say, domestic chickens, which spend most of their time on the ground in familiar environments and accordingly exhibit a much lower CFF (87 Hz)7. Raptors exhibit a range of visual acquisition capabilities depending on their specific hunting target (fast or slow moving prey)8. When vision is not an option, other sensory systems such as whisking, can be used to gather information about nearby objects. Rats use two sets of whiskers (macrovibrissae and microvibrissae) to sense their surroundings, sampling it at frequencies of 5-12 Hz9.
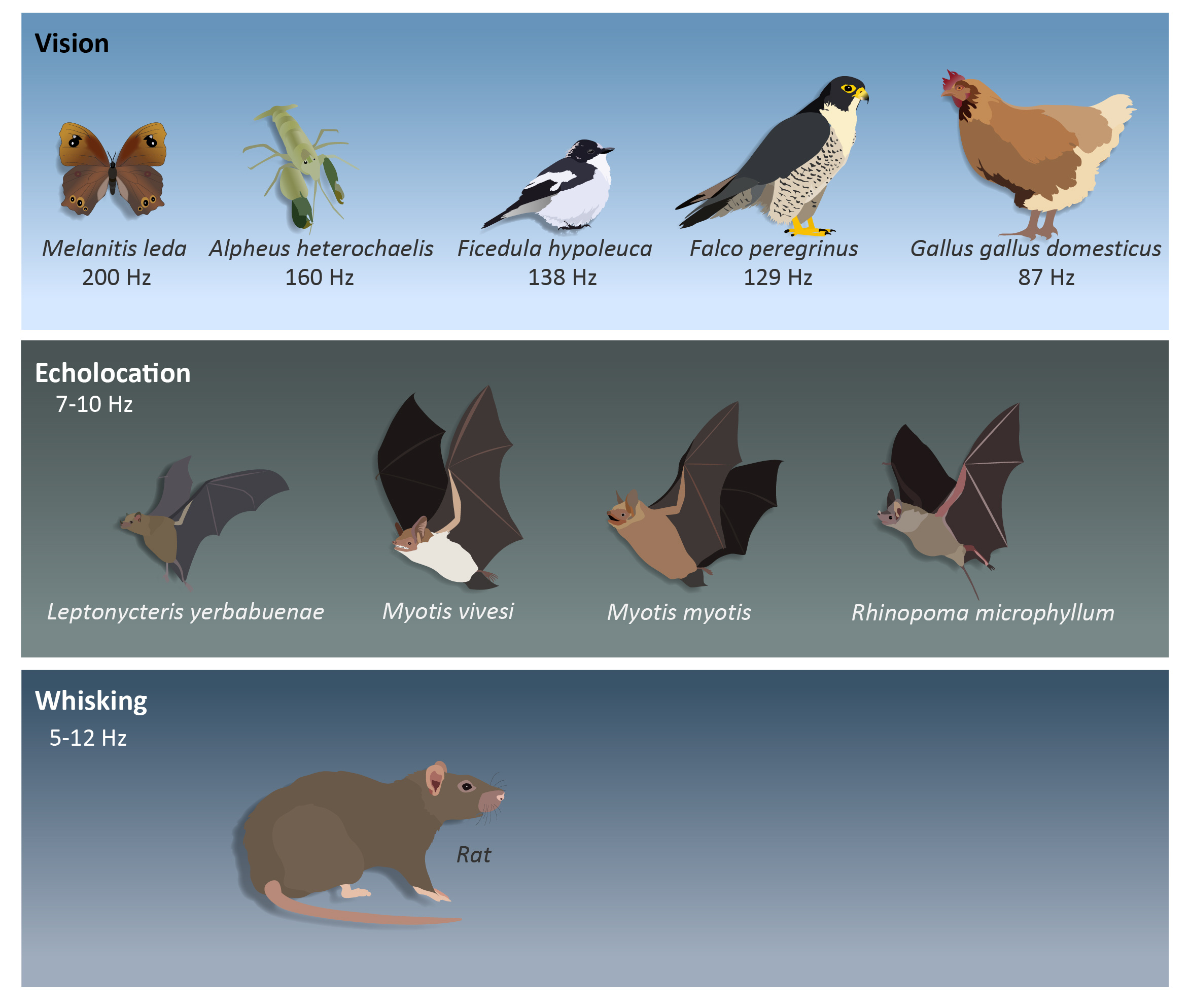
Figure 1: Sensory acquisition strategies.
Bats, being the only true flying mammals, mostly fly at night and rely on emitting ultrasonic signals to form a representation of the world around them. By controlling the properties of their emitted signals, such as their frequency, intensity, duration and directionality, bats can refine the acquisition resolution depending on the task at hand10. Adjusting the calling rate allows to control the temporal resolution with which bats perceive their world. Since flying expends a lot of energy, bats couple their call production with the wingbeat, conserving energy by performing two tasks at the price of one. This means that emitting calls too rapidly, and breaking this coupling, will inevitably lead to excess energy loss, but not calling often enough can lead to collisions with undetected obstacles or missing out on a juicy insect. This leads to the question - how often should a bat sample the environment using echolocation?
In our article “What determines the information update rate in echolocating bats”, recently published in Communications Biology, we try to answer this question.
Bats regulate the rate of sensory sampling by adjusting the inter-pulse interval (IPI), that is, the time-interval between two consecutive signals in the echolocation sequence. A useful way to assess sensory functionality is according to the sensory volume, i.e., the maximal volume of space in which an animal can detect objects (e.g., prey or obstacles)11,12. In echolocation, each emission covers a sensory volume that depends on the emitted signal’s parameters (e.g., its intensity and frequency). Depending on the bat’s flight speed, it will travel a certain distance between each two emissions, and if this distance exceeds the sensory volume of the echolocation signal, the bat will suffer from a ‘dead zone’ from which it will not receive any information (Fig. 2).
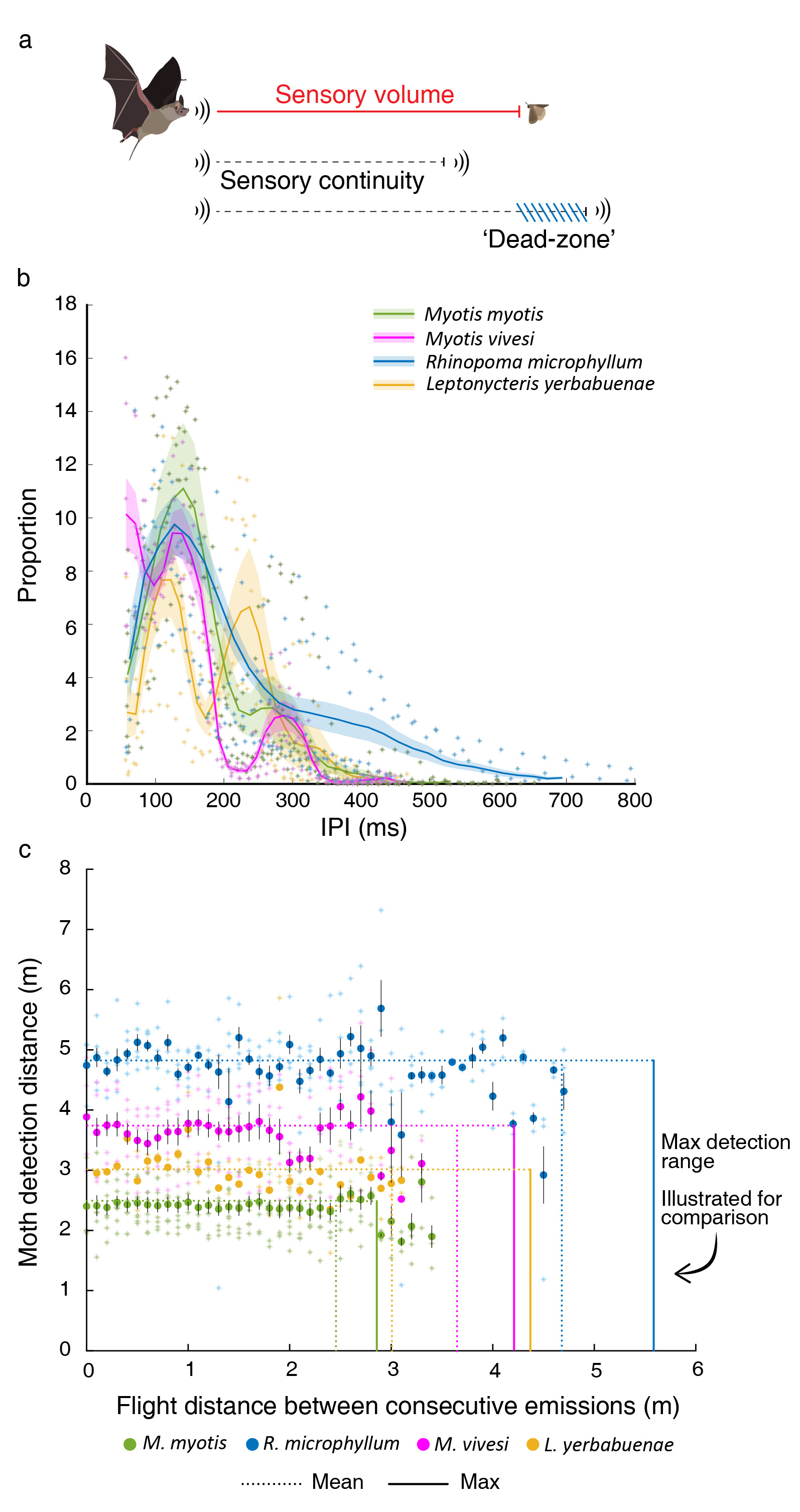
Figure 2: Sensing vs. movement. (a) Theoretical relationship between the maximum distance in which a bat can detect a moth (sensory volume) and the distance the bat travels between consecutive signals (flight distance). (b) IPI distribution. Lines represent the mean for all conspecifics and shaded areas represent the SE. Asterisks represent the individual data points for each species. N=25 bats from all four species in total (c) Moth detection distance (i.e., the 1-Dimensional equivalent of the sensory volume) compared to the flight distance between consecutive emissions of the four bat species. Circles represent the average detection distance for different flight distances based on the actual signal parameters and the flight speed and IPI. Asterisks represent the individual data points for each species. Error bars depict the SE. N=22 bats from all four species in total. Vertical lines show the mean (dotted line) and maximal (solid line) moth detection range for each species. Note that these lines represent values on the Y-axis, but are presented vertically to ease the comparison with the flight distance. These lines reveal that except for M. myotis the bats never fly between consecutive signals farther than the detection range thus maintaining sensing continuity for detecting a moth.
A commonly discussed hypothesis suggests that bats should always maintain an overlap between the sensory volumes covered by consecutive emissions13,14 thus maintaining sensory continuity. To test this we used on-board recordings - where we place a miniature microphone of the back of the bat - of four different species that greatly differ in their foraging strategies: the lesser long-nosed bat (Leptonycteris yerbabuenae) is a nectarivorous bat that flies long distances to reach the cacti fields were it feeds15,16; the greater mouse-tailed bat (Rhinopoma microphyllum), an aerial-hawker that forages in open spaces at relatively high altitudes17,18; the greater mouse-eared bat (Myotis myotis), a ground-gleaning bat which can also hunt on the wing19, and finally, the Mexican fish-eating bat (Myotis vivesi), a bat that specializes in hunting small oceanic fish and crustaceans above water at low altitudes20 (Fig. 3).
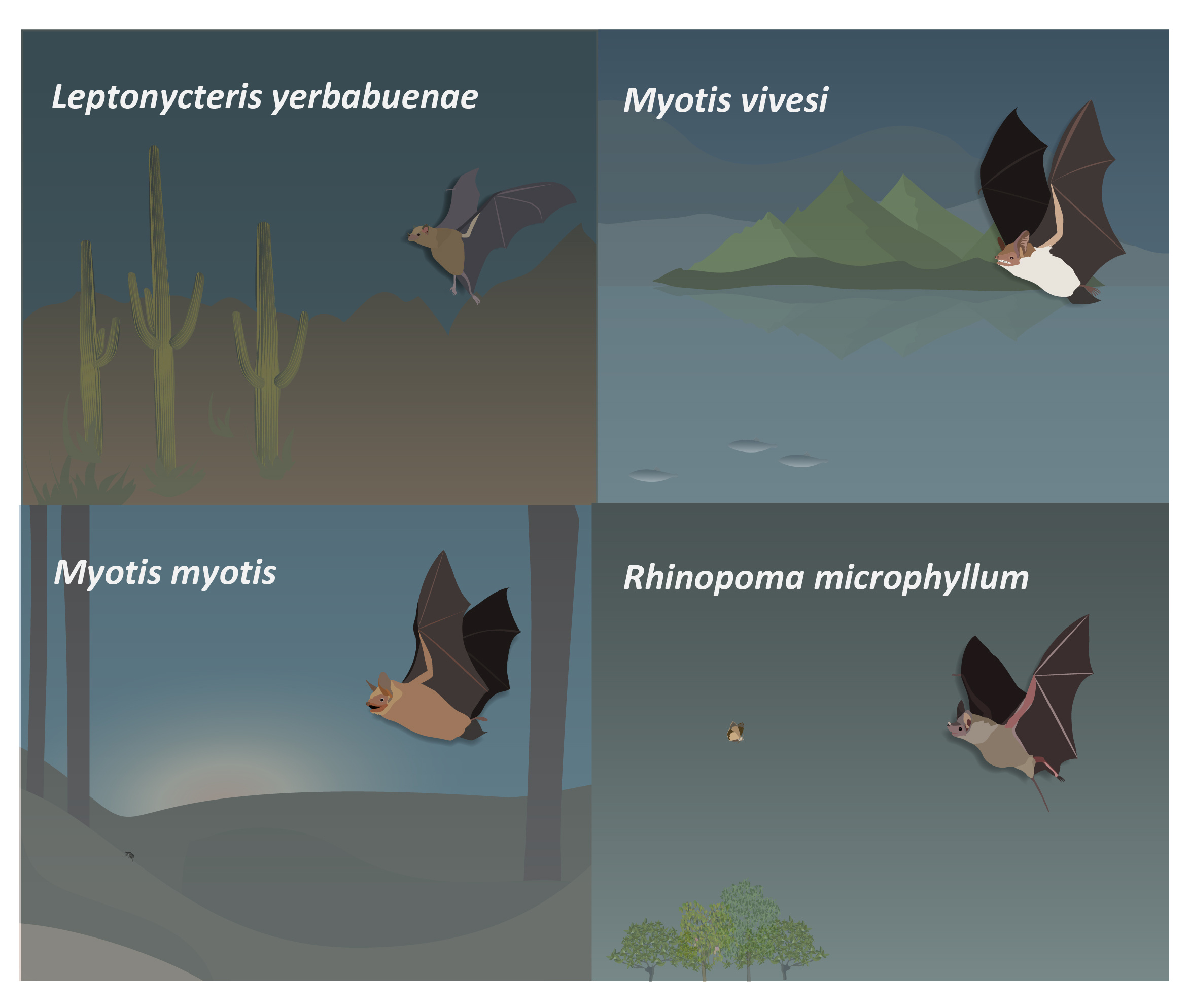
Figure 3: Foraging strategies. The lesser long-nosed bat (Leptonycteris yerbabuenae) is a nectarivorous bat that flies long distances to reach the cacti fields were it feeds; the greater mouse-tailed bat (Rhinopoma microphyllum), an aerial-hawker that forages in open spaces at relatively high altitudes; the greater mouse-eared bat (Myotis myotis), a ground-gleaning bat which can also hunt on the wing, and finally, the Mexican fish-eating bat (Myotis vivesi), a bat that specializes in hunting small oceanic fish and crustaceans above water at low altitudes
We found that all four species regulate their sampling behavior in very similar ways, despite their different styles of foraging. There was a strong positive correlation between the echolocation update rate and the signals’ peak-energy frequency and intensity and a negative correlation between the update rate and the flight speed and altitude. In general, all bats conformed to the continuity hypothesis, i.e., they maintained an overlap between the sensory volumes covered by consecutive emissions. However, we did not find any correlation between the distance traveled between consecutive calls and the sensory volume (Figure 2). This lead us to suggest that bats do maintain continuity, but that this is not what drives their adjustments of echolocation and that other factors such as flight altitude and their behavioral mode determine the sensory update rate. We identified two behavioral modes that are characterized by different calling and flight parameters. The first can be described as a low-attention mode and is characterized by faster flight, lower information update rate and weaker emissions at lower frequencies. This is typical of pure commuting - when a bat is moving from place to place without searching for prey. The second is a high-attention mode and is characterized by slower flight and a higher information update rate. We hypothesize that this high attention mode is related to searching for prey or attending potential obstacles such as background, conspecifics or the ground during commute. Bats are unique in their ability to finely control sensory temporal resolution, but by studying them, we can obtain insight about general questions, such as how and why animals regulate sensing.
References:
- Mankowska, N. D. et al. Critical flicker fusion frequency: a narrative review. Med. 57, 1–9 (2021).
- Kingston, A. C. N., Chappell, D. R. & Speiser, D. I. A snapping shrimp has the fastest vision of any aquatic animal. Biol. Lett. 16, (2020).
- Autrum, H. Etectrophysiological analysis of the visual systems in insects. Exp. Cell Res. 5, 426–439 (1958).
- Hammer, D. X., Schmitz, H., Schmitz, A., Grady Rylander, H. & Welch, A. J. Sensitivity threshold and response characteristics of infrared detection in the beetle Melanophila acuminata (Coleoptera: Buprestidae). Comp. Biochem. Physiol. - A Mol. Integr. Physiol. 128, 805–819 (2001).
- Chatterjee, P., Mohan, U., Krishnan, A. & Sane, S. P. Evolutionary constraints on flicker fusion frequency in Lepidoptera. J. Comp. Physiol. A Neuroethol. Sensory, Neural, Behav. Physiol. 206, 671–681 (2020).
- Boström, J. E. et al. Ultra-rapid vision in birds. PLoS One 11, 3–9 (2016).
- Lisney, T. J. et al. Behavioural assessment of flicker fusion frequency in chicken Gallus gallus domesticus. Vision Res. 51, 1324–1332 (2011).
- Potier, S., Lieuvin, M., Pfaff, M. & Kelber, A. How fast can raptors see? J. Exp. Biol. 223, (2020).
- Hartmann, M. J. Active sensing capabilities of the rat whisker system. Auton. Robots 11, 249–254 (2001).
- Nelson, M. E. & MacIver, M. A. Sensory acquisition in active sensing systems. J. Comp. Physiol. A Neuroethol. Sensory, Neural, Behav. Physiol. 192, 573–586 (2006).
- Lima, S. L. & Patrick A., Z. Towards a behavioral ecology of ecological landscapes. Trends Ecol. Evol. 11, 131–135 (1996).
- Stilz, W.-P. & Schnitzler, H.-U. Estimation of the acoustic range of bat echolocation for extended targets. J. Acoust. Soc. Am. 132, 1765–1775 (2012).
- Holderied, M. W. & Von Helversen, O. Echolocation range and wingbeat period match in aerial-hawking bats. Proc. R. Soc. B Biol. Sci. 270, 2293–2299 (2003).
- Surlykkc, A. et al. Echolocation in two very small bats from Thailand: Craseonycteris thonglongy and Myotis siligorensis. Behav. Ecol. Sociobiol. 33, 1–12 (1993).
- Goldshtein, A. et al. Reinforcement learning enables resource partitioning in foraging bats. Curr. Biol. 30, 4096–4102 (2020).
- Medellin, R. A. et al. Follow me: foraging distances of Leptonycteris yerbabuenae (Chiroptera: Phyllostomidae) in Sonora determined by fluorescent powder. J. Mammal. 99, 306–311 (2018).
- Norberg, U. M. & Rayner, J. M. V. Ecological morphology and flight in bats (Mammalia; Chiroptera): wing adaptations, flight performance, foraging strategy and echolocation. Philos. Trans. R. Soc. London. B, Biol. Sci. 316, 335–427 (1987).
- Levin, E., Yom-Tov, Y. & Barnea, A. Frequent summer nuptial flights of ants provide a primary food source for bats. Naturwissenschaften 96, 477–483 (2009).
- Arlettaz, R. Feeding behaviour and foraging strategy of free-living mouse-eared bats, Myotis myotis and Myotis blythii. Anim. Behav. 51, 1–11 (1996).
- Otálora-Ardila, A., Herrera, L. G., Juan Flores-Martínez, J. & Voigt, C. C. Marine and terrestrial food sources in the diet of the fish-eating myotis (Myotis vivesi). J. Mammal. 94, 1102–1110 (2013).
Follow the Topic
-
Communications Biology

An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Your space to connect: The Psychedelics Hub
A new Communities’ space to connect, collaborate, and explore research on Psychotherapy, Clinical Psychology, and Neuroscience!
Continue reading announcementRelated Collections
With Collections, you can get published faster and increase your visibility.
From RNA Detection to Molecular Mechanisms
Publishing Model: Open Access
Deadline: May 05, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in