Targeting intestinal macrophages as a therapeutic option in metabolic disease
Published in Healthcare & Nursing
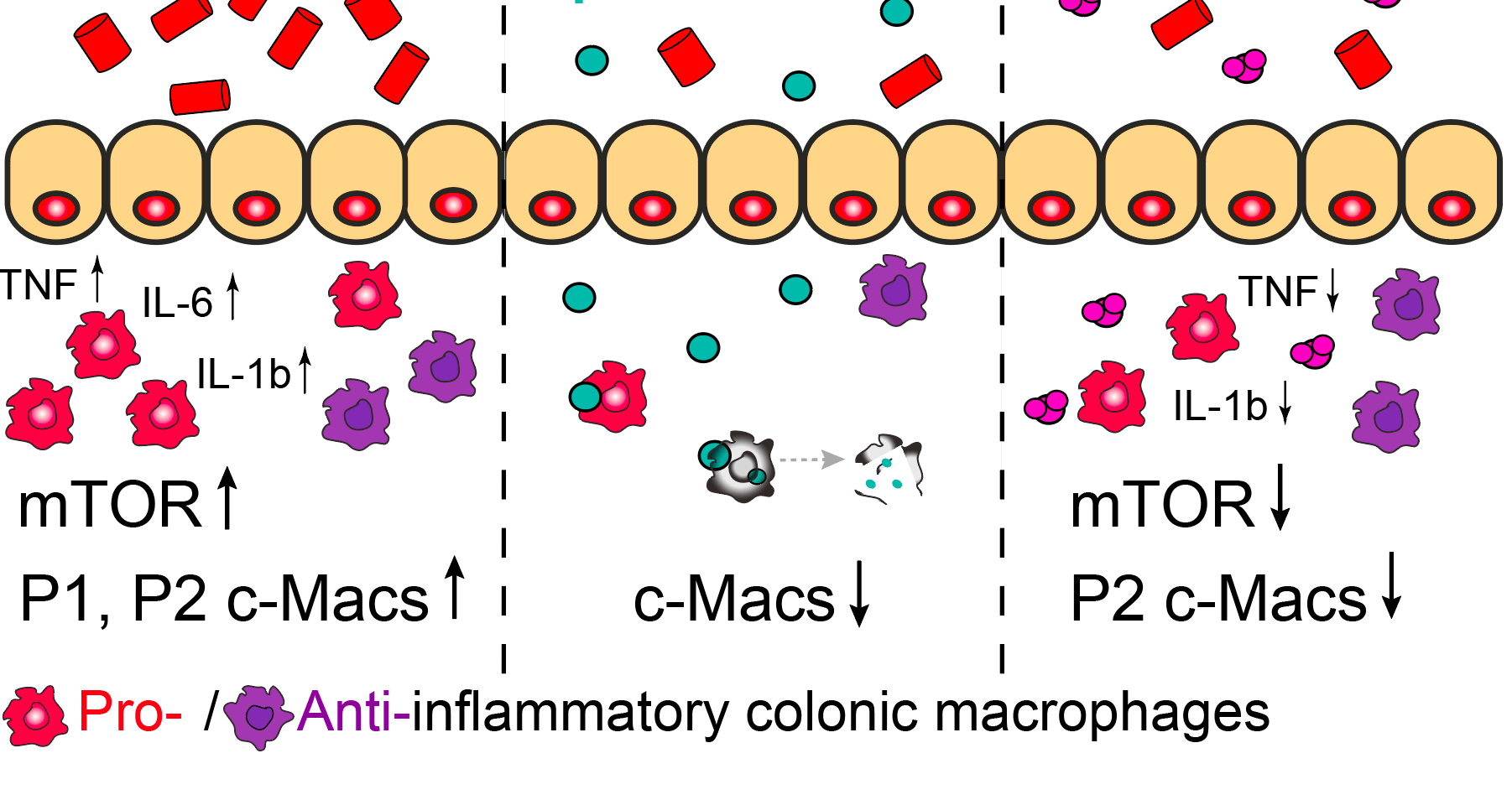
Inflammation in obesity and type 2 diabetes. Inflammation plays a significant role in metabolic diseases like obesity and type 2 diabetes. As the main drivers for low-grade inflammation in the context of obesity, adipose tissue macrophages have been intensively studied. However, the role intestinal macrophages play in glucose metabolism remains still unclear. The gut is the first organ constantly exposed to food antigens and gut microbiota. Therefore, we postulated that intestinal macrophages might initiate inflammation and glucose dysregulation in obesity.
Where does inflammation start in metabolic disease. In the gut, macrophages follow a differentiation trajectory. They differentiate from monocyte-derived/pro-inflammatory macrophage subpopulations (P1, P2), intermediate (P3), towards resident/anti-inflammatory macrophages (P4, P5)1,2. In inflammatory bowel diseases, this maturation process is disrupted, causing an accumulation in pro-inflammatory intestinal macrophages2-4. In the context of obesity and diabetes, an inflammatory tone in colonic macrophages has been described5. However, a distinct characterization of intestinal macrophage subpopulations, their activation status, and their impact on glycemic control remained elusive.
First important observations. Interestingly, we found that monocyte-derived/pro-inflammatory macrophages (P1, P2) were increased already after short-term high-fat diet feeding in mice. We were able to confirm our findings also in human obese subjects compared to lean controls, thus bridging our basic research to the clinic context6.
Further questions to be answered. Next, in our mouse study, we were interested in answering unsolved questions that are difficult to answer in the human setup.
- Is obesity-induced intestinal inflammation dependent on the microbiota? Yes, the gut microbiota is essential for macrophage influx into the gut and glucose intolerance.
- Does the source of the fat or fiber content of the diet influence tissue inflammation or glucose metabolism? The fat source modulates the magnitude of intestinal and adipose tissue inflammation, glucose intolerance, and insulin resistance.
- Can we ameliorate obesity-induced glucose homeostasis by depleting macrophages systemically and colon-specifically? Yes, a dose-dependent depletion of macrophages causes gradual improvements in metabolic disease. Also colon-specific macrophage depletion leads to improved glucose tolerance and insulin sensitivity.
- What are the mechanistic pathways involved in intestinal macrophage activation in obesity? High-fat diet induces an interferon response in intestinal macrophages, altered mitochondrial metabolism, which converge in mTOR as a common regulator. mTOR inhibition in colonic macrophages restores insulin secretion capacity in obesity.
Important collaborations. During our study, we collaborated with different research groups worldwide. I went to Marseille, France, for an expert exchange to validate isolating and analyzing intestinal macrophages. Prof. Sandrine Henri, Prof. Bernard Malissen, and Dr. Anna Baranska from the Centre d`Immunologie Marseille-Luminy were a great help in method optimization and led us in the right direction at a crucial early timepoint during the study. Furthermore, we collaborated with Prof. Stephanie C. Ganal-Vonarburg and Dr. Catherine Mooser, Dr. Bahtiyar Yilmaz, and Nerea Fernandez Trigo from the Inselspital in Bern, to answer important questions regarding the involvement of the microbiota. In addition, we enjoyed the interaction with several gastroenterology teams, such as from the University Hospital Basel, Clarunis, the University Center for Gastrointestinal and Liver Diseases in Basel, Lindenhof Hospital in Bern, and the MagenDarm Office in Basel. This allowed us to confirm our preclinical data in human obese patients.
Take home message. Obesity triggers an inflammatory response in the gut, causing the accumulation of pro-inflammatory intestinal macrophages. Importantly, colon-specific macrophage depletion or inhibition of colonic macrophage activation by blocking mTOR-signaling improves glycemic control and/or β-cell function. Overall, better knowledge regarding the initiation of inflammation in obesity and diabetes might lead to new immuno-modulatory strategies to prevent inflammation and glucose dysregulation early in the disease process.
To get more insights into our study, read our full article “Targeting colonic macrophages improves glycemic control in high-fat diet-induced obesity”, published in Nature Communications Biology on April 19, 2022 (10.1038/s42003-022-03305-z. or https://www.nature.com/articles/s42003-022-03305-z).
References
- Tamoutounour, S.et al. Origins and functional specialization of macrophages and of conventional and monocyte-derived dendritic cells in mouse skin. Immunity 39, 925-938, doi:10.1016/j.immuni.2013.10.004 (2013).
- Tamoutounour, S. et al. CD64 distinguishes macrophages from dendritic cells in the gut and reveals the Th1-inducing role of mesenteric lymph node macrophages during colitis. Eur J Immunol 42, 3150-3166, doi:10.1002/eji.201242847 (2012).
- Bain, C. C. et al. Resident and pro-inflammatory macrophages in the colon represent alternative context-dependent fates of the same Ly6Chi monocyte precursors. Mucosal Immunol 6, 498-510, doi:10.1038/mi.2012.89 (2013)
- Platt, A. M., Bain, C. C., Bordon, Y., Sester, D. P. & Mowat, A. M. An independent subset of TLR expressing CCR2-dependent macrophages promotes colonic inflammation. J Immunol 184, 6843-6854, doi:10.4049/jimmunol.0903987 (2010).
- Kawano, Y. et al. Colonic Pro-inflammatory Macrophages Cause Insulin Resistance in an Intestinal Ccl2/Ccr2-Dependent Manner. Cell Metab 24, 295-310, doi:10.1016/j.cmet.2016.07.009 (2016)
- Rohm, T. V. et al. Obesity in Humans Is Characterized by Gut Inflammation as Shown by Pro-Inflammatory Intestinal Macrophage Accumulation. Front Immunol 12, 668654, doi:10.3389/fimmu.2021.668654 (2021). https://pubmed.ncbi.nlm.nih.gov/34054838/or https://www.frontiersin.org/articles/10.3389/fimmu.2021.668654/full.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
Mechanistic insights into human host and microbiome interactions
Publishing Model: Open Access
Deadline: May 31, 2026
Advances in neurodegenerative diseases
Publishing Model: Hybrid
Deadline: Jun 30, 2026





Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in