The role of cell therapy in spinal cord injury: the application of in vivo bioluminescence imaging
Published in Bioengineering & Biotechnology
Given the marked challenge in regenerating the central nervous system following injury, patients with spinal cord injury (SCI) exhibit mostly irreversible impairment of motor, sensory, and autonomic functions. It is estimated that the annual incidence of SCI is approximately 5,000 in Japan and 18,000 in the United States. However, approaches for treating patients with SCI remain limited. In recent years, clinical trials of granulocyte-colony stimulating factor and hepatocyte growth factor in acute SCI have been conducted in Japan. Clinical trials are underway to examine the potential of transplanting neural stem/progenitor cells (NS/PCs) derived from human-induced pluripotent stem cells (iPSCs) for subacute SCI.
Pluripotent stem cells, including embryonic stem cells (ESCs) and iPSCs, are a promising source of NS/PCs and an attractive approach for promoting functional recovery. NS/PCs replace damaged tissue and differentiate into three neural lineage cell subtypes: neurones, astrocytes, and oligodendrocytes. Transplantation of human ESC-derived cells remains ethically controversial in clinical practice, and our laboratory has been working on realising human iPSC-derived NS/PC (hiPSC-NS/PC) transplantation therapy since the successful generation of iPSCs in 2006. We demonstrated the efficacy of hiPSC-NS/PC transplantation in treating subacute SCI in rodents and non-human primates. Based on these studies, we launched a clinical study (Phase I/IIa) of hiPSC-NS/PC transplantation for subacute SCI (Sugai et al., Regenerative Therapy, 2021).
Accumulated evidence from animal experiments utilising hiPSC-NS/PC as the cell source has demonstrated the efficacy and safety of cell transplantation therapy. Efficacy is mainly related to motor function recovery, and concerns regarding the risk of tumourigenesis or immune rejection must be considered. In addition to the above-mentioned three lineages of neural cells, some cell populations possibly maintain an undifferentiated state and self-proliferation. To improve the efficacy and reduce the risk of tumourigenesis, inhibiting Notch signalling of stem cells by gamma-secretase inhibitor (GSI) can be considered a valuable strategy, promoting differentiation into mature neurones (Okubo et al., Stem Cell Reports, 2016). This approach could be beneficial in chronic SCI animal experiments via axonal regrowth involving phosphorylation of p38 mitogen-activated protein kinase. Emerging methods attempt to enhance stem cell therapy, and determining which method is superior among various treatment strategies or whether combination therapy affords a synergistic effect remains an ongoing challenge. As we hope to apply cell therapy to chronic SCI in the future, further knowledge regarding therapeutic conditions will be necessary.
Stem cell therapy is distinct from conventional drug therapies, which aim to attenuate secondary injury of the spinal cord during the acute phase. NS/PCs are speculated to exhibit both cell-autonomous and non-cell-autonomous effects. The former includes synapse formation between host circuit neurones and graft neurones, reconstruction of neural circuits to their former structure, and remyelination of demyelinated axons with oligodendrocytes derived from grafted cells. The latter includes trophic effects released from grafted cells.
However, a limited number of reports have demonstrated how each of these mechanisms plays a role in functional recovery. In this context, we have also conducted basic research using chemogenetics to further improve therapeutic efficacy and elucidate mechanisms underlying the beneficial effects of cell therapy on the injured spinal cord.
In recent years, advances in the field of neuroscience have led to the development of various tools for controlling neuronal or glial cell activity in vivo. One such tool is the designer receptor exclusively activated by designer drugs (DREADD). These genetically modified G-protein-coupled receptors employ compounds such as clozapine N-oxide to selectively control neural activity in target neurones.
To confirm that graft-derived neurones serve as a neuronal relay to transmit an excitatory electrical signal across the lesion area, cell type-specific silencing has been previously examined. Indeed, we recently reported that selective inhibition of the neuronal activity of grafted cells by the inhibitory DREADD system (hM4Di) temporarily suppresses motor function recovery (Kitagawa et al., Stem Cell Reports, 2022). According to another study from our laboratory, longitudinal stimulation of grafted cells by an excitatory DREADD system (hM3Dq) could induce significant motor function recovery when compared with cell transplantation therapy alone, indicating that activating neural activity of the transplanted cells can improve the host motor function (Kawai et al., Cell Reports, 2021). This finding expands the possibility of enhancing the effect of cell therapy using a combination of transcranial magnetic stimulation and cell therapy.
In the present study, we successfully developed a monitoring system to visualise neuronal activity in living animals. Genetically encoded calcium indicator allows the visualisation of neuronal activity, previously utilised in combination with optogenetics by other groups (Ceto et al., Cell Stem Cell, 2020). This approach may reflect real-time neuronal activity, but monitoring cells deep within an animal remains technically challenging. We are familiar with in vivobioluminescence imaging using firefly luciferase (ffLuc), which is genetically encoded into grafted cells and can be consistently observed from outside the animal. We used this tool to assess cell survival. Herein, we further applied this strategy to visualise the neuronal activity of grafted cells, taking advantage of the newly developed techniques AkaBLI and E-SARE. Various experiments can be conducted using this method. We detected the longitudinal elevation of neuronal activity in the grafted cells in vivo.
Furthermore, combined with the DREADD system, we could survey how injured corticospinal neurones integrated with transplanted cells. The effect of corticospinal tract activation on graft activity increased gradually after transplantation, indicating that the connection between host corticospinal circuit neurones and grafted neurones has become increasingly rigorous (Figure 1). We speculate that this system will help estimate the therapeutic effects of various stem cell therapies in the future.
Based on the findings of these studies, we intend to pursue further research to improve cell transplantation therapy and chronic SCI, for which adequate therapeutic effects are yet to be established.
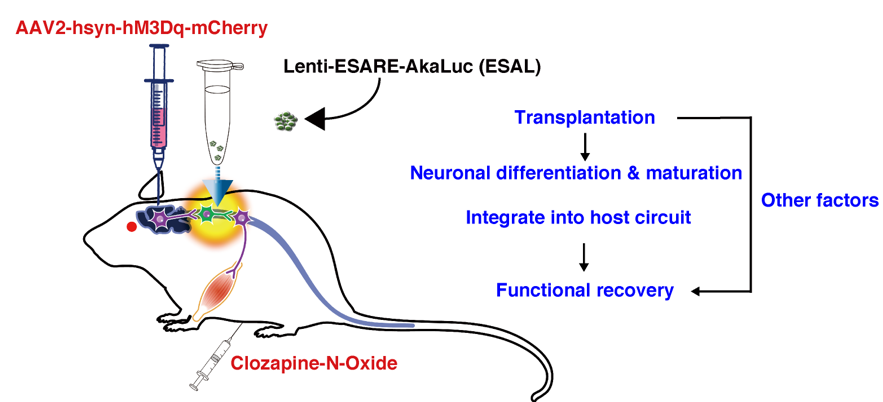
Figure 1: This E-SARE-AkaLuc (ESAL) system was used to detect the graft activity responding to host CST activation; it only shows the synapse linking upstream of graft neurones. Assessing the graft-to-host interaction downstream of the graft-derived neurons will be needed in the future. CST, corticospinal tract.
Follow the Topic
-
Communications Biology
An open access journal from Nature Portfolio publishing high-quality research, reviews and commentary in all areas of the biological sciences, representing significant advances and bringing new biological insight to a specialized area of research.
Related Collections
With Collections, you can get published faster and increase your visibility.
DNA repair and human disease
Publishing Model: Hybrid
Deadline: Oct 31, 2026
Cell death and inflammatory signalling
Publishing Model: Hybrid
Deadline: Oct 28, 2026



Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in