Transcranial electrical stimulation for the treatment of obsessive–compulsive disorder
Published in Neuroscience and General & Internal Medicine
Obsessive–compulsive disorder (OCD) is a condition with substantial disability and subclinical obsessive–compulsive behaviors affect up to 1 billion people globally. Transcranial electrical stimulation (tES) is emerging as a potential treatment for OCD, yet its effectiveness is uncertain due to the limited number of randomized controlled trials conducted so far. In this analysis study, we applied 3 meta-analytic approaches to evaluate the pooled therapeutic effect size of included randomized controlled trials (ntDCS = 15, ntACS = 1) (pair-wise meta-analysis), compare the efficacy of various tES interventions (network meta-analysis), and explore how treatment effect size correlates with induced electrical field in the target brain regions (meta-modeling). With these 3 distinct meta-analytic approaches, we first show a significant moderate effect of tES interventions (standardized mean difference 0.61; 95% confidence interval [0.37 to 0.85]; P < 0.001) as a whole on reducing OCD severity, with larger therapeutic effects of twice-daily intervention, and with other stimulation parameters also moderating efficacy. Next, we identify specific tES interventions with superior indicated and estimated effects, including entraining alpha frequency alternating stimulation of the medial prefrontal cortex, excitability-diminishing cathodal stimulation over the supplementary motor area and orbitofrontal cortex, and excitability-enhancing anodal stimulation of the lateral prefrontal cortex. Finally, we show based on computational approaches that longer stimulation targeting the medial prefrontal and frontopolar cortices has greater therapeutic effects and suggest an optimized intervention accordingly.
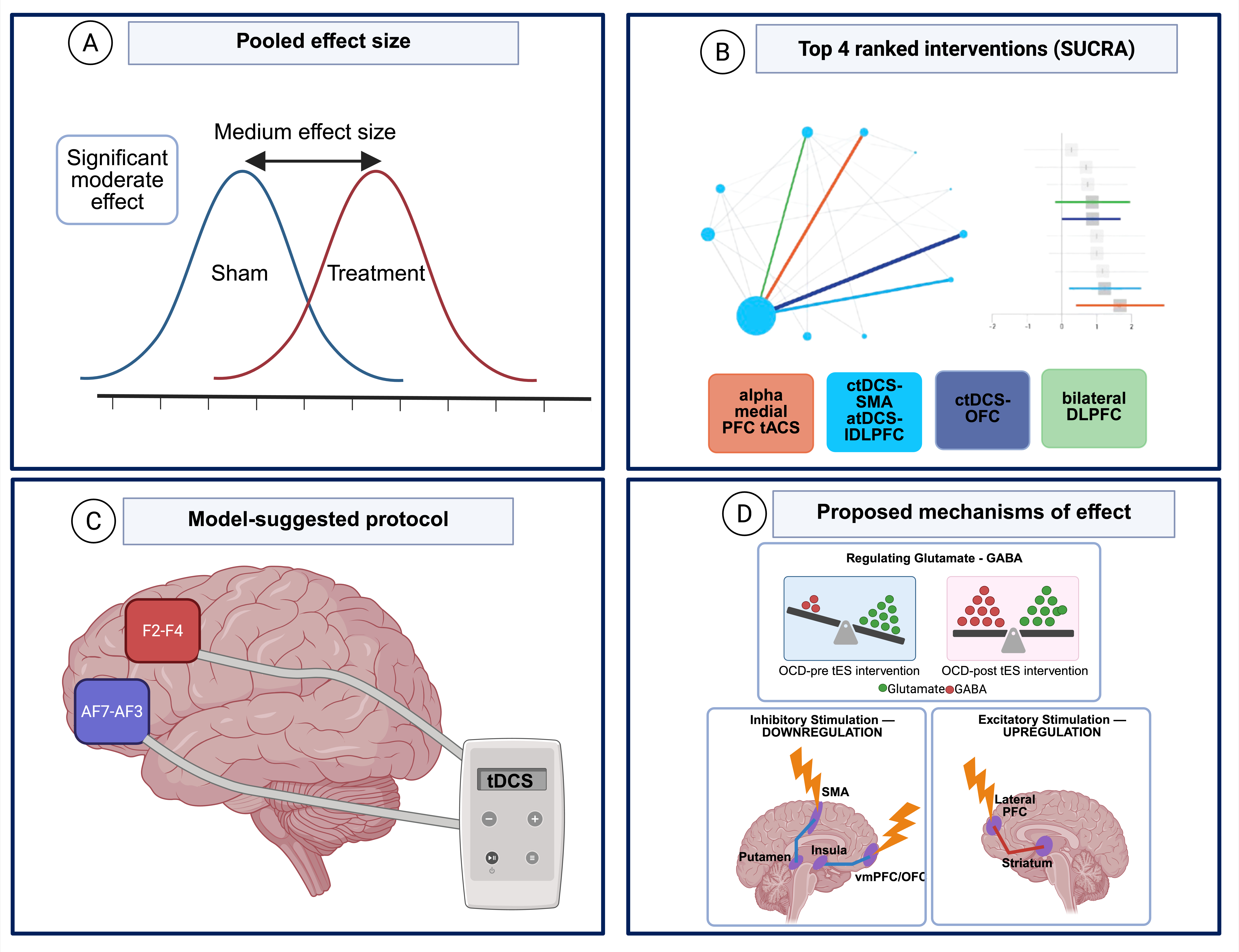
We also discussed other potentially effective tES interventions according to the latest neurobiological theory of OCD. The results contribute to establishing the clinical efficacy of tES for OCD treatment while suggesting future studies to further evaluate current and future approaches to enhance the efficacy of interventions. This study is published in Nature Mental Health and is open access to readers: https://www.nature.com/articles/s44220-026-00590-z

Follow the Topic
-
Nature Mental Health
This journal takes an expansive view of the relationship between mental health and human health. It brings together innovative investigation of the neurobiological and psychological factors that underpin psychiatric disorders to contemporary work examining the effects of public health crises.
Related Collections
With Collections, you can get published faster and increase your visibility.
Health outcomes of adversity in early life
Publishing Model: Hybrid
Deadline: Sep 30, 2026
The expanding therapeutic landscape of GLP 1 receptor agonists
Publishing Model: Hybrid
Deadline: Jan 23, 2027




Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in