Unravelling the genetic puzzle of COVID-19 vaccine response
Published in Protocols & Methods, Genetics & Genomics, and Biomedical Research
What prompted us to initiate this project?
In the first few months after vaccine administration, it quickly became clear that individual response to the drug was different, particularly in the levels of antibodies detected in the blood.
We and many other scientists therefore wondered about the genetic bases underlying this variability in response. Indeed, host genetic factors are believed to contribute to this diversity, as this has been observed for other vaccines as well.
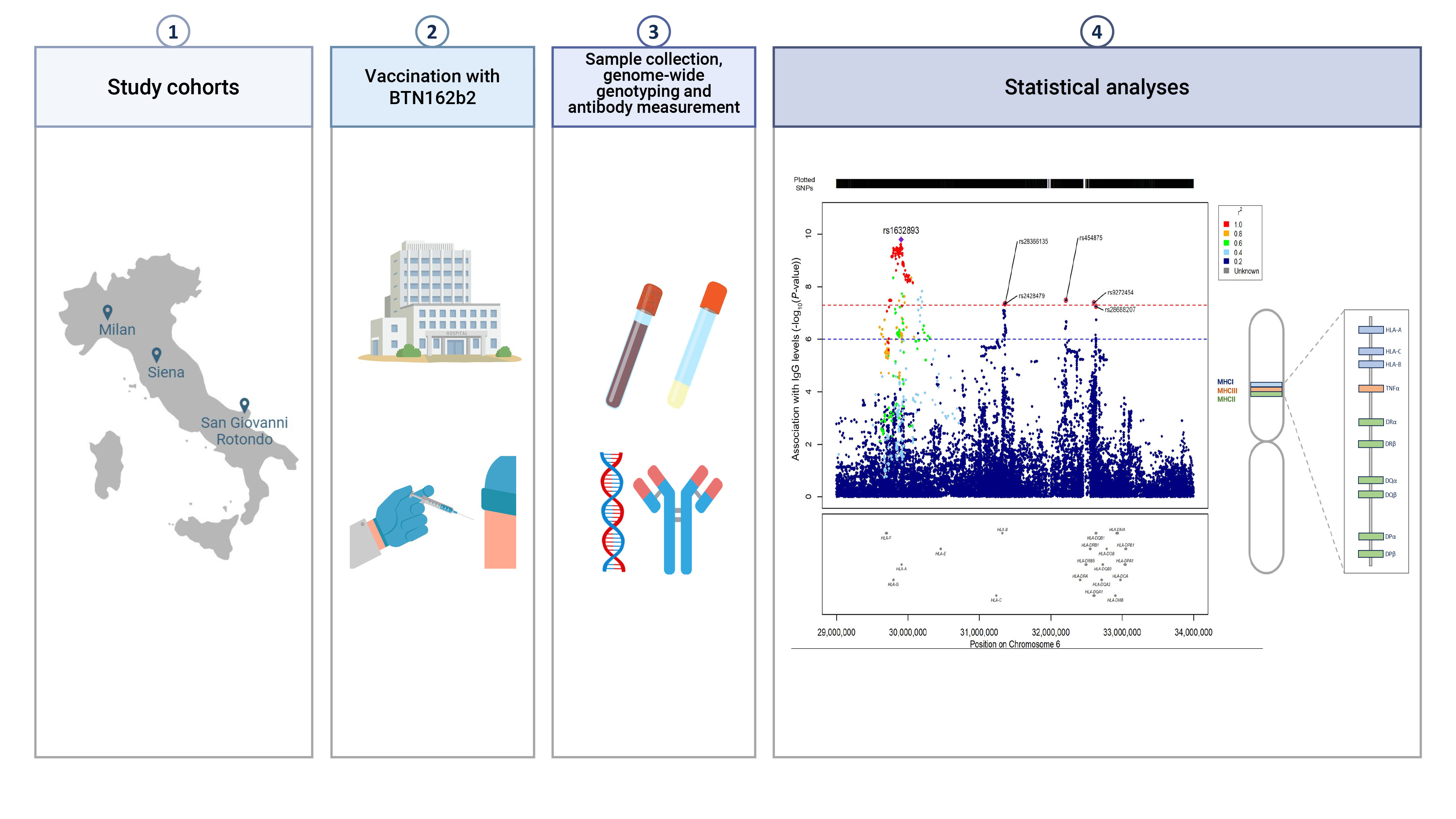
At that time, we and our colleagues from the Azienda Ospedaliero-Universitaria Senese in Siena, and Fondazione IRCCS Casa Sollievo della Sofferenza, San Giovanni Rotondo (FG) were already collaborating on the genetics of COVID-19 disease, and we immediately seized the opportunity to start investigating the genetics of the response to vaccine, as well, in the hospital workers vaccinated in these two hospitals. To increase the sample size, we proposed other colleagues from Fondazione IRCCS Istituto Neurologico Carlo Besta in Milan to contribute to the study. Thus, the willingness to collaborate for an important common goal and the foresight in biobanking valuable material, by some physicians and researchers, was crucial to the realization of the study. No less decisive was also the role of the funding agency (Istituto Buddista Italiano Soka Gakkai), which allowed the original research design to be modified on the fly, thus enabling the study of the genetics of vaccines, which did not yet exist at the time of the grant application.
Therefore, we collected blood samples and clinical data from 1,351 individuals who received two doses of the BNT162b2 vaccine (Pfizer-BioNTech). One month after complete vaccination, we measured the levels of anti-SARS-CoV-2 antibodies in the serum of the recruited individuals, and, as expected, large differences were found among the vaccinees. In addition, we determined the genetic constitution of these subjects, by analyzing more than seven million germline variants in their genomic DNA. Finally, statistical genetics analyses were performed to identify germline variants that correlated with antibody levels, taking into account for potential confounding variables (i.e., sex, age at vaccination, recruiting center, the first five principal components, and the time interval between the second dose of vaccine and serological testing.
What were our main findings?
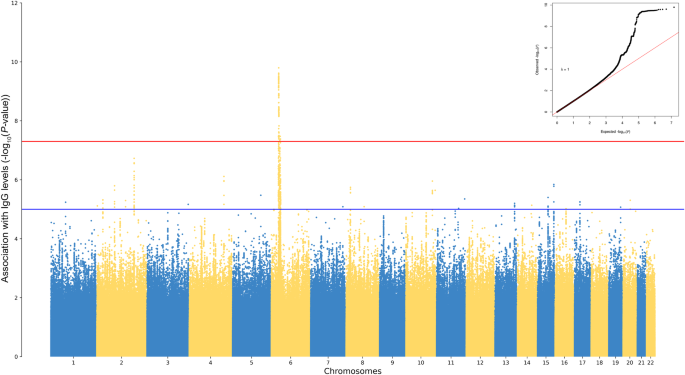
Our genome-wide association analysis consisted of a multivariable linear regression between normalized antibody levels and 7,339,393 imputed variants, in 1,351 individuals of European origin who received the two doses of the Pfizer-BioNTech vaccine.
We identified a statistically significant locus on chromosome 6, within the HLA locus, with multiple variants associated with immunoglobuline G (IgG) levels. A deeper investigation revealed additional nearby association peaks, in several HLA genes, suggesting the existence of multiple independent loci influencing the antibody response. In detail, we observed that subjects with particular variants ahead of the HLA-A and HLA-B genes had higher levels of anti-SARS-CoV-2 antibodies, while lower IgG levels were measured individuals with other variants in the introns of HLA-DQA1 and HLA-DQB1 genes. Also analyzing HLA alleles (i.e. combinations of variants), we observed significant associations with antibody levels. Of note, our analyses indicated that HLA alleles belonging to both MHC Class I (e.g. HLA-A*03:01), and Class II (HLA-DQB1*06:01) were involved in modulating the serum level of anti-SARS-CoV-2 IgG after vaccination.
The importance of independent validation
At the same time our group was working on these analyses, a British group was conducting a similar study, but on subjects vaccinated predominantly with the AZD1222, Oxford-AstraZeneca vaccine. Although they preceded us with their publication, we were excited that our results independently confirmed what colleagues had discovered, namely, the importance of the HLA locus in modulating the response to anti-COVID-19 vaccination. Independent validation of the role of specific alleles of HLA-DQB1 gene is important when we consider that genetic studies in different populations, with specific frequencies of genetic variants, do not always yield the same results.
Any Surprising Findings?
Our study, however, not only confirms the association reported in the British study. Indeed, we observed that MHC Class I molecules associate with anti-spike IgG after completion of the vaccine cycle. While our manuscript was under review, another single-center Italian study also reported an association of HLA-A*03:01 with antibody levels. Further investigation is needed to deepen the association between the HLA-A*03:01 allele, along with other MHC class I molecules, and the CD8+ T cell response following COVID-19 vaccination. Indeed, we can only speculate that CD8+ follicular T cells may play a helper role in enhancing B cell class-switch antibody production and increasing the antibody response, both through HLA binding mechanisms and non-HLA related effects such as the increased secretion of specific cytokines. This hypothesis is supported by the observation that mRNA vaccines against SARS-CoV-2 elicit neutralizing antibodies and stimulate CD8+ T cell responses.
What’s next?
Our study supports the role of the HLA locus in modulating antibody levels after anti-COVID-19 vaccination by highlighting differences in genetic determinants among populations. These findings underscore the importance of investigating genetic associations in diverse populations. We are already in touch with UK colleagues to share our data and collaborate on new analyses that will combine information from different cohorts around the world.
In conclusion, our results contribute to the understanding of the genetic basis of vaccine response, particularly in the context of COVID-19 vaccination. These insights pave the way for precision vaccination approaches tailored to individual genetic profiles. Moreover, our findings may stimulate further research into the genetic determinants of vaccine response in different diseases, advancing the field of vaccinogenomics.
Follow the Topic
-
Communications Medicine
A selective open access journal from Nature Portfolio publishing high-quality research, reviews and commentary across all clinical, translational, and public health research fields.
Related Collections
With Collections, you can get published faster and increase your visibility.
Healthy Aging
Publishing Model: Open Access
Deadline: Jun 01, 2026
Public health and health governance in China
Publishing Model: Open Access
Deadline: Jul 31, 2026


Please sign in or register for FREE
If you are a registered user on Research Communities by Springer Nature, please sign in